Abstract
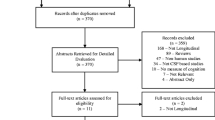
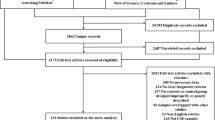
The cerebrospinal fluid (CSF) signature of reduced amyloid beta 1–42 (Aβ42), elevated total tau (t-tau), and phosphorylated tau181 (p-tau) is important for the early diagnosis of Alzheimer’s disease (AD). Aβ42, t-tau, and p-tau have been reported in numerous studies to contribute to predicting cognitive impairment in Parkinson’s disease (PDCI). However, no consistent conclusion can be drawn so far. Literatures regarding Aβ42, t-tau, and p-tau in CSF were systematically reviewed, and a meta-analysis was thus performed to evaluate the changes of these biomarkers in PDCI patients, including PD with mild cognitive impairment (PDMCI) and PD dementia (PDD) patients, relative to PD with normal cognition (PDNC) patients. Databases of “PubMed,” “EBSCO,” and “Springer” were retrieved for articles concerning Aβ42, t-tau, and p-tau in PDCI patients relative to those in PDNC patients published from January 1, 2000 to February 1, 2017. The following keywords were set, namely, “dementia” or “cognitive impairment” or “mild cognitive impairment” and “cerebrospinal fluid” and “Parkinson*.” Sixteen articles comprising 590 PDCI patients and 1182 PDNC patients were included. The results showed that CSF Aβ42 level in PDCI cohort was lower than that in PDNC cohort (pooled Std.MD = −0.44, 95% CI [−0.61, −0.26], p < 0.00001). Reduced Aβ42 (pooled Std.MD = −0.60, 95% CI [−0.75, −0.45], p < 0.00001) as well as elevated t-tau (pooled Std.MD = 0.21, 95% CI [0.06, 0.35], p = 0.006) and p-tau (pooled Std.MD = 0.36, 95% CI [0.02, 0.69], p = 0.04) could be observed in PDD cohort compared with PDNC cohort. Therefore, amyloid pathology and tauopathy may participate in the development of PDD, which is similar to AD.




Similar content being viewed by others
References
Santangelo G, Vitale C, Picillo M, Moccia M, Cuoco S, Longo K et al (2015) Mild cognitive impairment in newly diagnosed Parkinson’s disease: a longitudinal prospective study. Parkinsonism Relat Disord 21:1219–1226
Garcia-Ptacek S, Kramberger MG (2016) Parkinson disease and dementia. J Geriatr Psychiatry Neurol 29:261–270
Pfeiffer HC, Løkkegaard A, Zoetmulder M, Friberg L, Werdelin L (2014) Cognitive impairment in early-stage non-demented Parkinson’s disease patients. Acta Neurol Scand 129:307–318
Pedersen KF, Larsen JP, Tysnes OB, Alves G (2013) Prognosis of mild cognitive impairment in early Parkinson disease: the Norwegian ParkWest study. JAMA Neurol 70:580–586
Kotzbauer PT, Cairns NJ, Campbell MC, Willis AW, Racette BA, Tabbal SD et al (2012) Pathologic accumulation of alpha-synuclein and Abeta in Parkinson disease patients with dementia. Arch Neurol 69:1326–1331
Recasens A, Dehay B, Bové J, Carballo-Carbajal I, Dovero S, Pérez-Villalba A et al (2014) Lewy body extracts from Parkinson disease brains trigger alpha-synuclein pathology and neurodegeneration in mice and monkeys. Ann Neurol 75:351–362
Zhang QS, Heng Y, Yuan YH, Chen NH (2017) Pathological alpha-synuclein exacerbates the progression of Parkinson’s disease through microglial activation. Toxicol Lett 265:30–37
Olsson B, Lautner R, Andreasson U, Öhrfelt A, Portelius E, Bjerke M et al (2016) CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: a systematic review and meta-analysis. Lancet Neurol 15:673–684
Terrelonge M, Marder KS, Weintraub D, Alcalay RN (2016) CSF beta-amyloid 1-42 predicts progression to cognitive impairment in newly diagnosed Parkinson disease. J Mol Neurosci 58:88–92
Bäckström DC, Eriksson Domellöf M, Linder J, Olsson B, Öhrfelt A, Trupp M et al (2015) Cerebrospinal fluid patterns and the risk of future dementia in early, incident Parkinson disease. JAMA Neurol 72:1175–1182
Liu C, Cholerton B, Shi M, Ginghina C, Cain KC, Auinger P et al (2015) CSF tau and tau/Abeta42 predict cognitive decline in Parkinson’s disease. Parkinsonism Relat Disord 21:271–276
Stang A (2010) Critical evaluation of the Newcastle-Ottawa Scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605
Mollenhauer B, Trenkwalder C, von Ahsen N, Bibl M, Steinacker P, Brechlin P et al (2006) Beta-amlyoid 1-42 and tau-protein in cerebrospinal fluid of patients with Parkinson’s disease dementia. Dement Geriatr Cogn Disord 22:200–208
Parnetti L, Tiraboschi P, Lanari A, Peducci M, Padiglioni C, D’Amore C et al (2008) Cerebrospinal fluid biomarkers in Parkinson’s disease with dementia and dementia with Lewy bodies. Biol Psychiatry 64:850–855
Compta Y, Marti MJ, Ibarretxe-Bilbao N, Junque C, Valldeoriola F, Munoz E et al (2009) Cerebrospinal tau, phospho-tau, and beta-amyloid and neuropsychological functions in Parkinson’s disease. Mov Disord 24:2203–2210
Montine TJ, Shi M, Quinn JF, Peskind ER, Craft S, Ginghina C et al (2010) CSF Abeta(42) and tau in Parkinson’s disease with cognitive impairment. Mov Disord 25:2682–2685
Hall S, Öhrfelt A, Constantinescu R, Andreasson U, Surova Y, Bostrom F et al (2012) Accuracy of a panel of 5 cerebrospinal fluid biomarkers in the differential diagnosis of patients with dementia and/or Parkinsonian disorders. Arch Neurol 69:1445–1452
Maetzler W, Tian Y, Baur SM, Gauger T, Odoj B, Schmid B et al (2012) Serum and cerebrospinal fluid levels of transthyretin in Lewy body disorders with and without dementia. PLoS One 7:e48042
Beyer MK, Alves G, Hwang KS, Babakchanian S, Bronnick KS, Chou YY et al (2013) Cerebrospinal fluid Abeta levels correlate with structural brain changes in Parkinson’s disease. Mov Disord 28:302–310
Nutu M, Zetterberg H, Londos E, Minthon L, Nägga K, Blennow K et al (2013) Evaluation of the cerebrospinal fluid amyloid-beta1-42/amyloid-beta1-40 ratio measured by alpha-LISA to distinguish Alzheimer’s disease from other dementia disorders. Dement Geriatr Cogn Disord 36:99–110
Alves G, Lange J, Blennow K, Zetterberg H, Andreasson U, Førland MG et al (2014) CSF Abeta42 predicts early-onset dementia in Parkinson disease. Neurology 82:1784–1790
Vranová HP, Hényková E, Kaiserová M, Menšíková K, Vaštík M, Mareš J et al (2014) Tau protein, beta-amyloid(1)(−)(4)(2) and cluster in CSF levels in the differential diagnosis of Parkinsonian syndrome with dementia. J Neurol Sci 343:120–124
Yarnall AJ, Breen DP, Duncan GW, Khoo TK, Coleman SY, Firbank MJ et al (2014) Characterizing mild cognitive impairment in incident Parkinson disease: the ICICLE-PD study. Neurology 82:308–316
Yu SY, Zuo LJ, Wang F, Chen ZJ, Hu Y, Wang YJ et al (2014) Potential biomarkers relating pathological proteins, neuroinflammatory factors and free radicals in PD patients with cognitive impairment: a cross-sectional study. BMC Neurol 14:113. doi:10.1186/1471-2377-14-113
Skogseth RE, Bronnick K, Pereira JB, Mollenhauer B, Weintraub D, Fladby T et al (2015) Associations between cerebrospinal fluid biomarkers and cognition in early untreated Parkinson’s disease. J Parkinsons Dis 5:783–792
Compta Y, Buongiorno M, Bargalló N, Valldeoriola F, Muñoz E, Tolosa E et al (2016) White matter hyperintensities, cerebrospinal amyloid-beta and dementia in Parkinson’s disease. J Neurol Sci 367:284–290
Buongiorno M, Antonelli F, Compta Y, Fernandez Y, Pavia J, Lomeña F et al (2017) Cross-sectional and longitudinal cognitive correlates of FDDNP PET and CSF amyloid-beta and tau in Parkinson’s disease. J Alzheimers Dis 55:1261–1272
Schrag A, Siddiqui UF, Anastasiou Z, Weintraub D, Schott JM (2017) Clinical variables and biomarkers in prediction of cognitive impairment in patients with newly diagnosed Parkinson’s disease: a cohort study. Lancet Neurol 16:66–75
Lewczuk P, Matzen A, Blennow K, Parnetti L, Molinuevo JL, Eusebi P et al (2017) Cerebrospinal fluid Abeta42/40 corresponds better than Abeta42 to amyloid PET in Alzheimer’s disease. J Alzheimers Dis 55:813–822
Stewart T, Liu C, Ginghina C, Cain KC, Auinger P, Cholerton B et al (2014) Cerebrospinal fluid alpha-synuclein predicts cognitive decline in Parkinson disease progression in the DATATOP cohort. Am J Pathol 184:966–975
Leaver K, Poston KL (2015) Do CSF biomarkers predict progression to cognitive impairment in Parkinson’s disease patients? A systematic review. Neuropsychol Rev 25:411–423
Wills J, Jones J, Haggerty T, Duka V, Joyce JN, Sidhu A (2010) Elevated tauopathy and alpha-synuclein pathology in postmortem Parkinson’s disease brains with and without dementia. Exp Neurol 225:210–218
Haggerty T, Credle J, Rodriguez O, Wills J, Oaks AW, Masliah E et al (2011) Hyperphosphorylated tau in an alpha-synuclein-overexpressing transgenic model of Parkinson’s disease. Eur J Neurosci 33:1598–1610
Caspell-Garcia C, Simuni T, Tosun-Turgut D, Wu IW, Zhang Y, Nalls M et al (2017) Multiple modality biomarker prediction of cognitive impairment in prospectively followed de novo Parkinson disease. PLoS One 12:e0175674. doi:10.1371/journal.pone.0175674
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The research is supported by Jingzhou Science and Technology Bureau Foundation (grant number 2016073).
Rights and permissions
About this article
Cite this article
Hu, X., Yang, Y. & Gong, D. Changes of cerebrospinal fluid Aβ42, t-tau, and p-tau in Parkinson’s disease patients with cognitive impairment relative to those with normal cognition: a meta-analysis. Neurol Sci 38, 1953–1961 (2017). https://doi.org/10.1007/s10072-017-3088-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-3088-1