Abstract
Background
Case presentation of acute onset bilateral painless vision loss caused by bilateral infarction of the lateral geniculate bodies (LGB) and a systematic review of the literature.
Methods
A descriptive case report is presented on a 17-year-old female diagnosed with acute pancreatitis who developed acute onset bilateral painless vision loss. A systematic literature review of cases with bilateral LGB lesions was conducted across three electronic databases (PubMed/PubMed Central/MEDLINE, Scopus, and ScienceDirect). The review was conducted in concordance with PRISMA guidelines and prospectively registered on PROSPERO (CRD42022362491).
Results
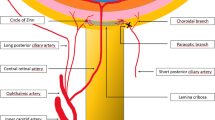
The reported 17-year-old female was found to have MRI findings consistent with bilateral hemorrhagic infarction of the LGB and Purtscher-like retinopathy. A systematic literature review of bilateral LGB infarction yielded 23 records for analysis. 19/23 (82.6%) of reported cases occurred in women. Bilateral vision loss was noted in all cases. The average reported age was 27 years old with a range from 2–50. Gastrointestinal pathology (e.g., pancreatitis, gastroenteritis) was present in 8/23 (34.7%) of cases. 8/23 (34.7%) cases had neuroimaging or pathological evidence of hemorrhagic transformation of the infarct. Most cases experienced partial recovery of visual loss; only one case (4.7%) had complete visual recovery. 9/23 (39.1%) cases were reported from the United States and 4/23 (17.3%) from India.
Conclusions
Bilateral LGB lesion is a rare cause of vision loss, typically caused by systemic diseases and with female preponderance. Purported pathophysiology relates to increased vulnerability of the LGB to ischemic and metabolic stress.



Similar content being viewed by others
Change history
17 May 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10072-024-07595-5
References
Mackenzie IM, Meighan S, Pollock EN (1933) On the projection of the retinal quadrants on the lateral geniculate bodies, and the relationship of the quadrants to the optic radiations. Trans Ophthalmol Soc U K 53:142–169
Karadan U, Supreeth RN, Chellenton J et al (2019) Bilateral lateral geniculate body lesions causing reversible blindness in a patient with posterior reversible encephalopathy syndrome. J R Coll Physicians Edinb 49:34–36. https://doi.org/10.4997/JRCPE.2019.107
Lefebvre PR, Cordonnier M, Baleriaux D et al (2004) An unusual cause of visual loss: involvement of bilateral lateral geniculate bodies. AJNR Am J Neuroradiol 25:1544–1548
Mariotti P, Iorio R, Frisullo G et al (2010) Acute necrotizing encephalopathy during novel influenza A (H1N1) virus infection. Ann Neurol 68:111–114. https://doi.org/10.1002/ana.21996
Mathew T, D'Souza D, Nadig R et al (2016) Bilateral lateral geniculate body hemorrhagic infarction: a rare cause of acute bilateral painless vision loss in female patients. Neurol India 64:160–162. https://doi.org/10.4103/0028-3886.173671
Merren MD (1972) Bilateral lateral geniculate body necrosis as a cause of amblyopia. Neurology 22:263–268. https://doi.org/10.1212/wnl.22.3.263
Moseman CP, Shelton S (2002) Permanent blindness as a complication of pregnancy induced hypertension, Obstet Gynecol. 100:943–945. https://doi.org/10.1016/s0029-7844(02)02250-0
Mudumbai RC, Bhandari A (2007) Bilateral isolated lateral geniculate body lesions in a patient with pancreatitis and microangiopathy. J Neuroophthalmol 27:169–175. https://doi.org/10.1097/WNO.0b013e31814a5921
Mulholland C, Best J, Rennie I et al (2010) Bilateral sectoranopia caused by bilateral geniculate body infarction in a 14-year-old boy with inflammatory bowel disease. J aapos 14:435–437. https://doi.org/10.1016/j.jaapos.2010.06.010
Ogawa Y, Nakagawa M, Yazaki M et al (2014) An abnormal MRI signal in both lateral geniculate bodies is a diagnostic indicator for patients with Behcet’s disease. Case Rep Neurol 6:78–82. https://doi.org/10.1159/000360848
Puppala GK, Prabhu AN, Gorthi SP et al (2019) Acute influenza A virus-associated encephalitis with bilateral lateral geniculate body hemorrhagic infarction-complicating pregnancy. Ann Indian Acad Neurol 22:355–357. https://doi.org/10.4103/aian.AIAN_319_18
Rausch C, Bartsch L, Gorodetski B et al (2021) Loss of vision because of bilateral lateral geniculate body infarction after traumatic brain injury. Neurol Clin Pract 11:e947–e948. https://doi.org/10.1212/CPJ.0000000000001064
Silva RR, Penna ER, Fusão EF et al (2014) An unusual cause of blindness: infarction in the bilateral lateral geniculate bodies. J Stroke Cerebrovasc Dis 23:1736–1737. https://doi.org/10.1016/j.jstrokecerebrovasdis.2014.01.004
Stem MS, Fahim A, Trobe JD et al (2014) Lateral geniculate lesions causing reversible blindness in a pre-eclamptic patient with a variant of posterior reversible encephalopathy syndrome. J Neuroophthalmol 34:372–376. https://doi.org/10.1097/WNO.0000000000000120
Viloria A, Jimenez B, Palacin M (2015) Reversible severe bilateral visual loss in an unusual case of bilateral lateral geniculate myelinolysis during acute pancreatitis. BMJ Case Rep. https://doi.org/10.1136/bcr-2015-212409
Cheng MF, Pang H (2020) Bilateral lateral geniculate body involvement causing visual impairment in a patient with metastatic carcinoma of lung. AJMHS:53
Murugesan S, Senthilkumar E, Kumar K et al (2022) Isolated bilateral lateral geniculate body necrosis following acute pancreatitis: a rare cause of bilateral loss of vision in a young female. J Postgrad Med. https://doi.org/10.4103/jpgm.jpgm_1134_21
Muller-Axt C, Eichner C, Rusch H et al (2021) Mapping the human lateral geniculate nucleus and its cytoarchitectonic subdivisions using quantitative MRI. Neuroimage 244:118559. https://doi.org/10.1016/j.neuroimage.2021.118559
Cooper RM (2002) Diffuse light increases metabolic activity in the lateral geniculate nucleus, visual cortex, and superior colliculus of the cone-dominated ground squirrel visual system. Vision Res 42:2899–2907. https://doi.org/10.1016/s0042-6989(02)00361-9
Fujii K, Lenkey C, Rhoton AL Jr (1980) Microsurgical anatomy of the choroidal arteries: lateral and third ventricles. J Neurosurg 52:165–188. https://doi.org/10.3171/jns.1980.52.2.0165
Siesjo BK, Ingvar M, Pelligrino D (1983) Regional differences in vascular autoregulation in the rat brain in severe insulin-induced hypoglycemia. J Cereb Blood Flow Metab 3:478–485. https://doi.org/10.1038/jcbfm.1983.74
Miguel AI, Henriques F, Azevedo LF et al (2013) Systematic review of Purtscher’s and Purtscher-like retinopathies. Eye (Lond) 27:1–13. https://doi.org/10.1038/eye.2012.222
Meys KME, de Vries LS, Groenendaal F et al (2022) The mammillary bodies: a review of causes of injury in infants and children. AJNR Am J Neuroradiol 43:802–812. https://doi.org/10.3174/ajnr.A7463
Nazir M, Lone R, Charoo BA (2019) Infantile thiamine deficiency: new insights into an old disease. Indian Pediatr 56:673–681
Baker CF, Jeerakathil T, Lewis JR et al (2006) Isolated bilateral lateral geniculate infarction producing bow-tie visual field defects. Can J Ophthalmol 41:609–613. https://doi.org/10.1016/s0008-4182(06)80033-5
Bartel MJ, Bhalla R, Lopez Chiriboga AS et al (2016) Vision loss: another ERCP-related adverse event, also known as isolated bilateral lateral geniculate body infarction. Gastrointest Endosc 83:474–475. https://doi.org/10.1016/j.gie.2015.07.031
Breker DA, Stacey AW, Srinivasan A et al (2015) Vision loss caused by retinal and lateral geniculate nucleus infarction in H1N1 influenza. J Neuroophthalmol 35:265–269. https://doi.org/10.1097/wno.0000000000000247
Donahue SP, Kardon RH, Thompson HS (1995) Hourglass-shaped visual fields as a sign of bilateral lateral geniculate myelinolysis. Am J Ophthalmol 119:378–380. https://doi.org/10.1016/s0002-9394(14)71190-0
Greenfield DS, Siatkowski RM, Schatz NJ et al (1996) Bilateral lateral geniculitis associated with severe diarrhea. Am J Ophthalmol 122:280–281. https://doi.org/10.1016/s0002-9394(14)72030-6
Imes RK, Barakos J (2006) Presumed bilateral lateral geniculate nuclei ischemia. AJNR Am J Neuroradiol 27:9–10
Gocht A, Colmant HJ (1987) Central pontine and extrapontine myelinolysis: a report of 58 cases. Clin Neuropathol 6:262–270
Lambeck J, Hieber M, Dressing A et al (2019) Central pontine myelinosis and osmotic demyelination syndrome. Dtsch Arztebl Int 116:600–606. https://doi.org/10.3238/arztebl.2019.0600
Fischer M, Schmutzhard E (2017) Posterior reversible encephalopathy syndrome. J Neurol 264:1608–1616. https://doi.org/10.1007/s00415-016-8377-8
Etherton MR, Wu O, Cougo P et al (2019) Sex-specific differences in white matter microvascular integrity after ischaemic stroke. Stroke Vasc Neurol 4:198–205. https://doi.org/10.1136/svn-2019-000268
Reeves MJ, Bushnell CD, Howard G et al (2008) Sex differences in stroke: epidemiology, clinical presentation, medical care, and outcomes. Lancet Neurol 7:915–926. https://doi.org/10.1016/S1474-4422(08)70193-5
Gupte R, Brooks W, Vukas R et al (2019) Sex differences in traumatic brain injury: what we know and what we should know. J Neurotrauma 36:3063–3091. https://doi.org/10.1089/neu.2018.6171
Faura J, Bustamante A, Miro-Mur F et al (2021) Stroke-induced immunosuppression: implications for the prevention and prediction of post-stroke infections. J Neuroinflammation 18:127. https://doi.org/10.1186/s12974-021-02177-0
Puschel A, Lindenblatt N, Katzfuss J et al (2012) Immunosuppressants accelerate microvascular thrombus formation in vivo: role of endothelial cell activation. Surgery 151:26–36. https://doi.org/10.1016/j.surg.2011.06.026
(2015) Bath PM and Wardlaw JM. Pharmacological treatment and prevention of cerebral small vessel disease: a review of potential interventions. Int J Stroke 10:469–478. https://doi.org/10.1111/ijs.12466
Author information
Authors and Affiliations
Contributions
Concept, design, drafting of manuscript: Srichawla, Henninger. Analysis or interpretation of data: Srichawla, Catton, Lichtenberg. Funding and study supervision: Henninger. All authors critically reviewed the report and approved the final version.
Corresponding author
Ethics declarations
Ethical Approval and Informed consent
Written informed consent was obtained from the patient for the publication of this manuscript.
Conflict of interest
Dr. Henninger serves as consultant to GentiBio, Inc and reports research support from the Department of Defense, National Institute of Neurological Disorders and Stroke (NINDS) of the National Institutes of Health (NIH), and the Angel Fund for ALS Research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The original article contains a reference citation error. Cheng et al. (2020) should be replaced to “Cheng and Pang (2020)” in Table 1 continued and Table 2. In Reference 16, Cheng MFP, H (2020) should be corrected to “Cheng MF, Pang H (2020)”.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Srichawla, B.S., Catton, R.M., Lichtenberg, A.A. et al. Clinical characteristics and risk factors for bilateral lateral geniculate body pathology: a systematic review of the literature. Neurol Sci 44, 3481–3493 (2023). https://doi.org/10.1007/s10072-023-06818-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-06818-5