Abstract
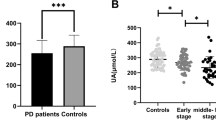
Uric acid (UA) is a natural antioxidant and iron scavenger in the human body, which has been hypothesized to exert an anti-oxidative effect in Parkinson’s disease (PD). This study aimed to investigate the relationship between serum UA levels and freezing of gait (FOG) in PD. A total of 321 Chinese PD patients with fasting serum UA evaluated were included in the cross-sectional study. Demographics, clinical features, and therapeutic regimen were collected. The Unified PD Rating Scale (UPDRS) III and Hoehn and Yahr (H and Y) stage were used to evaluate the severity of disease, and the Frontal Assessment Battery (FAB) and Montreal Cognitive Assessment (MoCA) scales were used to assess the cognitive function. Patients with FOG showed lower proportion of male, longer disease duration, lower body mass index, lower concentrations of serum UA, higher total levodopa equivalent daily dosage, higher UPDRS III score, greater median H and Y stage, lower scores of FAB and MoCA, and higher frequencies of motor fluctuation, dyskinesia, falls, and festination compared to patients without FOG (P < 0.05). The binary logistic regression model indicated that high UPDRS III score (OR = 1.049, P < 0.001), fluctuation (OR = 2.677, P = 0.035), dyskinesia (OR = 6.294, P = 0.003), festination (OR = 3.948, P < 0.001), falls (OR = 7.528, P < 0.001), and low serum UA levels (OR = 0.990, P < 0.001) were associated with FOG. Our study suggests that low serum UA concentration is associated with the occurrence of FOG in PD.
Similar content being viewed by others
References
Ames BN, Cathcart R, Schwiers E, Hochstein P (1981) Uric acid provides an antioxidant defense in humans against oxidant- and radical-caused aging and cancer: a hypothesis. Proc Natl Acad Sci USA 78(11):6858–6862
Bowman GL, Shannon J, Frei B, Kaye JA, Quinn JF (2010) Uric acid as a CNS antioxidant. J Alzheimers Dis JAD 19(4):1331–1336. doi:10.3233/jad-2010-1330
Ascherio A, LeWitt PA, Xu K, Eberly S, Watts A, Matson WR, Marras C, Kieburtz K, Rudolph A, Bogdanov MB, Schwid SR, Tennis M, Tanner CM, Beal MF, Lang AE, Oakes D, Fahn S, Shoulson I, Schwarzschild MA (2009) Urate as a predictor of the rate of clinical decline in Parkinson disease. Arch Neurol 66(12):1460–1468. doi:10.1001/archneurol.2009.247
de Lau LM, Koudstaal PJ, Hofman A, Breteler MM (2005) Serum uric acid levels and the risk of Parkinson disease. Ann Neurol 58(5):797–800. doi:10.1002/ana.20663
Picillo M, Santangelo G, Moccia M, Erro R, Amboni M, Prestipino E, Longo K, Vitale C, Spina E, Orefice G, Barone P, Pellecchia MT (2016) Serum uric acid is associated with apathy in early, drug-naive Parkinson’s disease. J Neural Transm (Vienna) 123(4):371–377. doi:10.1007/s00702-015-1502-5
Gonzalez-Aramburu I, Sanchez-Juan P, Sierra M, Fernandez-Juan E, Sanchez-Quintana C, Berciano J, Combarros O, Infante J (2014) Serum uric acid and risk of dementia in Parkinson’s disease. Parkinsonism Relat Dis 20(6):637–639. doi:10.1016/j.parkreldis.2014.02.023
Giladi N, Nieuwboer A (2008) Understanding and treating freezing of gait in parkinsonism, proposed working definition, and setting the stage. Mov Dis Off J Mov Dis Soc 23(Suppl 2):S423–S425. doi:10.1002/mds.21927
Morris ME, Iansek R, Galna B (2008) Gait festination and freezing in Parkinson’s disease: pathogenesis and rehabilitation. Mov Dis Off J Mov Dis Soc 23(Suppl 2):S451–S460. doi:10.1002/mds.21974
Amboni M, Cozzolino A, Longo K, Picillo M, Barone P (2008) Freezing of gait and executive functions in patients with Parkinson’s disease. Mov Dis Off J Mov Dis Soc 23(3):395–400. doi:10.1002/mds.21850
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55(3):181–184
Goetz CG, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stebbins GT, Stern MB, Tilley BC, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, Van Hilten JJ, LaPelle N (2007) Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): process, format, and clinimetric testing plan. Mov Dis Off J Mov Dis Soc 22(1):41–47. doi:10.1002/mds.21198
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17(5):427–442
Dubois B, Slachevsky A, Litvan I, Pillon B (2000) The FAB: a Frontal Assessment Battery at bedside. Neurology 55(11):1621–1626
Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53(4):695–699. doi:10.1111/j.1532-5415.2005.53221.x
Giladi N, Tal J, Azulay T, Rascol O, Brooks DJ, Melamed E, Oertel W, Poewe WH, Stocchi F, Tolosa E (2009) Validation of the freezing of gait questionnaire in patients with Parkinson’s disease. Mov Disord 24(5):655–661. doi:10.1002/mds.21745
Church WH, Ward VL (1994) Uric acid is reduced in the substantia nigra in Parkinson’s disease: effect on dopamine oxidation. Brain Res Bull 33(4):419–425
Lolekha P, Wongwan P, Kulkantrakorn K (2015) Association between serum uric acid and motor subtypes of Parkinson’s disease. J Clin Neurosci Off J Neurosurg Soc Australas 22(8):1264–1267. doi:10.1016/j.jocn.2015.02.015
Fukae J, Ishikawa K, Hatano T, Yoritaka A, Takanashi M, Shimo Y, Tsugawa J, Tsuboi Y, Hattori N (2014) Serum uric acid concentration is linked to wearing-off fluctuation in Japanese Parkinson’s disease patients. J Parkinsons Dis 4(3):499–505. doi:10.3233/jpd-140353
Moccia M, Picillo M, Erro R, Vitale C, Longo K, Amboni M, Santangelo G, Spina E, De Rosa A, De Michele G, Santoro L, Barone P, Pellecchia MT (2014) Is serum uric acid related to non-motor symptoms in de-novo Parkinson’s disease patients? Parkinsonism Relat Dis 20(7):772–775. doi:10.1016/j.parkreldis.2014.03.016
Cao B, Guo X, Chen K, Song W, Huang R, Wei QQ, Zhao B, Shang HF (2013) Uric acid is associated with the prevalence but not disease progression of multiple system atrophy in Chinese population. J Neurol 260(10):2511–2515. doi:10.1007/s00415-013-7006-z
Oropesa-Ruiz JM, Huertas-Fernandez I, Jesus S, Caceres-Redondo MT, Vargas-Gonzalez L, Carrillo F, Carballo M, Gomez-Garre P, Mir P (2016) Low serum uric acid levels in progressive supranuclear palsy. Mov Dis Off J Mov Dis Soc 31(3):402–405. doi:10.1002/mds.26466
Sakuta H, Suzuki K, Miyamoto T, Miyamoto M, Numao A, Fujita H, Watanabe Y, Hirata K (2017) Serum uric acid levels in Parkinson’s disease and related disorders. Brain Behav 7(1):e00598. doi:10.1002/brb3.598
Jenner P (2003) Oxidative stress in Parkinson’s disease. Ann Neurol 53(Suppl 3):S26–S36. doi:10.1002/ana.10483 (discussion S36–S28)
Hink HU, Santanam N, Dikalov S, McCann L, Nguyen AD, Parthasarathy S, Harrison DG, Fukai T (2002) Peroxidase properties of extracellular superoxide dismutase: role of uric acid in modulating in vivo activity. Arterioscler Thromb Vasc Biol 22(9):1402–1408
Iravani MM, Haddon CO, Rose S (1067) Jenner P (2006) 3-Nitrotyrosine-dependent dopaminergic neurotoxicity following direct nigral administration of a peroxynitrite but not a nitric oxide donor. Brain Res 1:256–262. doi:10.1016/j.brainres.2005.10.086
Davies KJ, Sevanian A, Muakkassah-Kelly SF, Hochstein P (1986) Uric acid-iron ion complexes. A new aspect of the antioxidant functions of uric acid. Biochem J 235(3):747–754
Jones DC, Gunasekar PG, Borowitz JL, Isom GE (2000) Dopamine-induced apoptosis is mediated by oxidative stress and Is enhanced by cyanide in differentiated PC12 cells. J Neurochem 74(6):2296–2304
Duan W, Ladenheim B, Cutler RG, Kruman II, Cadet JL, Mattson MP (2002) Dietary folate deficiency and elevated homocysteine levels endanger dopaminergic neurons in models of Parkinson’s disease. J Neurochem 80(1):101–110
Facheris MF, Hicks AA, Minelli C, Hagenah JM, Kostic V, Campbell S, Hayward C, Volpato CB, Pattaro C, Vitart V, Wright A, Campbell H, Klein C, Pramstaller PP (2011) Variation in the uric acid transporter gene SLC2A9 and its association with AAO of Parkinson’s disease. J Mol Neurosci MN 43(3):246–250. doi:10.1007/s12031-010-9409-y
Shine JM, Naismith SL, Lewis SJ (2011) The pathophysiological mechanisms underlying freezing of gait in Parkinson’s Disease. J Clin Neurosci Off J Neurosurg Soc Australas 18(9):1154–1157. doi:10.1016/j.jocn.2011.02.007
Teramoto H, Morita A, Ninomiya S, Shiota H, Kamei S (2014) Relation between freezing of gait and frontal function in Parkinson’s disease. Parkinsonism Relat Dis 20(10):1046–1049. doi:10.1016/j.parkreldis.2014.06.022
Yogev-Seligmann G, Hausdorff JM, Giladi N (2008) The role of executive function and attention in gait. Mov Dis Off J Mov Dis Soc 23(3):329–342. doi:10.1002/mds.21720 (quiz 472)
Annanmaki T, Pessala-Driver A, Hokkanen L, Murros K (2008) Uric acid associates with cognition in Parkinson’s disease. Parkinsonism Relat Dis 14(7):576–578. doi:10.1016/j.parkreldis.2007.11.001
Acknowledgements
The authors thank the patients and their families for their participation in the study. The present study was supported by the funding of the National Science Fund of China (Grant No. 81571247), supported in part by the National Basic Research Program of China (No: 2015CB856400), and the Science and Technology Bureau Fund of Sichuan Province (2014FZ0072).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ou, R., Cao, B., Wei, Q. et al. Serum uric acid levels and freezing of gait in Parkinson’s disease. Neurol Sci 38, 955–960 (2017). https://doi.org/10.1007/s10072-017-2871-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-2871-3