Abstract
Background
Epilepsy pathogenesis and progression are strongly influenced by inflammation. High-mobility group box-1 (HMGB1) is a key proinflammatory factor. The purpose of this study was to quantify and assess the relationship between HMGB1 level and epilepsy.
Methods
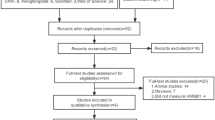
We searched Embase, Web of Science, PubMed, and the Cochrane Library for studies examining the relationship between HMGB1 and epilepsy. Two independent researchers extracted data and assessed quality using the Cochrane Collaboration tool. Data extracted were analyzed using Stata 15 and Review Manager 5.3. The study protocol was registered prospectively at INPLASY, ID: INPLASY2021120029.
Results
A total of 12 studies were eligible for inclusion. After exclusion of one study with reduced robustness, 11 studies were included, with a total of 443 patients and 333 matched controls. Two of the articles included cerebrospinal fluid and serum HMGB1 data, which were distinguished by “a” and “b,” respectively. The meta-analysis indicated that in comparison with the control group, the HMGB1 level was higher in epilepsy patients (SMD = 0.56, 95% CI = 0.27–0.85, P = 0.0002). Subgroup analysis of specimen types indicated that both serum HMGB1 and cerebrospinal fluid HMGB1 were higher in epilepsy patients than in the control group, with the increase in cerebrospinal fluid HMGB1 being more obvious. Subgroup analysis of disease types demonstrated that the serum HMGB1 level of epileptic seizure patients (including febrile and nonfebrile seizures) was significantly higher than that of matched controls. However, serum HMGB1 levels did not differ significantly between mild epilepsy patients and severe epilepsy patients. Patient age subgroup analysis showed higher HMGB1 in adolescents with epilepsy. Begg’s test did not indicate publication bias.
Conclusions
This is the first meta-analysis to summarize the association between HMGB1 level and epilepsy. The results of this meta-analysis indicate that epilepsy patients have elevated HMGB1. Large-scale studies with a high level of evidence are needed to reveal the exact relationship between HMGB1 level and epilepsy.





Similar content being viewed by others
References
Fisher RS, Acevedo C, Arzimanoglou A et al (2014) ILAE official report: a practical clinical definition of epilepsy[J]. Epilepsia 55(4):475–482
Ngugi AK, Bottomley C, Kleinschmidt I et al (2010) Estimation of the burden of active and life-time epilepsy: a meta-analytic approach[J]. Epilepsia 51(5):883–890
Thijs RD, Surges R, O’Brien TJ et al (2019) Epilepsy in adults[J]. Lancet 393(10172):689–701
Sanz P, Garcia-Gimeno MA (2020) Reactive glia inflammatory signaling pathways and epilepsy [J]. Int J Mol Sci 21(11):4096
Rana A, Musto AE (2018) The role of inflammation in the development of epilepsy[J]. J Neuroinflammation 15(1):144
Alyu F, Dikmen M (2017) Inflammatory aspects of epileptogenesis: contribution of molecular inflammatory mechanisms[J]. Acta Neuropsychiatr 29(1):1–16
Eyo UB, Murugan M, Wu LJ (2017) Microglia-neuron communication in epilepsy[J]. Glia 65(1):5–18
Mo M, Eyo UB, Xie M et al (2019) Microglial P2Y12 receptor regulates seizure-induced neurogenesis and immature neuronal projections[J]. J Neurosci 39(47):9453–9464
Hemmer B, Kerschensteiner M, Korn T (2015) Role of the innate and adaptive immune responses in the course of multiple sclerosis[J]. Lancet Neurol 14(4):406–419
Corps KN, Roth TL, Mcgavern DB (2015) Inflammation and neuroprotection in traumatic brain injury[J]. JAMA Neurol 72(3):355–362
Vezzani B, Carinci M, Patergnani S et al (2020) The dichotomous role of inflammation in the CNS: a mitochondrial point of view [J]. Biomolecules 10(10):1437
Vezzani A, Balosso S, Ravizza T (2019) Neuroinflammatory pathways as treatment targets and biomarkers in epilepsy[J]. Nat Rev Neurol 15(8):459–472
Zhang Z, Liu Q, Liu M et al (2018) Upregulation of HMGB1-TLR4 inflammatory pathway in focal cortical dysplasia type II[J]. J Neuroinflammation 15(1):27
Iori V, Maroso M, Rizzi M et al (2013) Receptor for advanced glycation endproducts is upregulated in temporal lobe epilepsy and contributes to experimental seizures[J]. Neurobiol Dis 58:102–114
Maroso M, Balosso S, Ravizza T et al (2010) Toll-like receptor 4 and high-mobility group box-1 are involved in ictogenesis and can be targeted to reduce seizures[J]. Nat Med 16(4):413–419
Shi Y, Zhang L, Teng J et al (2018) HMGB1 mediates microglia activation via the TLR4/NF-kappaB pathway in coriaria lactone induced epilepsy[J]. Mol Med Rep 17(4):5125–5131
Iori V, Frigerio F, Vezzani A (2016) Modulation of neuronal excitability by immune mediators in epilepsy[J]. Curr Opin Pharmacol 26:118–123
Zhao J, Zheng Y, Liu K et al (2020) HMGB1 is a therapeutic target and biomarker in diazepam-refractory status epilepticus with wide time window[J]. Neurotherapeutics 17(2):710–721
Ravizza T, Terrone G, Salamone A et al (2018) High mobility group box 1 is a novel pathogenic factor and a mechanistic biomarker for epilepsy[J]. Brain Behav Immun 72:14–21
Wykes RC, Heeroma JH, Mantoan L et al (2012) Optogenetic and potassium channel gene therapy in a rodent model of focal neocortical epilepsy[J]. Sci Transl Med 4(161):152r–161r
Fu L, Liu K, Wake H et al (2017) Therapeutic effects of anti-HMGB1 monoclonal antibody on pilocarpine-induced status epilepticus in mice[J]. Sci Rep 7(1):1179
Paudel YN, Shaikh MF, Chakraborti A et al (2018) HMGB1: a common biomarker and potential target for TBI, neuroinflammation, epilepsy, and cognitive dysfunction[J]. Front Neurosci 12:628
Choi J, Min HJ, Shin J (2011) Increased levels of HMGB1 and pro-inflammatory cytokines in children with febrile seizures [J]. J Neuroinflammation 8:135
Asano T, Ichiki K, Koizumi S et al (2011) High mobility group box 1 in cerebrospinal fluid from several neurological diseases at early time points[J]. Int J Neurosci 121(8):480–484
Issac MSM, Girgis M, Haroun M et al (2015) Association of genetic polymorphism of pre-MicroRNA-146a rs2910164 and serum high-mobility group box 1 with febrile seizures in Egyptian children[J]. J Child Neurol 30(4):437–444
Mahmoud A, Alnabya SA, Debdeb M (2018) High-mobility group box 1 protein serum level in children with febrile seizures [J]. Menoufia Med J 31:1005–1010
Salih KS, Hamdan FB, Al-Mayah QS (2020) Diagnostic value of matrix metalloproteinase-2 and high mobility group box 1 in patients with refractory epilepsy [J]. Egyptian J Neurol Psychiatry Neurosurg 56:102
Kamaşak T, Dilber B, Yaman SÖ et al (2020) HMGB-1, TLR4, IL-1R1, TNF-α, and IL-1β: novel epilepsy markers?[J]. Epileptic Disord 22(2):183–193
Kaya M A, Erin N, Bozkurt O et al (2021) Changes of HMGB-1 and sTLR4 levels in cerebrospinal fluid of patients with febrile seizures [J]. Epilepsy Res 169:106516
Morichi S, Yamanaka G, Watanabe Y et al (2021) High mobility group box 1 and angiogenetic growth factor levels in children with central nerve system infections [J]. J Infect Chemother 27:840–844
Wang N, Liu H, Ma B et al (2021) CSF high-mobility group box 1 is associated with drug-resistance and symptomatic etiology in adult patients with epilepsy[J]. Epilepsy Res 177:106767
Walker LE, Sills GJ, Jorgensen A et al (2022) High-mobility group box 1 as a predictive biomarker for drug-resistant epilepsy: a proof-of-concept study[J]. Epilepsia 63(1):e1–e6
Stang A (2010) Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses[J]. Eur J Epidemiol 25(9):603–605
Kwan P, Brodie MJ (2000) Early identification of refractory epilepsy[J]. N Engl J Med 342(5):314–319
Pitkanen A, Loscher W, Vezzani A et al (2016) Advances in the development of biomarkers for epilepsy[J]. Lancet Neurol 15(8):843–856
Vezzani A, Balosso S, Ravizza T (2008) The role of cytokines in the pathophysiology of epilepsy[J]. Brain Behav Immun 22(6):797–803
Ravizza T, Gagliardi B, Noe F et al (2008) Innate and adaptive immunity during epileptogenesis and spontaneous seizures: evidence from experimental models and human temporal lobe epilepsy[J]. Neurobiol Dis 29(1):142–160
Zhen C, Wang Y, Li D et al (2019) Relationship of high-mobility group box 1 levels and multiple sclerosis: a systematic review and meta-analysis[J]. Mult Scler Relat Disord 31:87–92
Le K, Mo S, Lu X et al (2018) Association of circulating blood HMGB1 levels with ischemic stroke: a systematic review and meta-analysis[J]. Neurol Res 40(11):907–916
Xia Q, Tao P, Xu J (2021) Association of polymorphism rs1045411 in the HMGB1 gene with cancer risk: evidence from a meta-analysis[J]. Int J Med Sci 18(6):1348–1355
Walker LE, Griffiths MJ, Mcgill F et al (2017) A comparison of HMGB1 concentrations between cerebrospinal fluid and blood in patients with neurological disease[J]. Biomarkers 22(7):635–642
Berger RP, Beers SR, Papa L et al (2012) Common data elements for pediatric traumatic brain injury: recommendations from the biospecimens and biomarkers workgroup[J]. J Neurotrauma 29(4):672–677
Pedersen BK, Steensberg A, Fischer C et al (2001) Exercise and cytokines with particular focus on muscle-derived IL-6[J]. Exerc Immunol Rev 7:18–31
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical approval
The authors declare that all procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• This is the first meta-analysis to investigate the association between HMGB1 level and epilepsy.
• HMGB1 levels are elevated in epileptic patients, particularly children.
• HMGB1 has the potential to serve as a biomarker for epilepsy.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, Y., Chen, X. & Liang, Y. Meta-analysis of HMGB1 levels in the cerebrospinal fluid and serum of patients with epilepsy. Neurol Sci 44, 2329–2337 (2023). https://doi.org/10.1007/s10072-023-06720-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-06720-0