Abstract
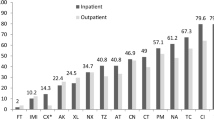
Motility mediated by the flagella of Escherichia coli is important for the bacteria to move toward host cells. Here, we present the relationship among bacterial motility, virulence factors, antimicrobial susceptibility, and types of infection. A total of 231 clinical E. coli isolates from different infections were collected and analyzed. Higher-motility strains (motility diameter ≥6.6 mm) were more common in spontaneous bacterial peritonitis (SBP) (SBP 59 %, colonization 32 %, urinary tract infection 16 %, urosepsis 34 %, and biliary tract infection 29 %; p < 0.0001). Compared with the higher-motility group, there was a higher prevalence of afa and ompT genes (p = 0.0160 and p = 0.0497, respectively) in E. coli strains with lower motility. E. coli isolates with higher and lower motility were in different phylogenetic groups (p = 0.018), with a lower prevalence of A and B1 subgroups in higher-motility strains. Also, the patterns of virulence factors and antibiotic susceptibility of E. coli isolates derived from various infections were significantly different. This study demonstrates that the prevalence of higher-motility strains was greater in E. coli isolates from SBP compared to other types of infection. Various types of E. coli infection were associated with differences in bacterial motility, virulence factors, and antibiotic susceptibility. More bacterial virulence factors may be necessary for the development of extraintestinal infections caused by E. coli isolates with lower motility.

Similar content being viewed by others
References
Wang MC, Tseng CC, Chen CY, Wu JJ, Huang JJ (2002) The role of bacterial virulence and host factors in patients with Escherichia coli bacteremia who have acute cholangitis or upper urinary tract infection. Clin Infect Dis 35:1161–1166
Burall LS, Harro JM, Li X, Lockatell CV, Himpsl SD, Hebel JR, Johnson DE, Mobley HL (2004) Proteus mirabilis genes that contribute to pathogenesis of urinary tract infection: identification of 25 signature-tagged mutants attenuated at least 100-fold. Infect Immun 72:2922–2938
Mobley HL, Belas R, Lockatell V, Chippendale G, Trifillis AL, Johnson DE, Warren JW (1996) Construction of a flagellum-negative mutant of Proteus mirabilis: effect on internalization by human renal epithelial cells and virulence in a mouse model of ascending urinary tract infection. Infect Immun 64:5332–5340
Schmitt CK, Ikeda JS, Darnell SC, Watson PR, Bispham J, Wallis TS, Weinstein DL, Metcalf ES, O’Brien AD (2001) Absence of all components of the flagellar export and synthesis machinery differentially alters virulence of Salmonella enterica serovar Typhimurium in models of typhoid fever, survival in macrophages, tissue culture invasiveness, and calf enterocolitis. Infect Immun 69:5619–5625
Zeng H, Carlson AQ, Guo Y, Yu Y, Collier-Hyams LS, Madara JL, Gewirtz AT, Neish AS (2003) Flagellin is the major proinflammatory determinant of enteropathogenic Salmonella. J Immunol 171:3668–3674
Terry K, Williams SM, Connolly L, Ottemann KM (2005) Chemotaxis plays multiple roles during Helicobacter pylori animal infection. Infect Immun 73:803–811
Girón JA, Torres AG, Freer E, Kaper JB (2002) The flagella of enteropathogenic Escherichia coli mediate adherence to epithelial cells. Mol Microbiol 44:361–379
Kao CY, Sheu BS, Sheu SM, Yang HB, Chang WL, Cheng HC, Wu JJ (2012) Higher motility enhances bacterial density and inflammatory response in dyspeptic patients infected with Helicobacter pylori. Helicobacter 17:411–416
Simms AN, Mobley HL (2008) Multiple genes repress motility in uropathogenic Escherichia coli constitutively expressing type 1 fimbriae. J Bacteriol 190:3747–3756
Lane MC, Lockatell V, Monterosso G, Lamphier D, Weinert J, Hebel JR, Johnson DE, Mobley HL (2005) Role of motility in the colonization of uropathogenic Escherichia coli in the urinary tract. Infect Immun 73:7644–7656
Lane MC, Alteri CJ, Smith SN, Mobley HL (2007) Expression of flagella is coincident with uropathogenic Escherichia coli ascension to the upper urinary tract. Proc Natl Acad Sci U S A 104:16669–16674
Schwan WR (2008) Flagella allow uropathogenic Escherichia coli ascension into murine kidneys. Int J Med Microbiol 298:441–447
Pichon C, Héchard C, du Merle L, Chaudray C, Bonne I, Guadagnini S, Vandewalle A, Le Bouguénec C (2009) Uropathogenic Escherichia coli AL511 requires flagellum to enter renal collecting duct cells. Cell Microbiol 11:616–628
Tseng CC, Huang JJ, Ko WC, Yan JJ, Wu JJ (2001) Decreased predominance of papG class II allele in Escherichia coli strains isolated from adults with acute pyelonephritis and urinary tract abnormalities. J Urol 166:1643–1646
Wang MC, Tseng CC, Wu AB, Huang JJ, Sheu BS, Wu JJ (2009) Different roles of host and bacterial factors in Escherichia coli extra-intestinal infections. Clin Microbiol Infect 15:372–379
Wang MC, Lin WH, Tseng CC, Wu AB, Teng CH, Yan JJ, Wu JJ (2013) Role of K1 capsule antigen in cirrhotic patients with Escherichia coli spontaneous bacterial peritonitis in southern Taiwan. Eur J Clin Microbiol Infect Dis 32:407–412
Clermont O, Bonacorsi S, Bingen E (2000) Rapid and simple determination of the Escherichia coli phylogenetic group. Appl Environ Microbiol 66:4555–4558
Emődy L, Kerényi M, Nagy G (2003) Virulence factors of uropathogenic Escherichia coli. Int J Antimicrob Agents 22(Suppl 2):S29–S33
Mobley HL, Island MD, Massad G (1994) Virulence determinants of uropathogenic Escherichia coli and Proteus mirabilis. Kidney Int Suppl 47:S129–S136
Donnelly MA, Steiner TS (2002) Two nonadjacent regions in enteroaggregative Escherichia coli flagellin are required for activation of toll-like receptor 5. J Biol Chem 277:40456–40461
Lee JM, Han KH, Ahn SH (2009) Ascites and spontaneous bacterial peritonitis: an Asian perspective. J Gastroenterol Hepatol 24:1494–1503
Căruntu FA, Benea L (2006) Spontaneous bacterial peritonitis: pathogenesis, diagnosis, treatment. J Gastrointest Liver Dis 15:51–56
Parthasarathy G, Yao Y, Kim KS (2007) Flagella promote Escherichia coli K1 association with and invasion of human brain microvascular endothelial cells. Infect Immun 75:2937–2945
Duan Q, Zhou M, Zhu X, Bao W, Wu S, Ruan X, Zhang W, Yang Y, Zhu J, Zhu G (2012) The flagella of F18ab Escherichia coli is a virulence factor that contributes to infection in a IPEC-J2 cell model in vitro. Vet Microbiol 160:132–140
Haiko J, Westerlund-Wikström B (2013) The role of the bacterial flagellum in adhesion and virulence. Biology (Basel) 2:1242–1267
Wright KJ, Seed PC, Hultgren SJ (2005) Uropathogenic Escherichia coli flagella aid in efficient urinary tract colonization. Infect Immun 73:7657–7668
Acknowledgments
We thank Robert Jonas for the helpful comments on this manuscript. This study was supported, in part, by grants NSC 99-2314-B-006-017-MY3 from the National Science Council and NCKUH-10006001 from the National Cheng Kung University Hospital, Taiwan.
Conflict of interest
No conflict of interest declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Y. Kao and W. H. Lin contributed equally to the article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 30 kb)
Rights and permissions
About this article
Cite this article
Kao, C.Y., Lin, W.H., Tseng, C.C. et al. The complex interplay among bacterial motility and virulence factors in different Escherichia coli infections. Eur J Clin Microbiol Infect Dis 33, 2157–2162 (2014). https://doi.org/10.1007/s10096-014-2171-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-014-2171-2