Abstract
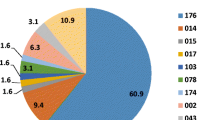
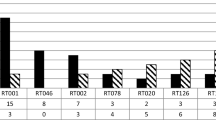
Reduced susceptibility to metronidazole and vancomycin in Clostridium difficile has been reported, which emphasises the need for simple antimicrobial susceptibility testing methods. The aim of this study was to apply a published disc diffusion method and zone diameter breakpoint correlates to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) epidemiological minimum inhibitory concentration (MIC) cut-off values in a routine setting. Metronidazole and vancomycin zone diameters from 2702 isolates were recorded. Fifteen isolates had a metronidazole zone diameter below the published breakpoint (<23 mm) and five isolates had a vancomycin zone diameter below the published breakpoint (<19 mm), most of which were polymerase chain reaction (PCR) ribotype 027. The total number of PCR ribotype 027 was 29 (1.1 %). Overall, C. difficile PCR ribotype 027 isolates had smaller zone diameters than non-027 isolates. The disc diffusion method is very simple and inexpensive, and the published zone diameter breakpoints will detect C. difficile isolates with reduced susceptibility to metronidazole and vancomycin.

Similar content being viewed by others
References
Bouza E (2012) Consequences of Clostridium difficile infection: understanding the healthcare burden. Clin Microbiol Infect 18(Suppl 6):5–12
Debast SB, Bauer MP, Kuijper EJ et al (2014) European Society of Clinical Microbiology and Infectious Diseases: update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect 20(Suppl 2):1–26
Brazier JS, Fawley W, Freeman J et al (2001) Reduced susceptibility of Clostridium difficile to metronidazole. J Antimicrob Chemother 48:741–742
Wong SS, Woo PC, Luk WK et al (1999) Susceptibility testing of Clostridium difficile against metronidazole and vancomycin by disk diffusion and Etest. Diagn Microbiol Infect Dis 34:1–6
Peláez T, Alcalá L, Alonso R et al (2002) Reassessment of Clostridium difficile susceptibility to metronidazole and vancomycin. Antimicrob Agents Chemother 46:1647–1650
Baines SD, O’Connor R, Freeman J et al (2008) Emergence of reduced susceptibility to metronidazole in Clostridium difficile. J Antimicrob Chemother 62:1046–1052
Mutlu E, Wroe AJ, Sanchez-Hurtado K et al (2007) Molecular characterization and antimicrobial susceptibility patterns of Clostridium difficile strains isolated from hospitals in south-east Scotland. J Med Microbiol 56:921–929
Freeman J, Vernon J, Morris K et al (2015) Pan-European longitudinal surveillance of antibiotic resistance among prevalent Clostridium difficile ribotypes. Clin Microbiol Infect 21:248.e9–248.e16
Erikstrup LT, Danielsen TK, Hall V et al (2012) Antimicrobial susceptibility testing of Clostridium difficile using EUCAST epidemiological cut-off values and disk diffusion correlates. Clin Microbiol Infect 18:E266–E272
de Boer RF, Wijma JJ, Schuurman T et al (2010) Evaluation of a rapid molecular screening approach for the detection of toxigenic Clostridium difficile in general and subsequent identification of the tcdC Delta117 mutation in human stools. J Microbiol Methods 83:59–65
Hoegh AM, Nielsen JB, Lester A et al (2012) A multiplex, internally controlled real-time PCR assay for detection of toxigenic Clostridium difficile and identification of hypervirulent strain 027/ST-1. Eur J Clin Microbiol Infect Dis 31:1073–1079
Justesen US, Erikstrup LT, Danielsen TK et al (2012) The Brucella blood agar disc diffusion antimicrobial susceptibility testing—reproducibility results for Clostridium difficile ATCC 700057. Clin Microbiol Infect 18(Suppl 3):S117–S118
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Holt, H.M., Danielsen, T.K. & Justesen, U.S. Routine disc diffusion antimicrobial susceptibility testing of Clostridium difficile and association with PCR ribotype 027. Eur J Clin Microbiol Infect Dis 34, 2243–2246 (2015). https://doi.org/10.1007/s10096-015-2475-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-015-2475-x