Abstract
Introduction
GNE myopathy is an autosomal recessive distal myopathy caused by a biallelic mutation in UDP-N-acetylglucosamine 2-epomerase/N-acetylmannosamine kinase. In this study, we discuss the clinical features, pathological characteristics, genetic profiles, and atypical clinical manifestations of 22 Chinese GNE patients.
Materials and methods
Retrospective analysis was performed for GNE myopathy patients at our institute between 2005 and 2021. Histopathological analysis and gene testing were done according to standard protocols.
Results
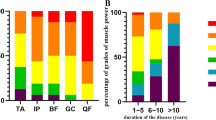
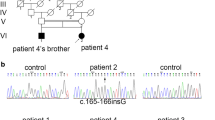
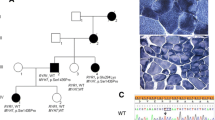
Molecular analysis revealed 14-reported and 7 novel mutations, including c.125G > A (p.P42Q), c.226G > A (p.V76I), c.970C > G (p.H324D), c.155A > G (p.D52G), c.1055G > A (p.R352H), c.1064G > A (p.G355E), and c.491 T > C (p.I164T) in GNE. D207V was the most frequent mutation showing an allele frequency of 25%. A total of 21 patients presented classic clinical manifestation, and only 1 patient had signs of proximal muscle weakness. A patient containing p.V603L and p.R160X mutations showed idiopathic thrombocytopenia and distal weakness. There were 4 female patients who experienced rapid deterioration after pregnancy.
Discussion
Our study revealed 7 novel mutations in GNE, where p.D207V was shown as a potential hotspot mutation in Chinese patients. Idiopathic thrombocytopenia should be a concern in GNE myopathy patients. Twenty-seven percent of female patients experienced rapid deterioration during pregnancy or after delivery.


Similar content being viewed by others
References
Nishino I, Carrillo-Carrasco N, Argov Z (2015) GNE myopathy: current update and future therapy. J Neurol Neurosurg Psychiatry 86:385–392. https://doi.org/10.1136/jnnp-2013-307051
Carrillo N, Malicdan MC, Huizing M (2018) GNE myopathy: etiology, diagnosis, and therapeutic challenges. Neurotherapeutics 15:900–914. https://doi.org/10.1007/s13311-018-0671-y
Nonaka I, Sunohara N, Ishiura S et al (1981) Familial distal myopathy with rimmed vacuole and lamellar (myeloid) body formation. J Neurol Sci 51:141–155. https://doi.org/10.1016/0022-510x(81)90067-8
Argov Z, Yarom R (1984) “Rimmed vacuole myopathy” sparing the quadriceps. A unique disorder in Iranian Jews. J Neurol Sci 64:33–43. https://doi.org/10.1016/0022-510x(84)90053-4
Tasca G, Ricci E, Monforte M et al (2012) Muscle imaging findings in GNE myopathy. J Neurol 259:1358–1365. https://doi.org/10.1007/s00415-011-6357-6
Liu CY, Yao J, Kovacs WC et al (2021) Skeletal muscle magnetic resonance biomarkers in GNE myopathy. Neurology 96:e798–e808. https://doi.org/10.1212/WNL.0000000000011231
Pogoryelova O, Gonzalez CJ, Nikolenko N et al (2018) GNE myopathy: from clinics and genetics to pathology and research strategies. Orphanet J Rare Dis 13:70. https://doi.org/10.1186/s13023-018-0802-x
Park YE, Kim HS, Choi ES et al (2012) Limb-girdle phenotype is frequent in patients with myopathy associated with GNE mutations. J Neurol Sci 321:77–81. https://doi.org/10.1016/j.jns.2012.07.061
de Dios JK, Shrader JA, Joe GO et al (2014) Atypical presentation of GNE myopathy with asymmetric hand weakness. Neuromuscul Disord 24:1063–1067. https://doi.org/10.1016/j.nmd.2014.07.006
Futterer J, Dalby A, Lowe GC et al (2018) Mutation in GNE is associated with a severe form of congenital thrombocytopenia. Blood 132:1855–1858. https://doi.org/10.1182/blood-2018-04-847798
Tanboon J, Rongsa K, Pithukpakorn M et al (2014) A novel mutation of the GNE gene in distal myopathy with rimmed vacuoles: a case with inflammation. Case Rep Neurol 6:55–59. https://doi.org/10.1159/000360730
Eisenberg I, Avidan N, Potikha T et al (2001) The UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene is mutated in recessive hereditary inclusion body myopathy. Nat Genet 29:83–87. https://doi.org/10.1038/ng718
Celeste FV, Vilboux T, Ciccone C et al (2014) Mutation update for GNE gene variants associated with GNE myopathy. Hum Mutat 35:915–926. https://doi.org/10.1002/humu.22583
Zhao J, Wang Z, Hong D et al (2015) Mutational spectrum and clinical features in 35 unrelated mainland Chinese patients with GNE myopathy. J Neurol Sci 354:21–26. https://doi.org/10.1016/j.jns.2015.04.028
Chen Y, Xi J, Zhu W et al (2019) GNE myopathy in Chinese population: hotspot and novel mutations. J Hum Genet 64:11–16. https://doi.org/10.1038/s10038-018-0525-9
Huizing M, Carrillo-Carrasco N, Malicdan MC et al (2014) GNE myopathy: new name and new mutation nomenclature. Neuromuscul Disord 24:387–389. https://doi.org/10.1016/j.nmd.2014.03.004
Li MM, Datto M, Duncavage EJ et al (2017) Standards and guidelines for the interpretation and reporting of sequence variants in cancer: a joint consensus recommendation of the association for molecular pathology, American Society of Clinical Oncology, and College of American Pathologists. J Mol Diagn 19:4–23. https://doi.org/10.1016/j.jmoldx.2016.10.002
Carrillo N, Malicdan M C, Huizing M (2020) GNE myopathy. In: GeneReviews® [Internet]. University of Washington, Seattle, WA, pp 1993–2022
Cho A, Hayashi YK, Monma K et al (2014) Mutation profile of the GNE gene in Japanese patients with distal myopathy with rimmed vacuoles (GNE myopathy). J Neurol Neurosurg Psychiatry 85:914–917. https://doi.org/10.1136/jnnp-2013-305587
Pogoryelova O, Wilson IJ, Mansbach H et al (2019) GNE genotype explains 20% of phenotypic variability in GNE myopathy. Neurol Genet 5(1):e308. https://doi.org/10.1212/NXG.0000000000000308
Grandis M, Gulli R, Cassandrini D et al (2010) The spectrum of GNE mutations: allelic heterogeneity for a common phenotype. Neurol Sci 31(3):377–380. https://doi.org/10.1007/s10072-010-0248-y
Chu CC, Kuo HC, Yeh TH et al (2007) Heterozygous mutations affecting the epimerase domain of the GNE gene causing distal myopathy with rimmed vacuoles in a Taiwanese family. Clin Neurol Neurosurg 109:250–256. https://doi.org/10.1016/j.clineuro.2006.09.008
Liewluck T, Pho-Iam T, Limwongse C et al (2006) Mutation analysis of the GNE gene in distal myopathy with rimmed vacuoles (DMRV) patients in Thailand. Muscle Nerve 34:775–778. https://doi.org/10.1002/mus.20583
Sim JE, Hong JM, Suh GI et al (2013) A case of GNE myopathy presenting a rapid deterioration during pregnancy. J Clin Neurol 9:280–282. https://doi.org/10.3988/jcn.2013.9.4.280
Yoshioka W, Miyasaka N, Okubo R et al (2020) Pregnancy in GNE myopathy patients: a nationwide repository survey in Japan. Orphanet J Rare Dis 15:245. https://doi.org/10.1186/s13023-020-01487-5
Crook M (1991) Sialic acid: its importance to platelet function in health and disease. Platelets 2:1–10. https://doi.org/10.3109/09537109109005496
Ferroni P, Lenti L, Martini F et al (1997) Ganglioside content of human platelets–differences in resting and activated platelets. Thromb Haemost 77:548–554
Sorensen AL, Rumjantseva V, Nayeb-Hashemi S (2009) Role of sialic acid for platelet life span: exposure of beta-galactose results in the rapid clearance of platelets from the circulation by asialoglycoprotein receptor-expressing liver macrophages and hepatocytes. Blood 114:1645–1654. https://doi.org/10.1182/blood-2009-01-199414
Kotze HF, van Wyk V, Badenhorst PN et al (1993) Influence of platelet membrane sialic acid and platelet-associated IgG on ageing and sequestration of blood platelets in baboons. Thromb Haemost 70:676–680
Zhen C, Guo F, Fang X et al (2014) A family with distal myopathy with rimmed vacuoles associated with thrombocytopenia. Neurol Sci 35:1479–1481. https://doi.org/10.1007/s10072-014-1790-9
Izumi R, Niihori T, Suzuki N et al (2014) GNE myopathy associated with congenital thrombocytopenia: a report of two siblings. Neuromuscul Disord 24:1068–1072. https://doi.org/10.1016/j.nmd.2014.07.008
Ricci E, Broccolini A, Gidaro T et al (2006) NCAM is hyposialylated in hereditary inclusion body myopathy due to GNE mutations. Neurology 66:755–758. https://doi.org/10.1212/01.wnl
Acknowledgements
We thank the patients for their participation in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Ethics Committee of the Third Hospital of Hebei University.
Informed consent
Informed consent was obtained from all patients and their legal representatives.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, X., Zhao, Z., Shen, H. et al. Gene analysis and clinical features of 22 GNE myopathy patients. Neurol Sci 43, 5049–5056 (2022). https://doi.org/10.1007/s10072-022-06023-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06023-w