Abstract
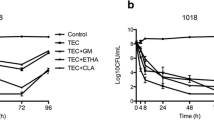
Vancomycin lock solution (LS) is recommended for the conservative treatment of subcutaneous injection port (SIP)-related infections, but may be associated with failure. We used an in vitro dynamic model of biofilm formation in an SIP, based on a continuous flow circulating via a real SIP, to assess the effectiveness of vancomycin (5 mg/ml), daptomycin (5 mg/ml) and ethanol 40 % LS in eradicating a pre-established Staphylococcus epidermidis biofilm. Heparin, Ringer’s lactate and enoxaparin sodium LS were used as controls. The logarithmic reductions of colony-forming units (CFU) were compared by Student’s t-test. After 24 h of exposure, the vancomycin LS did not exert a greater bactericidal effect than the heparin LS control (mean logarithmic reduction: 2.27 ± 0.58 vs. 1.34 ± 0.22, respectively, p = 0.3). The mean logarithmic reduction was greater with daptomycin LS (5.45 ± 0.14 vs. 0.39 ± 0.12, p < 0.01) and ethanol LS (6.79 ± 1.03 vs. 1.43 ± 0.54, p = 0.02). Bacterial revival after exposure to 24 h of LS was assessed. The mean viable bacteria count was significantly higher for vancomycin LS (9.36 ± 0.10 log10CFU) and daptomycin LS (9.16 ± 0.02 log10CFU) than for ethanol LS (2.95 ± 1.65 log10CFU). Ethanol appeared to be the most attractive option to treat SIP-related infection, but its poor ability to entirely disrupt the biofilm structure may require its use in association with a dispersal agent to avoid renewal of the biofilm.




Similar content being viewed by others
References
Walser EM (2012) Venous access ports: indications, implantation technique, follow-up, and complications. Cardiovasc Intervent Radiol 35:751–764
Maki DG, Kluger DM, Crnich CJ (2006) The risk of bloodstream infection in adults with different intravascular devices: a systematic review of 200 published prospective studies. Mayo Clin Proc 81:1159–1171
Crisinel M, Mahy S, Ortega-Debalon P, Buisson M, Favre JP, Chavanet P, Piroth L (2009) Incidence, prévalence et facteurs de risque de survenue d’une première complication infectieuse sur chambres à cathéter implantables. Méd Mal Infect 39:252–258
Opilla M (2008) Epidemiology of bloodstream infection associated with parenteral nutrition. Am J Infect Control 36:S173.e5–S173.e8
Simon A, Ammann RA, Bode U, Fleischhack G, Wenchel HM, Schwamborn D, Gravou C, Schlegel PG, Rutkowski S, Dannenberg C, Körholz D, Laws HJ, Kramer MH (2008) Healthcare-associated infections in pediatric cancer patients: results of a prospective surveillance study from university hospitals in Germany and Switzerland. BMC Infect Dis 8:70
Adler A, Yaniv I, Solter E, Freud E, Samra Z, Stein J, Fisher S, Levy I (2006) Catheter-associated bloodstream infections in pediatric hematology–oncology patients: factors associated with catheter removal and recurrence. J Pediatr Hematol Oncol 28:23–28
Chang YF, Lo AC, Tsai CH, Lee PY, Sun S, Chang TH, Chen CC, Chang YS, Chen JR (2011) Higher complication risk of totally implantable venous access port systems in patients with advanced cancer—a single institution retrospective analysis. Palliat Med (in press)
Flynn PM, Willis B, Gaur AH, Shenep JL (2003) Catheter design influences recurrence of catheter-related bloodstream infection in children with cancer. J Clin Oncol 21:3520–3525
Raad I, Kassar R, Ghannam D, Chaftari AM, Hachem R, Jiang Y (2009) Management of the catheter in documented catheter-related coagulase-negative staphylococcal bacteremia: remove or retain? Clin Infect Dis 49:1187–1194
Donlan RM (2011) Biofilm elimination on intravascular catheters: important considerations for the infectious disease practitioner. Clin Infect Dis 52:1038–1045
Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O’Grady NP, Raad II, Rijnders BJ, Sherertz RJ, Warren DK (2009) Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis 49(1):1–45
Del Pozo JL, Alonso M, Serrera A, Hernaez S, Aguinaga A, Leiva J (2009) Effectiveness of the antibiotic lock therapy for the treatment of port-related enterococci, Gram-negative, or Gram-positive bacilli bloodstream infections. Diagn Microbiol Infect Dis 63:208–212
Fernandez-Hidalgo N, Almirante B, Calleja R, Ruiz I, Planes AM, Rodriguez D, Pigrau C, Pahissa A (2006) Antibiotic-lock therapy for long-term intravascular catheter-related bacteraemia: results of an open, non-comparative study. J Antimicrob Chemother 57:1172–1180
Messing B, Peitra-Cohen S, Debure A, Beliah M, Bernier JJ (1988) Antibiotic-lock technique: a new approach to optimal therapy for catheter-related sepsis in home-parenteral nutrition patients. JPEN J Parenter Enter Nutr 12:185–189
Guedon C, Nouvellon M, Lalaude O, Lerebours E (2002) Efficacy of antibiotic-lock technique with teicoplanin in Staphylococcus epidermidis catheter-related sepsis during long-term parenteral nutrition. JPEN J Parenter Enter Nutr 26:109–113
Giacometti A, Cirioni O, Ghiselli R, Orlando F, Mocchegiani F, Silvestri C, Licci A, De Fusco M, Provinciali M, Saba V, Scalise G (2005) Comparative efficacies of quinupristin–dalfopristin, linezolid, vancomycin, and ciprofloxacin in treatment, using the antibiotic-lock technique, of experimental catheter-related infection due to Staphylococcus aureus. Antimicrob Agents Chemother 49:4042–4045
Wiederhold NP, Coyle EA, Raad II, Prince RA, Lewis RE (2005) Antibacterial activity of linezolid and vancomycin in an in vitro pharmacodynamic model of gram-positive catheter-related bacteraemia. J Antimicrob Chemother 55:792–795
Lee JY, Ko KS, Peck KR, Oh WS, Song JH (2006) In vitro evaluation of the antibiotic lock technique (ALT) for the treatment of catheter-related infections caused by staphylococci. J Antimicrob Chemother 57(6):1110–1115
Van Praagh AD, Li T, Zhang S, Arya A, Chen L, Zhang XX, Bertolami S, Mortin LI (2011) Daptomycin antibiotic lock therapy in a rat model of staphylococcal central venous catheter biofilm infections. Antimicrob Agents Chemother 55(9):4081–4089
Rijnders BJ, Van Wijngaerden E, Vandecasteele SJ, Stas M, Peetermans WE (2005) Treatment of long-term intravascular catheter-related bacteraemia with antibiotic lock: randomized, placebo-controlled trial. J Antimicrob Chemother 55:90–94
Fortún J, Grill F, Martín-Dávila P, Blázquez J, Tato M, Sánchez-Corral J, García-San Miguel L, Moreno S (2006) Treatment of long-term intravascular catheter-related bacteraemia with antibiotic-lock therapy. J Antimicrob Chemother 58(4):816–821
LaPlante KL, Mermel LA (2007) In vitro activity of daptomycin and vancomycin lock solutions on staphylococcal biofilms in a central venous catheter model. Nephrol Dial Transplant 22:2239–2246
Qu Y, Istivan TS, Daley AJ, Rouch DA, Deighton MA (2009) Comparison of various antimicrobial agents as catheter lock solutions: preference for ethanol in eradication of coagulase-negative staphylococcal biofilms. J Med Microbiol 58:442–450
Stevens DL (2006) The role of vancomycin in the treatment paradigm. Clin Infect Dis 42(Suppl 1):S51–S57
Aumeran C, Thibert E, Chapelle FA, Hennequin C, Lesens O, Traoré O (2012) Assessment on experimental bacterial biofilms and in clinical practice of the efficacy of sampling solutions for microbiological testing of endoscopes. J Clin Microbiol 50:938–942
Pineau L, Roques C, Luc J, Michel G (1997) Automatic washer disinfector for flexible endoscopes: a new evaluation process. Endoscopy 29:372–379
Curtin J, Cormican M, Fleming G, Keelehan J, Colleran E (2003) Linezolid compared with eperezolid, vancomycin, and gentamicin in an in vitro model of antimicrobial lock therapy for Staphylococcus epidermidis central venous catheter-related biofilm infections. Antimicrob Agents Chemother 47(10):3145–3148
Chauhan A, Lebeaux D, Decante B, Kriegel I, Escande MC, Ghigo JM, Beloin C (2012) A rat model of central venous catheter to study establishment of long-term bacterial biofilm and related acute and chronic infections. PLoS One 7(5):e37281, Epub 2012 May 16
Balestrino D, Souweine B, Charbonnel N, Lautrette A, Aumeran C, Traoré O, Forestier C (2009) Eradication of microorganisms embedded in biofilm by an ethanol-based catheter lock solution. Nephrol Dial Transplant 24:3204–3209
Chaieb K, Zmantar T, Souiden Y, Mahdouani K, Bakhrouf A (2011) XTT assay for evaluating the effect of alcohols, hydrogen peroxide and benzalkonium chloride on biofilm formation of Staphylococcus epidermidis. Microb Pathog 50:1–5
Cazander G, van de Veerdonk MC, Vandenbroucke-Grauls CMJE, Schreurs MWJ, Jukema GN (2010) Maggot excretions inhibit biofilm formation on biomaterials. Clin Orthop Relat Res 468:2789–2796
Acknowledgements
Damien Balestrino, Nicolas Charbonnel [Clermont Université, UMR CNRS 6023 Laboratoire Microorganismes: Génome Environnement (LMGE), Université d’Auvergne, CHU Clermont Ferrand, 63003 Clermont-Ferrand, France], Henri Laurichesse, Jean Beytout, Violaine Corbin (Service des Maladies Infectieuses, Pôle REUNNHIR, CHU Clermont-Ferrand, Clermont-Ferrand, France), Bertrand Souweine, Alexandre Lautrette, Natacha Mrozek (Réanimation Médicale, Pôle REUNNHIR, CHU Clermont-Ferrand, Clermont-Ferrand, France), Christelle Blavignac (CICS platform, Clermont University, electron microscopy).
This research was supported, in part, by an unrestricted, investigator-initiated grant from Cubist Pharmaceuticals, Inc., 65 Hayden Avenue, Lexington, MA 02421, USA. We thank Bard Access Systems, Salt Lake City, UT, USA, for providing the titanium central venous pressure port (TICVP) and Jeffrey Watts for reviewing the manuscript (Université Clermont 1, Faculté de Médecine de Clermont Ferrand, 63003 Clermont-Ferrand, France).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Aumeran, C., Guyot, P., Boisnoir, M. et al. Activity of ethanol and daptomycin lock on biofilm generated by an in vitro dynamic model using real subcutaneous injection ports. Eur J Clin Microbiol Infect Dis 32, 199–206 (2013). https://doi.org/10.1007/s10096-012-1732-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-012-1732-5