Abstract
Background
Migraine is a common neurovascular disease associated with vascular risks, especially in young adult females, but the mechanism underlying these associations remains unknown. This study evaluated the relationships between plasma endothelial dysfunction biomarkers and carotid intima–media thickness (IMT) in young adult females with migraine.
Methods
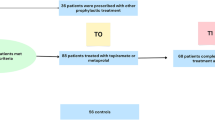
This case–control study included 148 female patients (age range: 18–50 years). Migraine was diagnosed according to the International Headache Society-IIIb criteria. Endothelial dysfunction biomarkers, such as von Willebrand factor (vWF), C-reactive protein (CRP), homocysteine, total nitrate/nitrite concentration, and thiobarbituric acid-reactive substances (TBARS), were evaluated in plasma. Carotid IMT was measured by a radiologist with sonography.
Results
The CRP, TBARS, vWF, and IMT levels were increased in the migraine compared with the control group (p < 0.001, p = 0.02, p < 0.001, and p < 0.001, respectively). After adjusting for confounders, multiple linear regression analysis revealed that systolic arterial blood pressure, CRP, vWF, TBARS, and right and left internal carotid artery (ICA) IMT were independently positively correlated with migraine (p < 0.01, p = 0.004, p = 0.023, p = 0.024, p = 0.032, and p = 0.048, respectively). Multiple logistic regression analysis revealed that right ICA IMT was independently associated with ergotamine and triptan and left ICA IMT was independently associated with ergotamine (p = 0.013, p = 0.026, and p = 0.017, respectively). In addition, significant correlations were found between LDL lipoprotein and carotid IMT in the migraine group (p < 0.05).
Conclusions
Carotid IMT enhancement and elevated TBARS, vWF, and CRP levels in migraine subjects during a migraine attack could be regarded as consequences of migraine attack pathophysiology. The independent associations between triptan and ergotamine consumption and enhanced carotid IMT suggest that repeated use of these vasoconstrictive antimigraine agents may have additional effects on carotid IMT.
Similar content being viewed by others
Abbreviations
- BMI:
-
body mass index
- CCA:
-
common carotid artery
- CRP:
-
C-reactive protein
- ICA:
-
internal carotid artery
- ICHD:
-
International Headache Society
- IMT:
-
intima–media thickness
- LDL:
-
low-density lipoprotein
- NSAID:
-
nonsteroidal anti-inflammatory drug
- TBARS:
-
thiobarbituric acid-reactive substances
- VAS:
-
visual analog score
- vWF:
-
von Willebrand factor
References
Headache Classification Committee of the International Headache Society (2013) The international classification of headache disorders, 3rd edition (beta version). Cephalalgia 33:629–808. https://doi.org/10.1177/0333102413485658
Lipton RB, Stewart WF, Diamond S, Diamond ML, Reed M (2001) Prevalence and burden of migraine in the United States: data from the American migraine study II. Headache 41:646–657
Waeber C, Moskowitz MA (2005) Migraine as an inflammatory disorder. Neurology 64(10 suppl 2):S9–S15
Moskowitz MA (2007) Pathophysiology of headache—past and present. Headache 47:558–567
Spector JT, Kahn SR, Jones MR, Jayakumar M, Dalal D, Nazarian S (2010) Migraine headache and ischemic stroke risk: an updated meta-analysis. Am J Med 123:612–624
Kurth T, Schürks M, Logroscino G, Gaziano JM, Buring JE (2008) Migraine, vascular risk, and cardiovascular events in women: prospective cohort study. BMJ 337:1–9. https://doi.org/10.1136/bmj.a636
Rajan R, Kuhrana D, Lal V (2014) Interictal cerebral and systemic endothelial dysfunction in patients with migraine: a case–control study. J Neurol Neurosurg Psychiatry 1–5. https://doi.org/10.1136/jnnp-2014-309571
Avci AY, Lakadamyali H, Arikan S, Benli US, Kilinc M (2015) High sensitivity C-reactive protein and cerebral white matter hyperintensities on magnetic resonance imaging in migraine patients. J Headache Pain 16:9. https://doi.org/10.1186/1129-2377-16-9
Hamed SA, Hamed EA, Eldin AME, Mahmoud NM (2010) Vascular risk factors, endothelial function, and carotid thickness in patients with migraine: relationship to atherosclerosis. J Stroke Cerebrovasc Dis 19:92–103. https://doi.org/10.1016/j.jstrokecerebrovasdis.2009.04.007
Tietjen GE, Herial NA, White L, Utley C, Kosmyna JM, Khuder SA (2009) Migraine and biomarkers of endothelial activation in young women. Stroke 40:2977–2982. https://doi.org/10.1161/STROKEAHA.109.547901
Yang EY, Nimbi V (2011) Ultrasound imaging of carotid intima–media thickness: an office-based tool to assist physicians in cardiovascular risk assessment. Curr Atheroscler Rep 13:431–436. https://doi.org/10.1007/s11883-011-0200-5
Naqvi TZ, Lee MS (2014) Carotid intima–media thickness and plaque in cardiovascular risk assessment. JACC Cardiovasc Imaging 7(10):1025–1038. https://doi.org/10.1016/j.jcmg.2013.11.014
Tietjen GE, Khubchandani J, Herial N, Palm-Meinders IH, Koppen H, Terwindt GM, Buchem MA, Launer LJ, Ferrari MD, Kruit MC (2017) Migraine and vascular disease biomarkers: a population-based case–control study. Cephalalgia 1–8. https://doi.org/10.1177/0333102417698936
Gonzales-Quintanilla V, Toriello M, Palacio E, Gonzalez-Gay MA, Castillo J, Montes S, Martinez-Nieto R, Fernandez J, Rojo A, Gutierrez S, Pons E, Otreino A (2015) Systemic and cerebral endothelial dysfunction in chronic migraine. A case–control study with an active comparator. Cephalalgia 36:552–560. https://doi.org/10.1177/0333102415607857
Stam AH, Weller CM, Janssens ACJW, Aulchenko YS, Oostra BA, Frants RR, Maagdenberg AMJM, Ferrari MD, Dujin CM, Terwindt GM (2012) Migraine is not associated with enhanced atherosclerosis. Cephalalgia 33:228–235. https://doi.org/10.1177/0333102412466966
Bassuk SS, Rifai N, Ridker PM (2004) High-sensitivity C-reactive protein: clinical importance. Curr Probl Cardiol 29:439–493
Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, Csiba L, Desvarieux M, Ebrahim S, Hernandez R, Jaff M, Kownator S, Naqvi T, Prati P, Rundek T, Sitzer M, Schminke U, Tardif JC, Taylor A, Vicaut E, Woo KS (2012) Mannheim carotid intima–media thickness and plaque consensus (2004-2006-2011). Cerebrovasc Dis 34:290–296. https://doi.org/10.1159/000343145
Albieri V, Olsen TS, Andersen KK (2016) Risk of stroke in migraineurs using triptans. Association with age, sex, stroke severity and subtype. EBioMedicine 6:199–205. https://doi.org/10.1016/j.ebiom.2016.02.039
Valentin JP, Bonnafous R, John GW (1996) Influence of the endothelium and nitric oxide on the contractile responses evoked by 5-HT1D receptor agonist in the rabbit isolated saphenous vein. Br J Pharmacol 119:35–42
Tfelt-Hansen P, Nilsson E, Edvinsson L (2007) Contractile responses to ergotamine and dihydroergotamine in the perfused middle cerebral artery of rat. J Headache Pain 8:83–89. https://doi.org/10.1007/s10194-007-0368-9
Vanmolkot FH, Hoon JN (2010) Endothelial function in migraine: a cross-sectional study. BMC Neurol 10:119. https://doi.org/10.1186/1471-2377-10-119
Fischer M, Gaul C, Shanib H, Holle D, Loacker L, Griesmacher A, Lackner P, Broessner G (2014) Markers of endothelial function in migraine patients: results from a bi-center prospective study. Cephalalgia 35:877–885. https://doi.org/10.1177/0333102414564890
Tzourio C, Gagniere B, El Amrani E, Alperovitch A, Bousser MG (2003) Relationship between migraine, blood pressure and carotid thickness. A population-based study in the elderly. Cephalalgia 23:914–920. https://doi.org/10.1046/j.1468-2982.2003.00613.x
Neri M, Frustaci A, Millic M, Valdiglesias V, Fini M, Bonassi S, Barbanti P (2015) A meta-analysis of biomarkers related to oxidative stress and nitric oxide pathway in migraine. Cephalalgia 35:931–937. https://doi.org/10.1177/0333102414564888
Silva FA, Rueda-Clausen CF, Silva SY, Zarruk JG, Guzman JC, Morillo CA, Vesga B, Pradilla G, Flórez M, López-Jaramillo P (2006) Endothelial function in patients with migraine during the interictal period. Headache 47:45–51. https://doi.org/10.1111/j.1526-4610.2006.00532.x
Heshmat-Ghahdarijani K, Javanmard SH, Sonbolestan SA, Saadatnia M, Sonbolestan SA (2015) Endothelial function in patients with migraine without aura during the interictal period. Int J Prev Med 6(2). https://doi.org/10.4103/2008-7802-151432
Ciancarelli I, Tozzi-Ciancarelli MG, Massimo CD, Marini C, Carolei A (2003) Urinary nitric oxide metabolites and lipid peroxidation by-products in migraine. Cephalalgia 23:39–42. https://doi.org/10.1046/j.1468-2982.2003.00447.x
Lippi G, Mattiuzzi C, Meschi T, Cervellin G, Borghi L (2014) Homocysteine and migraine. A narrative review. Clin Chim Acta 433:5–11. https://doi.org/10.1016/j.cca.2014.02.028
Moschiano F, D’Amico D, Usai S, Grazzi L, Stefano MD, Ciusani E, Erba N, Bussone G (2008) Homocysteine plasma levels in patients with migraine with aura. Neurol Sci 29:173–175. https://doi.org/10.1007/s10072-008-0917-2
Ipcioglu OM, Ozcan O, Gultepe M, Tekeli H, Senol MG (2008) Functional vitamin B12 deficiency represented by elevated urine methylmalonic acid levels in patients with migraine. Turk J Med Sci 38:409–414
Xiang Y, Hwa J (2016) Regulation of vWF expression, and secretion in health and disease. Curr Opin Hematol 23:288–293. https://doi.org/10.1097/MOH.0000000000000230
Cesar JM, Garcia-Avello A, Vecino AM, Sastre JL, Alvarez-Cermeno JC (1995) Increased levels of plasma von Willebrand factor in migraine crisis. Acta Neurol Scand 91:412–413
Furie B, Furie BC (1990) Molecular basis of vitamin K-dependent γ-carboxylation. Blood 75:1753–1762
Gudmundsson LS, Aspelund T, Scher AI, Thorgeirsson G, Johannsson M, Launer LJ, Gudnason V (2009) C-reactive protein in migraine sufferers similar to that of non-migraineurs: the Reykjavik study. Cephalalgia 29:1301–1310. https://doi.org/10.1111/j.1468-2982.2009.01865.x
Goadsby PJ, Edvinsson L, Ekman R (1990) Vasoactive peptide release in the extracerebral circulation of humans during headache. Ann Neurol 28:183–187
Gudmundsson LS, Thorgeirsson G, Sigfusson N, Sigvaldason H, Johannsson M (2005) Migraine patients have lower systolic but higher diastolic blood pressure compared with controls in a population-based study of 21537 subjects. The Reykjavik study. Cephalalgia 26:436–444. https://doi.org/10.1111/j.1468-2982.2005.01057.x
Agostoni E, Aliprandi A (2008) Migraine and hypertension. Neurol Sci 29:S37–S39. https://doi.org/10.1007/s10072-008-0883-8
Finocchi C, Sassos D (2017) Headache and arterial hypertension. Neurol Sci 38(suppl1):s67–s72. https://doi.org/10.1007/s10072-017-2893-x
Folsom AR, Eckfeldt JH, Weitzman S, Ma J, Chambless LE, Barnes RW, Cram KB, Hutchinson RG (1994) Relation of carotid artery wall thickness to diabetes mellitus, fasting glucose and insulin, body size, and physical activity. Atherosclerosis risk in communities (ARIC) study investigators. Stroke 25:66–73
Howard G, Sharrett AR, Heiss G, Evans GW, Chambless LE, Riley WA, Burke GL (1993) Carotid artery intimal–medial thickness distribution in general populations as evaluated by B-mode ultrasound. ARIC investigators. Stroke 24:1297–1304
Funding
This study was supported by the Baskent University Research Fund.
Author information
Authors and Affiliations
Contributions
A.Y.A. and M.H.A. conceived and designed the study; E.T. measured the carotid IMT; S.A. acquired and analyzed the laboratory data; M.A.T analyzed the statistics; A.Y.A. acquired and interpreted the data and wrote the paper; A.Y.A. and U.C. critically revised the manuscript for important intellectual content.
Corresponding author
Ethics declarations
The study was approved by the Baskent University Institutional Review Board and Ethics Committee (project no: KA15/133) and conducted in accordance with the Declaration of Helsinki. All M and control subjects gave informed consent for participation in the study and for carotid artery Doppler ultrasound and laboratory tests.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by Baskent University Institutional Review Board and Ethics Committee (project no: KA15/133) and conducted in accordance with the Declaration of Helsinki of 1964.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yilmaz Avci, A., Akkucuk, M.H., Torun, E. et al. Migraine and subclinical atherosclerosis: endothelial dysfunction biomarkers and carotid intima-media thickness: a case-control study. Neurol Sci 40, 703–711 (2019). https://doi.org/10.1007/s10072-019-3710-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-019-3710-5