Abstract
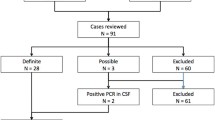
Herpes simplex virus (HSV) encephalitis is associated with a high risk of mortality and sequelae, and early diagnosis and treatment in the emergency department are necessary. However, most patients present with non-specific febrile, acute neurologic impairment; this may lead clinicians to overlook the diagnosis of HSV encephalitis. We aimed to identify which data collected in the first hours in a medical setting were associated with the diagnosis of HSV encephalitis. We conducted a multicenter retrospective case–control study in four French public hospitals from 2007 to 2013. The cases were the adult patients who received a confirmed diagnosis of HSV encephalitis. The controls were all the patients who attended the emergency department of Grenoble hospital with a febrile acute neurologic impairment, without HSV detection by polymerase chain reaction (PCR) in the cerebrospinal fluid (CSF), in 2012 and 2013. A multivariable logistic model was elaborated to estimate factors significantly associated with HSV encephalitis. Finally, an HSV probability score was derived from the logistic model. We identified 36 cases and 103 controls. Factors independently associated with HSV encephalitis were the absence of past neurological history (odds ratio [OR] 6.25 [95 % confidence interval (CI): 2.22–16.7]), the occurrence of seizure (OR 8.09 [95 % CI: 2.73–23.94]), a systolic blood pressure ≥140 mmHg (OR 5.11 [95 % CI: 1.77–14.77]), and a C-reactive protein <10 mg/L (OR 9.27 [95 % CI: 2.98–28.88]). An HSV probability score was calculated summing the value attributed to each independent factor. HSV encephalitis diagnosis may benefit from the use of this score based upon some easily accessible data. However, diagnostic evocation and probabilistic treatment must remain the rule.
Similar content being viewed by others
References
Mailles A, Stahl JP; Steering Committee and Investigators Group (2009) Infectious encephalitis in France in 2007: a national prospective study. Clin Infect Dis 49:1838–1847
Gordon B, Selnes OA, Hart J Jr, Hanley DF, Whitley RJ (1990) Long-term cognitive sequelae of acyclovir-treated herpes simplex encephalitis. Arch Neurol 47:646–647
Klapper PE, Cleator GM (1998) European guidelines for diagnosis and management of patients with suspected herpes simplex encephalitis. Clin Microbiol Infect 4:178–180
Greenland S (2004) Model-based estimation of relative risks and other epidemiologic measures in studies of common outcomes and in case–control studies. Am J Epidemiol 160:301–305
Raschilas F, Wolff M, Delatour F et al (2002) Outcome of and prognostic factors for herpes simplex encephalitis in adult patients: results of a multicenter study. Clin Infect Dis 35:254–260
Sili U, Kaya A, Mert A; HSV Encephalitis Study Group (2014) Herpes simplex virus encephalitis: clinical manifestations, diagnosis and outcome in 106 adult patients. J Clin Virol 60:112–118
McGrath N, Anderson NE, Croxson MC, Powell KF (1997) Herpes simplex encephalitis treated with acyclovir: diagnosis and long term outcome. J Neurol Neurosurg Psychiatry 63:321–326
Tan IL, McArthur JC, Venkatesan A, Nath A (2012) Atypical manifestations and poor outcome of herpes simplex encephalitis in the immunocompromised. Neurology 79:2125–2132
Chavanet P, Schaller C, Levy C et al (2007) Performance of a predictive rule to distinguish bacterial and viral meningitis. J Infect 54:328–336
Kulik DM, Uleryk EM, Maguire JL (2013) Does this child have bacterial meningitis? A systematic review of clinical prediction rules for children with suspected bacterial meningitis. J Emerg Med 45:508–519
Thwaites GE, Chau TT, Stepniewska K et al (2002) Diagnosis of adult tuberculous meningitis by use of clinical and laboratory features. Lancet 360:1287–1292
Cohn KA, Thompson AD, Shah SS et al (2012) Validation of a clinical prediction rule to distinguish Lyme meningitis from aseptic meningitis. Pediatrics 129:e46–e53
Lewthwaite P, Begum A, Ooi MH et al (2010) Disability after encephalitis: development and validation of a new outcome score. Bull World Health Organ 88:584–592
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
None.
Conflict of interest
None.
Ethical approval
The protocol has been declared to the French data protection authority (Commission Nationale de l’Informatique et des Libertés). The Institutional Ethics Review Board waived the requirement for informed consent and approved the study protocol.
Informed consent
Informed consents were given to each patient included in the study.
Rights and permissions
About this article
Cite this article
Gennai, S., Rallo, A., Keil, D. et al. Elaboration of a clinical and paraclinical score to estimate the probability of herpes simplex virus encephalitis in patients with febrile, acute neurologic impairment. Eur J Clin Microbiol Infect Dis 35, 935–939 (2016). https://doi.org/10.1007/s10096-016-2619-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-016-2619-7