Abstract
Background
Disturbance of the triple network model was recently proposed to be associated with the occurrence of posttraumatic stress disorder (PTSD) symptoms. Based on resting-state dynamic causal modeling (rs-DCM) analysis, we investigated the neurobiological model at a neuronal level along with potential neuroimaging biomarkers for identifying individuals with PTSD.
Methods
We recruited survivors of a devastating typhoon including 27 PTSD patients, 33 trauma-exposed controls (TECs), and 30 healthy controls without trauma exposure. All subjects underwent resting-state functional magnetic resonance imaging. Independent components analysis was used to identify triple networks. Detailed effective connectivity patterns were estimated by rs-DCM analysis. Spearman correlation analysis was performed on aberrant DCM parameters with clinical assessment results relevant to PTSD diagnosis. We also carried out step-wise binary logistic regression and receiver operating characteristic curve (ROC) analysis to confirm the capacity of altered effective connectivity parameters to distinguish PTSD patients.
Results
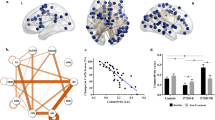
Within the executive control network, enhanced positive connectivity from the left posterior parietal cortex to the left dorsolateral prefrontal cortex was correlated with intrusion symptoms and showed good performance (area under the receiver operating characteristic curve = 0.879) in detecting PTSD patients. In the salience network, we observed a decreased causal flow from the right amygdala to the right insula and a lower transit value for the right amygdala in PTSD patients relative to TECs.
Conclusion
Altered effective connectivity patterns in the triple network may reflect the occurrence of PTSD symptoms, providing a potential biomarker for detecting patients. Our findings shed new insight into the neural pathophysiology of PTSD.




Similar content being viewed by others
References
Benjet C, Bromet E, Karam EG, Kessler RC, McLaughlin KA, Ruscio AM, Shahly V, Stein DJ, Petukhova M, Hill E, Alonso J, Atwoli L, Bunting B, Bruffaerts R, Caldas-de-Almeida JM, de Girolamo G, Florescu S, Gureje O, Huang Y, Lepine JP, Kawakami N, Kovess-Masfety V, Medina-Mora ME, Navarro-Mateu F, Piazza M, Posada-Villa J, Scott KM, Shalev A, Slade T, ten Have M, Torres Y, Viana MC, Zarkov Z, Koenen KC (2016) The epidemiology of traumatic event exposure worldwide: results from the World Mental Health Survey Consortium. Psychol Med 46:327–343. https://doi.org/10.1017/S0033291715001981
Shalev A, Liberzon I, Marmar C (2017) Post-traumatic stress disorder. N Engl J Med 376:2459–2469. https://doi.org/10.1056/NEJMra1612499
American Psychiatric Association (2013) DSM-V, Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Inc, Arlington
Green JD, Annunziata A, Kleiman SE, Bovin Mj, Harwell AM, Fox AML et al (2017) Examining the diagnostic utility of the DSM-5 PTSD symptoms among male and female returning veterans. Depress Anxiety 34:752–760. https://doi.org/10.1002/da.2266
Menon V (2011) Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn Sci 15:483–506. https://doi.org/10.1016/j.tics.2011.08.003
Enyuan Y, Zhengluan L, Yunfei T, Yaju Q, Zhu J, Han Z et al (2017) High-sensitive neuroimaging biomarkers for the identification of amnestic mild cognitive impairment based on resting-state fMRI and a triple network model. Brain Imaging Behav. https://doi.org/10.1007/s11682-017-9727-6
Biswal BB, Mennes M, Zuo XN, Gohel S, Kelly C, Smith SM et al (2017) Toward discovery science of human brain function. Proc Natl Acad Sci U S A 107:4734–4739. https://doi.org/10.1073/pnas.0911855107
Jovanovic T, Kazama A, Bachevalier J, Davis M (2012) Impaired safety signal learning may be a biomarker of PTSD. Neuropharmacology 62:695–704. https://doi.org/10.1016/j.neuropharm.2011.02.023
Sripada RK, King AP, Welsh RC, Garfinkel SN, Wang X, Sprpada CS et al (2012a) Neural dysregulation in posttraumatic stress disorder: evidence for disrupted equilibrium between salience and default mode brain networks. Psychosom Med 74:904–911. https://doi.org/10.1097/PSY.0b013e318273bf33
Koch SB, van Zuiden M, Nawijn L, Nawjin L, Frijling JL, Veltman DJ, Olff M (2016) Aberrant resting-state brain activity in posttraumatic stress disorder: a meta-analysis and systematic review. Depress Anxiety 33:592–605. https://doi.org/10.1002/da.22478
Patel R, Spreng RN, Shin LM, Girard TA (2012) Neurocircuitry models of posttraumatic stress disorder and beyond: a meta-analysis of functional neuroimaging studies. Neurosci Biobehav Rev 36:2130–2142. https://doi.org/10.1016/j.neubiorev.2012.06.003
Sheynin J, Liberzon I (2017) Circuit dysregulation and circuit-based treatments in posttraumatic stress disorder. Neurosci Lett 649:133–138. https://doi.org/10.1016/j.neulet.2016.11.014
Bluhm RL, Williamson PC, Osuch EA (2009) Alterations in default network connectivity in posttraumatic stress disorder related to early-life trauma. J Psychiatry Neurosci 34:187–194
Qin P, Northoff G (2011) How is our self related to midline regions and the default-mode network? NeuroImage 57:1221–1233. https://doi.org/10.1016/j.neuroimage.2011.05.028
Aupperle RL, Melrose AJ, Stein MB, Paulus MP (2012) Executive function and PTSD: disengaging from traums. Neuropharmacology 62:686–694. https://doi.org/10.1016/j.neuropharm.2011.02.008
St Jacques PL, Kragel PA, Rubin DC (2013) Neural networks supporting autobiographical memory retrieval in posttraumatic stress disorder. Cogn Affect Behav Neurosci 13:554–566. https://doi.org/10.3758/s13415-013-0157-7
Lanius RA, Frewen PA, Tursich M, Jetly R, Mckinnon MC (2015) Restoring large-scale brain networks in PTSD and related disorders: a proposal for neuroscientifically-informed treatment interventions. Eur J Psychotraumatol 6:27313. https://doi.org/10.3402/ejpt.v6.27313
Sripada RK, King AP, Garfinkel SN, Wang X, Sripada CS, Welsh RC (2012b) Altered resting-state amygdala functional connectivity in men with posttraumatic stress disorder. J Psychiatry Neurosci 37:241–249. https://doi.org/10.1503/jpn.110069
Rabinak CA, Angstadt M, Welsh RC, Kenndy AE, Lyubkin M, Martis B, Phan KL (2011) Altered amygdala resting-state functional connectivity in post-traumatic stress disorder. Front Psychiatry 2:62. https://doi.org/10.3389/fpsyt.2011.00062
Brown VM, LaBar KS, Haswell CC, Gold AL, Mid-Atlantic, Mirecc Workgroup, McCarthy G et al (2014) Altered resting-state functional connectivity of basolateral and centromedial amygdala complexes in posttraumatic stress disorder. Neuropsychopharmacology 39:351–359. https://doi.org/10.1038/npp.2013.197
Jin C, Qi R, Yin Y, Yin Y, Hu X, Duan L et al (2014) Abnormalities in whole-brain functional connectivity observed in treatment-naive post-traumatic stress disorder patients following an earthquake. Psychol Med 44:1927–1936. https://doi.org/10.1017/S003329171300250X
Zhu H, Qiu C, Meng Y, Cui H, Zhang Y, Huang X, Zhang J, Li T, Gong Q, Zhang W, Lui S (2015) Altered spontaneous neuronal activity in chronic posttraumatic stress disorder patients before and after a 12-week paroxetine treatment. J Affect Disord 174:257–264. https://doi.org/10.1016/j.jad.2014.11.053
Ke J, Chen F, Qi R, Xu Q, Zhong Y, Chen L, Li J, Zhang L, Lu G (2016) Post-traumatic stress influences local and remote functional connectivity: a resting-state functional magnetic resonance imaging study. Brain Imaging Behav 11:1316–1325. https://doi.org/10.1007/s11682-016-9622-6
Chen CC, Henson RN, Stephan KE, Kilner JM, Friston KJ (2009) Forward and backward connections in the brain: a DCM study of functional asymmetries. NeuroImage 45:453–462. https://doi.org/10.1016/j.neuroimage.2008.12.041
Friston KJ, Kahan J, Biswal B, Razi A (2014) A DCM for resting state fMRI. NeuroImage 94:396–407. https://doi.org/10.1016/j.neuroimage.2013.12.009
Udomratn P (2008) Mental health and the psychosocial consequences of natural disasters in Asia. Int Rev Psychiatry 20:441–444. https://doi.org/10.1080/09540260802397487
Weather F, Litz B, Herman D, Huska J, Keane T (1994) The PTSD Checklist-Civilian Version (PCL-C). National Center for PTSD, Boston
Weathers FW, Keane TM, Davidson JR (2001) Clinician-administered PTSD scale: a review of the first ten years of research. Depress Anxiety 13:132–156
First M, Spitzer R, Gibbons M, Williams J (1995) Structured clinical interview for DSM-IV. Biometrics Research Department. New York State Psychiatric Institute, New York
Weschler D (1987) WMS-R: Wechsler Memory Scale-Revised. Psychological Corporation, New York
Zung WW (1971) A rating instrument for anxiety disorders. Psychosomatics 12:371–379. https://doi.org/10.1016/S0033-3182(71)71479-0
Zung WW (1965) A self-rating depression scale. Arch Gen Psychiatry 12:63–70
Yan CG, Zhang YF (2010) DPARSF: a MATLAB toolbox for “pipeline” data analysis of resting-state fMRI. Front Syst Neurosci 4:13. https://doi.org/10.3389/fnsys.2010.00013
Calhoun VD, Adali T, Pearlson GD, Pekar JJ (2001) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14:140–151
Shirer WR, Ryali S, Rykhlevskaia E, Menon V, Greicius MD (2012) Decoding subject-driven cognitive states with whole-brain connectivity patterns. Cereb Cortex 22:158–165. https://doi.org/10.1093/cercor/bhr099
Tsvetanov KA, Henson RN, Tyler LK, Tyler LK, Razi A, Geerligs L et al (2016) Extrinsic and intrinsic brain network connectivity maintains cognition across the lifespan despite accelerated decay of regional brain activation. J Neurosci 36:3115–3126. https://doi.org/10.1523/JNEUROSCI.2733-15.2016
Rosa MJ, Friston K, Penny W (2012) Post-hoc selection of dynamic causal models. J Neurosci Methods 208:66–78. https://doi.org/10.1016/j.jneumeth.2012.04.013
Crone JS, Schurz M, Holler Y, Bergmann J, Monti M, Schmid E et al (2015) Impaired consciousness is linked to changes in effective connectivity of the posterior cingulate cortex within the default mode network. NeuroImage 110:101–109
Friston KJ, Harrison L, Penny W (2003) Dynamic causal modelling. NeuroImage 19:466–477
Tu YK, Wu YC (2017) Using structural equation modeling for network meta-analysis. BMC Med Res Methodol 17:104. https://doi.org/10.1186/s12874-017-0390-9
Friston K (2011) Dynamic causal modeling and Granger causality comments on: the identification of interacting networks in the brain using fMRI: model selection, causality and deconvolution. NeuroImage 58:303–305. https://doi.org/10.1016/j.neuroimage.2009.09.031
Zhang Y, Xie B, Chen H, Li M, Liu F, Chen F (2016) Abnormal functional connectivity density in post-traumatic stress disorder. Brain Topogr 29:405–411. https://doi.org/10.1007/s10548-016-0472-8
Hutchinson JB, Uncapher MR, Wagner AD (2009) Posterior parietal cortex and episodic retrieval: convergent and divergent effects of attention and memory. Learn Mem 16:343–356. https://doi.org/10.1101/lm.919109
Moores KA, Clark CR, McFarlane AC, Brown GC, Puce A, Taylor DJ (2008) Abnormal recruitment of working memory updating networks during maintenance of trauma-neutral information in post-traumatic stress disorder. Psychiatry Res 163:156–170. https://doi.org/10.1016/j.pscychresns.2007.08.011
Fried PJ, Rushmore RJ, Moss MB, Valero-Cabre A, Pascual-Leone A (2014) Causal evidence supporting functional dissociation of verbal and spatial working memory in the human dorsolateral prefrontal cortex. Eur J Neurosci 39:1973–1981. https://doi.org/10.1111/ejn.12584
Scolari M, Seidl-Rathkopf KN, Kastner S (2015) Functions of the human frontoparietal attention network: evidence from neuroimaging. Curr Opin Behav Sci 1:32–39. https://doi.org/10.1016/j.cobeha.2014.08.003
White SF, Costanzo ME, Blair JR, Roy MJ (2015) PTSD symptom severity is associated with increased recruitment of top-down attentional control in a trauma-exposed sample. NeuroImage Clin 7:19–27. https://doi.org/10.1016/j.nicl.2014.11.012
Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, Reiss AL, Greicius MD (2007) Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 27:2349–2356. https://doi.org/10.1523/JNEUROSCI.5587-06.2007
McTeague LM, Goodkind MS, Etkin A (2016) Transdiagnostic impairment of cognitive control in mental illness. J Psychiatr Res 83:37–46. https://doi.org/10.1016/j.jpsychires.2016.08.001
Shiba Y, Oikonomidis L, Sawiak S (2017) Converging prefronto-insula-amygdala pathways in negative emotion regulation in marmoset monkeys. Biol Psychiatry 17:31715–31718. https://doi.org/10.1016/j.biopsych.2017.06.016
Friston KJ, Mechelli A, Turner R, Price GJ (2000) Nonlinear responses in fMRI: the balloon model, Volterra kernels, and other hemodynamics. NeuroImage 12:466–477. https://doi.org/10.1006/nimg.2000.0630
Jun K, Li Z, Rongfeng Q, Qiang X, Yuan Z, Tao L, Jianjun L et al (2018) Typhoon-related post-traumatic stress disorder and trauma might lead to functional integration abnormalities in intra- and inter-resting state networks: a resting-state fMRI independent component analysis. Cell Physiol Biochem 48:99–110. https://doi.org/10.1159/000491666
Funding
This work was supported by the grants from the National Nature Science Foundation of China [Grant Numbers 81671672, 81301209, 81301155, 81460261, and 81201077]; the Key Science and Technology Project of Hainan Province [grant number ZDYF2016156]; and the Chinese Key Grant [Grant Number BWS11J063]; Jiangsu Provincial Medical Youth Talent [grant number QNRC2016888]; Foundations of Commission of Health and Family Planning of Wuxi and Jiangsu Province [grant number Z201610, H201656].
Author information
Authors and Affiliations
Contributions
YW was involved in the experimental design, data analysis, and writing of the manuscript. RQ, ZC, FC, and GL designed the study. JK, QX, and YZ analyzed the data. YL, JL, and LZ acquired the data.
Corresponding authors
Ethics declarations
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. It was approved by the ethics committee of Jinling Hospital, People’s Hospital of Hainan Province, and the Second Xiangya Hospital of Central South University. All participants provided written informed consent after a detailed description of this study.
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Supplemental Table 1
(DOCX 65 kb)
Appendix Figure 1
Results of the post-hoc model selection procedure. In each network model of per group, both the log-posterior and model posterior probabilities are examined. The full model is the winning model in each group with a maximum posterior probability of almost 1. (PNG 271 kb)
High resolution image
(TIF 365 kb)
Appendix Figure 2
Result of neural and hemodynamic parameters. All the detailed neural and hemodynamic parameters are displayed in the form of bar chart. The transit values in right amygdala of PTSD group are lower than that of TEC group. * Significant different (P < 0.05); error bar, standard error (S.E.). (PNG 307 kb)
High resolution image
(TIF 631 kb)
Rights and permissions
About this article
Cite this article
Weng, Y., Qi, R., Zhang, L. et al. Disturbed effective connectivity patterns in an intrinsic triple network model are associated with posttraumatic stress disorder. Neurol Sci 40, 339–349 (2019). https://doi.org/10.1007/s10072-018-3638-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-018-3638-1