Abstract
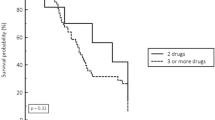
Clinical reports on infections by pandrug-resistant (PDR) bacteria are scarce. This observational case series study was conducted during a 2-year period at a university hospital. Patients infected by PDR gram-negative bacteria comprised the study cohort. An isolate was defined as PDR if it was resistant to all antibiotic classes available for empirical treatment. A total of 21 patients infected by PDR gram-negative bacteria were recorded. The mean APACHE II score on admission was 18.8, the mean Charlson comorbidity index was 2.9, and 20 (95.2%) patients had a history of intensive care unit hospitalization. All patients had recent exposure to multiple antibiotics (median, 6 antibiotic groups). Infections occurred at a mean of 41.5 days after admission. The mean length of stay after infection was 54.6 days and 5 (23.8%) patients died due to the infection. Treatment was mainly based on a colistin-containing regimen (47.6%) or tigecycline (33.3%). All patients treated with tigecycline had total resolution of the infection and a notably shorter length of hospital stay after infection. In conclusion, PDR gram-negative bacterial infections are associated with considerable prolongation of hospitalization and mortality, although the mortality is not as high as that expected. Tigecycline appears to be effective for the successful treatment of PDR infections.
Similar content being viewed by others
References
Beno P, Krcmery V, Demitrovicova A (2006) Bacteraemia in cancer patients caused by colistin-resistant Gram-negative bacilli after previous exposure to ciprofloxacin and/or colistin. Clin Microbiol Infect 12:497–498. doi:10.1111/j.1469-0691.2006.01364.x
Falagas ME, Bliziotis IA, Kasiakou SK, Samonis G, Athanassopoulou P, Michalopoulos A (2005) Outcome of infections due to pandrug-resistant (PDR) Gram-negative bacteria. BMC Infect Dis 5:24–30. doi:10.1186/1471-2334-5-24
Falagas ME, Rafailidis PI, Matthaiou DK, Virtzili S, Nikita D, Michalopoulos A (2008) Pandrug-resistant Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumannii infections: characteristics and outcome in a series of 28 patients. Int J Antimicrob Agents 32(5):450–454. doi:10.1016/j.ijantimicag.2008.05.016
Mentzelopoulos SD, Pratikaki M, Platsouka E, Kraniotaki H, Zervakis D, Koutsoukou A, Nanas S, Paniara O, Roussos C, Giamarellos-Bourboulis E, Routsi C, Zakynthinos SG (2007) Prolonged use of carbapenems and colistin predisposes to ventilator-associated pneumonia by pandrug-resistant Pseudomonas aeruginosa. Intensive Care Med 33:1524–1532. doi:10.1007/s00134-007-0683-2
Elemam A, Rahimian J, Mandell W (2009) Infection with panresistant Klebsiella pneumoniae: a report of 2 cases and a brief review of the literature. Clin Infect Dis 49(2):271–274. doi:10.1086/600042
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13(10):818–829
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383
Falagas ME, Bliziotis IA (2007) Pandrug-resistant Gram-negative bacteria: the dawn of the post-antibiotic era? Int J Antimicrob Agents 29:630–636. doi:10.1016/j.ijantimicag.2006.12.012
Horan TC, Gaynes RP (2004) CDC definitions for nosocomial infections. In: Mayhall CG (ed) Hospital epidemiology and infection control, 3rd edn. Lippincott Williams & Wilkins, Philadelphia, pp 1659–1702
Clinical and Laboratory Standards Institute (2007) Performance standards for antimicrobial susceptibility testing; seventeenth informational supplement. CLSI document M100-S17. Clinical and Laboratory Standards Institute, Wayne
European Committee on Antimicrobial Susceptibility Testing (EUCAST) Steering Committee (2006) EUCAST technical note on tigecycline. Clin Microbiol Infect 12:1147–1149. doi:10.1111/j.1469-0691.2006.01578.x
Kelesidis T, Karageorgopoulos DE, Kelesidis I, Falagas ME (2008) Tigecycline for the treatment of multidrug-resistant Enterobacteriaceae: a systematic review of the evidence from microbiological and clinical studies. J Antimicrob Chemother 62:895–904. doi:10.1093/jac/dkn311
Vasilev K, Reshedko G, Orasan R, Sanchez M, Teras J, Babinchak T, Dukart G, Cooper A, Dartois N, Gandjini H, Orrico R, Ellis-Grosse E; 309 Study Group (2008) A Phase 3, open-label, non-comparative study of tigecycline in the treatment of patients with selected serious infections due to resistant Gram-negative organisms including Enterobacter species, Acinetobacter baumannii and Klebsiella pneumoniae. J Antimicrob Chemother 62(Suppl 1):i29–i40. doi:10.1093/jac/dkn249
Navon-Venezia S, Leavitt A, Carmeli Y (2007) High tigecycline resistance in multidrug-resistant Acinetobacter baumannii. J Antimicrob Chemother 59:772–774. doi:10.1093/jac/dkm018
Peleg AY, Potoski BA, Rea R, Adams J, Sethi J, Capitano B, Husain S, Kwak EJ, Bhat SV, Paterson DL (2007) Acinetobacter baumannii bloodstream infection while receiving tigecycline: a cautionary report. J Antimicrob Chemother 59:128–131. doi:10.1093/jac/dkl441
Conflict of interest
None.
Financial support
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsioutis, C., Kritsotakis, E.I., Maraki, S. et al. Infections by pandrug-resistant gram-negative bacteria: clinical profile, therapeutic management, and outcome in a series of 21 patients. Eur J Clin Microbiol Infect Dis 29, 301–305 (2010). https://doi.org/10.1007/s10096-009-0857-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-009-0857-7