Abstract
Background
N-Pep-12 is a dietary supplement with neuroprotective and pro-cognitive effects, as shown in experimental models and clinical studies on patients after ischemic stroke. We tested the hypothesis that N-Pep-12 influences quantitative electroencephalography (QEEG) parameters in patients with subacute to chronic supratentorial ischemic lesions.
Methods
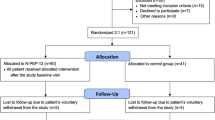
We performed secondary data analysis on an exploratory clinical trial (ISRCTN10702895), assessing the efficacy and safety of 90 days of once-daily treatment with 90 mg N-Pep-12 on neurocognitive function and neurorecovery outcome in patients with post-stroke cognitive impairment against a control group. All participants performed two 32-channel QEEG in resting and active states at baseline (30–120 days after stroke) and 90 days later. Power spectral density on the alpha, beta, theta, delta frequency bands, delta/alpha power ratio (DAR), and (delta+theta)/(alpha+beta) ratio (DTABR) were computed and compared across study groups using means comparison and descriptive methods. Secondarily, associations between QEEG parameters and available neuropsychological tests were explored.
Results
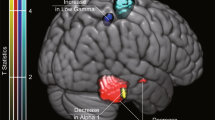
Our analysis showed a statistically significant main effect of EEG segments (p<0.001) in alpha, beta, delta, theta, DA, and DTAB power spectral density. An interaction effect between EEG segments and time was noticed in the alpha power. There was a significant difference in theta spectral power between patients with N-Pep-12 supplementation versus placebo at 0.05 alpha level (p=0.023), independent of time points.
Conclusion
A 90-day, 90 mg daily administration of N-Pep-12 had significant impact on some QEEG indicators in patients after supratentorial ischemic stroke, confirming possible enhancement of post-stroke neurorecovery. Further research is needed to consolidate our findings.


Similar content being viewed by others
Code availability
Not applicable.
References
Cucchiara B, George DK, Kasner SE, Knutsson M, Denison H, Ladenvall P, Amarenco P, Johnston SC (2019) Disability after minor stroke and TIA: a secondary analysis of the SOCRATES trial. Neurology 93:e708–e716. https://doi.org/10.1212/WNL.0000000000007936
Shavelle RM, Brooks JC, Strauss DJ, Turner-Stokes L (2019) Life expectancy after stroke based on age, sex, and rankin grade of disability: a synthesis. J Stroke Cerebrovasc Dis 28:104450. https://doi.org/10.1016/j.jstrokecerebrovasdis.2019.104450
Gorelick PB (2019) The global burden of stroke: persistent and disabling. The Lancet Neurology 18:417–418. https://doi.org/10.1016/S1474-4422(19)30030-4
Mijajlović MD, Pavlović A, Brainin M, Heiss WD, Quinn TJ, Ihle-Hansen HB, Hermann DM, Assayag EB, Richard E, Thiel A, Kliper E, Shin YI, Kim YH, Choi SH, Jung S, Lee YB, Sinanović O, Levine DA, Schlesinger I, Mead G, Milošević V, Leys D, Hagberg G, Ursin MH, Teuschl Y, Prokopenko S, Mozheyko E, Bezdenezhnykh A, Matz K, Aleksić V, Muresanu DF, Korczyn AD, Bornstein NM (2017) Post-stroke dementia - a comprehensive review. BMC Med 15:11–11. https://doi.org/10.1186/s12916-017-0779-7
O’Brien JT, Erkinjuntti T, Reisberg B et al (2003) Vascular cognitive impairment. Lancet Neurol 2:89–98. https://doi.org/10.1016/s1474-4422(03)00305-3
Graff-Radford J (2019) Vascular Cognitive Impairment. Continuum (Minneap Minn) 25:147–164. https://doi.org/10.1212/CON.0000000000000684
Petrovic J, Milosevic V, Zivkovic M, Stojanov D, Milojkovic O, Kalauzi A, Saponjic J (2017) Slower EEG alpha generation, synchronization and “flow”-possible biomarkers of cognitive impairment and neuropathology of minor stroke. PeerJ 5:e3839. https://doi.org/10.7717/peerj.3839
Foreman B, Claassen J (2012) Quantitative EEG for the detection of brain ischemia. Crit Care 16:216. https://doi.org/10.1186/cc11230
Bentes C, Peralta AR, Viana P, Martins H, Morgado C, Casimiro C, Franco AC, Fonseca AC, Geraldes R, Canhão P, Pinho e Melo T, Paiva T, Ferro JM (2018) Quantitative EEG and functional outcome following acute ischemic stroke. Clin Neurophysiol 129:1680–1687. https://doi.org/10.1016/j.clinph.2018.05.021
Hernández-Hernández EM, Caporal Hernandez K, Vázquez-Roque RA, Díaz A, de la Cruz F, Florán B, Flores G (2018) The neuropeptide-12 improves recognition memory and neuronal plasticity of the limbic system in old rats. Synapse 72:e22036. https://doi.org/10.1002/syn.22036
Hutter-Paier B, Reininger-Gutmann B, Wronski R, Doppler E, Moessler H (2015) Long-term treatment of aged Long Evans rats with a dietary supplement containing neuroprotective peptides (N-PEP-12) to prevent brain aging: effects of three months daily treatment by oral gavage. J Med Life 8:207–212
Windisch M, Hutter-Paier B, Grygar E, Doppler E, Moessler H (2005) N-PEP-12 – a novel peptide compound that protects cortical neurons in culture against different age and disease associated lesions. J Neural Transm 112:1331–1343. https://doi.org/10.1007/s00702-005-0283-7
Alvarez XA, Corzo L, Laredo M et al (2005) Neuropeptide dietary supplement N-PEP-12 enhances cognitive function and activates brain bioelectrical activity in healthy elderly subjects. Methods Find Exp Clin Pharmacol 27:483–487. https://doi.org/10.1358/mf.2005.27.7.914776
Balea M, Birle C, Costin C, Marton J, Muresanu IA, Jemna N, Popa LL, Slavoaca D, Rosu OV, Stan A, Vacaras V, Strilciuc S, Muresanu DF (2020) Effects of N-Pep-12 dietary supplementation on neurorecovery after ischemic stroke. Neurol Sci 42:2031–2037. https://doi.org/10.1007/s10072-020-04707-9
Crook TH, Ferris SH, Alvarez XA, Laredo M, Moessler H (2005) Effects of N-PEP-12 on memory among older adults. Int Clin Psychopharmacol 20:97–100. https://doi.org/10.1097/00004850-200503000-00006
Friedrich S, Konietschke F, Pauly M (2019) Resampling-based analysis of multivariate data and repeated measures designs with the R package MANOVA.RM. The R Journal 11:380–400
Aminov A, Rogers JM, Johnstone SJ, Middleton S, Wilson PH (2017) Acute single channel EEG predictors of cognitive function after stroke. PLoS One 12:e0185841. https://doi.org/10.1371/journal.pone.0185841
Cuspineda E, Machado C, Galán L, Aubert E, Alvarez MA, Llopis F, Portela L, García M, Manero JM, Ávila Y (2007) QEEG prognostic value in acute stroke. Clin EEG Neurosci 38:155–160. https://doi.org/10.1177/155005940703800312
Ghorbani M, Clark CCT (2021) Brain function during central fatigue induced by intermittent high-intensity cycling. Neurol Sci. https://doi.org/10.1007/s10072-020-04965-7
Finnigan S, van Putten MJAM (2013) EEG in ischaemic stroke: quantitative EEG can uniquely inform (sub-)acute prognoses and clinical management. Clin Neurophysiol 124:10–19. https://doi.org/10.1016/j.clinph.2012.07.003
Schleiger (2017) Poststroke QEEG informs early prognostication of cognitive impairment. Psychophysiology 54(2):301–309. https://doi.org/10.1111/psyp.12785
Schleiger E, Sheikh N, Rowland T, Wong A, Read S, Finnigan S (2014) Frontal EEG delta/alpha ratio and screening for post-stroke cognitive deficits: the power of four electrodes. Int J Psychophysiol 94:19–24. https://doi.org/10.1016/j.ijpsycho.2014.06.012
Muresanu DF, Strilciuc S, Stan A (2019) Current drug treatment of acute ischemic stroke: challenges and opportunities. CNS Drugs 33:841–847. https://doi.org/10.1007/s40263-019-00663-x
Bernhardt J, Hayward KS, Kwakkel G, Ward NS, Wolf SL, Borschmann K, Krakauer JW, Boyd LA, Carmichael ST, Corbett D, Cramer SC (2017) Agreed definitions and a shared vision for new standards in stroke recovery research: The Stroke Recovery and Rehabilitation Roundtable taskforce. Int J Stroke 12:444–450. https://doi.org/10.1177/1747493017711816
Seleznov I, Zyma I, Kiyono K, Tukaev S, Popov A, Chernykh M, Shpenkov O (2019) Detrended fluctuation, coherence, and spectral power analysis of activation rearrangement in EEG dynamics during cognitive workload. Front Hum Neurosci 13:270. https://doi.org/10.3389/fnhum.2019.00270
Crk (2014) Toward using alpha and theta brain waves to quantify programmer expertise 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society 5373-5376 https://doi.org/10.1109/EMBC.2014.6944840
Mitchell DJ, McNaughton N, Flanagan D, Kirk IJ (2008) Frontal-midline theta from the perspective of hippocampal “theta.”. Prog Neurobiol 86:156–185. https://doi.org/10.1016/j.pneurobio.2008.09.005
Wichniak (2014) Electroencephalographic theta activity and cognition in schizophrenia: preliminary results
Ozgören M, Başar-Eroğlu C, Başar E (2005) Beta oscillations in face recognition. Int J Psychophysiol 55:51–59. https://doi.org/10.1016/j.ijpsycho.2004.06.005
Schneider JM, Abel AD, Ogiela DA, Middleton AE, Maguire MJ (2016) Developmental differences in beta and theta power during sentence processing. Dev Cogn Neurosci 19:19–30. https://doi.org/10.1016/j.dcn.2016.01.001
Bastiaansen MCM, Oostenveld R, Jensen O, Hagoort P (2008) I see what you mean: theta power increases are involved in the retrieval of lexical semantic information. Brain Lang 106:15–28. https://doi.org/10.1016/j.bandl.2007.10.006
Demiralp T, Başar E (1992) Theta rhythmicities following expected visual and auditory targets. Int J Psychophysiol 13:147–160. https://doi.org/10.1016/0167-8760(92)90054-f
Tolonen U, Sulg IA (1981) Comparison of quantitative EEG parameters from four different analysis techniques in evaluation of relationships between EEG and CBF in brain infarction. Electroencephalogr Clin Neurophysiol 51:177–185. https://doi.org/10.1016/0013-4694(81)90007-9
Jordan KG (2004) Emergency EEG and continuous EEG monitoring in acute ischemic stroke. J Clin Neurophysiol 21:341–352
Gotman J, Skuce D, Thompson C (1973) Clinical applications of spectral analysis and extraction of features from electroencephalograms with slow waves in adult patients. Electroencephalogr Clin Neurophysiol 35(3):225–235. https://doi.org/10.1016/0013-4694(73)90233-2
Muresanu DF, Alvarez XA (2008) A pilot study to evaluate the effects of Cerebrolysin on cognition and qEEG in vascular dementia: Cognitive improvement correlates with qEEG acceleration. J Neurol Sci 267(1-2):112–119. https://doi.org/10.1016/j.jns.2007.10.016
Muresanu DF (2010) Persistence of the effects of Cerebrolysin on cognition and qEEG slowing in vascular dementia patients: Results of a 3-month extension study. J Neurol Sci 299(1-2):179–183. https://doi.org/10.1016/j.jns.2010.08.040
Iyer KK (2017) Effective assessments of electroencephalography during stroke recovery: contemporary approaches and considerations. J Neurophysiol 118:2521–2525. https://doi.org/10.1152/jn.00206.2017
Klimesch W (2012) α-band oscillations, attention, and controlled access to stored information. Trends Cogn Sci 16:606–617. https://doi.org/10.1016/j.tics.2012.10.007
Hurford R, Charidimou A, Fox Z, Cipolotti L, Werring DJ (2013) Domain-specific trends in cognitive impairment after acute ischaemic stroke. J Neurol 260:237–241. https://doi.org/10.1007/s00415-012-6625-0
Finnigan S, Robertson IH (2011) Resting EEG theta power correlates with cognitive performance in healthy older adults. Psychophysiology 48:1083–1087. https://doi.org/10.1111/j.1469-8986.2010.01173.x
Cummins TDR, Finnigan S (2007) Theta power is reduced in healthy cognitive aging. Int J Psychophysiol 66:10–17. https://doi.org/10.1016/j.ijpsycho.2007.05.008
Lithfous S, Tromp D, Dufour A, Pebayle T, Goutagny R, Després O (2015) Decreased theta power at encoding and cognitive mapping deficits in elderly individuals during a spatial memory task. Neurobiol Aging 36:2821–2829. https://doi.org/10.1016/j.neurobiolaging.2015.07.007
Kardos Z, Tóth B, Boha R, File B, Molnár M (2014) Age-related changes of frontal-midline theta is predictive of efficient memory maintenance. Neuroscience 273:152–162. https://doi.org/10.1016/j.neuroscience.2014.04.071
Tóth B, Kardos Z, File B, Boha R, Stam CJ, Molnár M (2014) Frontal midline theta connectivity is related to efficiency of WM maintenance and is affected by aging. Neurobiol Learn Mem 114:58–69. https://doi.org/10.1016/j.nlm.2014.04.009
Hughes LE, Rittman T, Robbins TW, Rowe JB (2018) Reorganization of cortical oscillatory dynamics underlying disinhibition in frontotemporal dementia. Brain 141:2486–2499. https://doi.org/10.1093/brain/awy176
Musaeus CS, Engedal K, Høgh P, Jelic V, Mørup M, Naik M, Oeksengaard AR, Snaedal J, Wahlund LO, Waldemar G, Andersen BB (2018) EEG Theta Power Is an Early Marker of Cognitive Decline in Dementia due to Alzheimer’s Disease. J Alzheimers Dis 64:1359–1371. https://doi.org/10.3233/JAD-180300
Song Y, Zang D-W, Jin Y-Y, Wang ZJ, Ni HY, Yin JZ, Ji DX (2015) Background rhythm frequency and theta power of quantitative EEG analysis: predictive biomarkers for cognitive impairment post-cerebral infarcts. Clin EEG Neurosci 46:142–146. https://doi.org/10.1177/1550059413517492
Finnigan S, Wong A, Read S (2016) Defining abnormal slow EEG activity in acute ischaemic stroke: Delta/alpha ratio as an optimal QEEG index. Clin Neurophysiol 127:1452–1459. https://doi.org/10.1016/j.clinph.2015.07.014
Zahiriev Z (1998) One-year dynamic follow-up of patients with reversible ischaemic neurological deficit using quantitative electroencephalography. Folia Med (Plovdiv) 40(2):26–32
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The original N-PEP-12 study was approved on 10/12/2015 by the Ethics Committee of the Iuliu Hatieganu University of Medicine and Pharmacy (8 Babeş Street, 400012 Cluj-Napoca, Romania; +40-264-597-256; contact@umfcluj.ro), ref: 507/10.12.2015. Amended twice refs: 82/24.03.2016;104/12.02.2018.
Conflict of interest
None
Ethical approval
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 94 kb)
The following are available online: Table 3. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the alpha band in each of the 7 EEG segments; Table 4. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the beta band in each of the 7 EEG segments; Table 5. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the delta band in each of the 7 EEG segments; Table 6. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the theta band in each of the 7 EEG segments; Table 7. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the DA band in each of the 7 EEG segments; Table 8. Spearman correlations coefficients (ρ) and p-values between spectral power measured on the DTAB band in each of the 7 EEG segments. (DOCX 94 kb)
Rights and permissions
About this article
Cite this article
Popa, L.L., Iancu, M., Livint, G. et al. N-Pep-12 supplementation after ischemic stroke positively impacts frequency domain QEEG. Neurol Sci 43, 1115–1125 (2022). https://doi.org/10.1007/s10072-021-05406-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-021-05406-9