Abstract
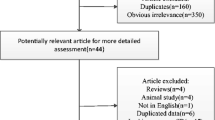
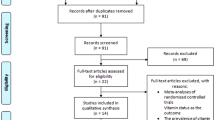
Although some studies have reported the associations between specific metal element intake and risk of Parkinson’s disease (PD), the associations between specific metal element intake such as iron intake and PD are still conflicted. We aimed to determine whether intake of iron, zinc, and copper increases/decreases the risk of PD. PubMed, Embase, Web of Knowledge, and Google Scholar were searched. We pooled the multivariate-adjusted relative risks (RRs) or odds ratios using random effects. Study quality was evaluated by the Newcastle–Ottawa Scale. Five studies including 126,507 individuals remained for inclusion, pooled RRs of Parkinson’s disease for moderate dietary iron intake was 1.08 (95 % CI 0.61–1.93, P = 0.787), and for high dietary iron intake was (1.03, 95 % CI 0.83–1.30, P = 0.766), respectively. The pooled RRs of Parkinson’s disease for the highest compared with the lowest dietary iron intake were 1.47 (95 % CI 1.17–1.85, P = 0.001) in western population and in males (RR = 1.43, 95 % CI 1.01–2.01, P = 0.041). The pooled RRs of Parkinson’s disease for moderate or high intake of zinc, and copper were not statistically different (P > 0.05). PD increased by 18 % (RR 1.18, 95 % CI 1.02–1.37) for western population by every 10-mg/day increment in iron intake. Higher iron intake appears to be not associated with overall PD risk, but may be associated with risk of PD in western population. Sex may be a factor influencing PD risk for higher iron intake. However, further studies are still needed to confirm the sex-selective effects.



Similar content being viewed by others
References
Samii A, Nutt JG, Ransom BR (2004) Parkinson’s disease. Lancet 363:1783–1793
Gorell JM, Johnson CC, Rybicki BA, Peterson EL, Richardson RJ (1998) The risk of Parkinson’s disease with exposure to pesticides, farming, well water, and rural living. Neurology 50:1346–1350
Kamel F (2013) Epidemiology. Paths from pesticides to Parkinson’s. Science 341:722–723
Steece-Collier K, Maries E, Kordower JH (2002) Etiology of Parkinson’s disease: genetics and environment revisited. Proc Natl Acad Sci USA 99:13972–13974
Trinh J, Farrer M (2013) Advances in the genetics of Parkinson disease. Nat Rev Neurol 9:445–454
Van Den Eeden SK, Tanner CM, Bernstein AL, Fross RD, Leimpeter A, Bloch DA et al (2003) Incidence of Parkinson’s disease: variation by age, gender, and race/ethnicity. Am J Epidemiol 157:1015–1022
Barnham KJ, Bush AI (2008) Metals in Alzheimer’s and Parkinson’s diseases. Curr Opin Cheml Biolo 12:222–228
Fang Y-Z, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18:872–879
Machlin LJ, Bendich A (1987) Free radical tissue damage: protective role of antioxidant nutrients. FASEB J 1:441–445
Oakley AE, Collingwood JF, Dobson J, Love G, Perrott HR, Edwardson JA et al (2007) Individual dopaminergic neurons show raised iron levels in Parkinson disease. Neurology 68:1820–1825
Yu X, Du T, Song N, He Q, Shen Y, Jiang H et al (2013) Decreased iron levels in the temporal cortex in postmortem human brains with Parkinson disease. Neurology 80:492–495
Hunt JR (2003) Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. Am J Clin Nutr 78:633s–639s
Lee HP, Zhu X, Liu G, Chen SG, Perry G, Smith MA et al (2010) Divalent metal transporter, iron, and Parkinson’s disease: a pathological relationship. Cell Res 20:397–399
Sandstrom B, Arvidsson B, Cederblad A, Bjorn-Rasmussen E (1980) Zinc absorption from composite meals. I. The significance of wheat extraction rate, zinc, calcium, and protein content in meals based on bread. Am J Clin Nutr 33:739–745
Johnson CC, Gorell JM, Rybicki BA, Sanders K, Peterson EL (1999) Adult nutrient intake as a risk factor for Parkinson’s disease. Int J Epidemiol 28:1102–1109
Logroscino G, Gao X, Chen H, Wing A, Ascherio A (2008) Dietary iron intake and risk of Parkinson’s disease. Am J Epidemiol 168:1381–1388
Miyake Y, Tanaka K, Fukushima W, Sasaki S, Kiyohara C, Tsuboi Y et al (2011) Dietary intake of metals and risk of Parkinson’s disease: a case–control study in Japan. J Neurol Sci 306:98–102
Powers KM, Smith-Weller T, Franklin GM, Longstreth WT Jr, Swanson PD, Checkoway H (2003) Parkinson’s disease risks associated with dietary iron, manganese, and other nutrient intakes. Neurology 60:1761–1766
Powers KM, Smith-Weller T, Franklin GM, Longstreth WT Jr, Swanson PD, Checkoway H (2009) Dietary fats, cholesterol and iron as risk factors for Parkinson’s disease. Parkinsonism Relat Disord 15:47–52
Ownby RL, Crocco E, Acevedo A, John V, Loewenstein D (2006) Depression and risk for Alzheimer disease: systematic review, meta-analysis, and metaregression analysis. Arch Gen Psychiatry 63:530–538
Stang A (2010) Critical evaluation of the Newcastle–Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 25:603–605
Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M (2015) The Newcastle–Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford_web.ppt. Accessed Apr 12 2015
Etminan M, Gill SS, Samii A (2005) Intake of vitamin E, vitamin C, and carotenoids and the risk of Parkinson’s disease: a meta-analysis. Lancet Neurol 4:362–365
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Greenland S, Longnecker MP (1992) Methods for trend estimation from summarized dose–response data, with applications to meta-analysis. Am J Epidemiol 135:1301–1309
Orsini N, Bellocco R, Greenland S (2006) Generalized least squares for trend estimation of summarized dose-response data. Stata J 6:40
Berlin JA, Longnecker MP, Greenland S (1993) Meta-analysis of epidemiologic dose–response data. Epidemiology 4:218–228
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629-634
Kozlowski H, Janicka-Klos A, Brasun J, Gaggelli E, Valensin D, Valensin G (2009) Copper, iron, and zinc ions homeostasis and their role in neurodegenerative disorders (metal uptake, transport, distribution and regulation). Coord Chem Rev 253:2665–2685
Gaggelli E, Kozlowski H, Valensin D, Valensin G (2006) Copper homeostasis and neurodegenerative disorders (Alzheimer’s, prion, and Parkinson’s diseases and amyotrophic lateral sclerosis). Chem Rev 106:1995–2044
Kozlowski H, Luczkowski M, Remelli M, Valensin D (2012) Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases). Coord Chem Rev 256:2129–2141
Dexter DT, Carayon A, Javoy-Agid F, Agid Y, Wells FR, Daniel SE et al (1991) Alterations in the levels of iron, ferritin and other trace metals in Parkinson’s disease and other neurodegenerative diseases affecting the basal ganglia. Brain 114(Pt 4):1953–1975
Van Den Eeden SK, Tanner CM, Bernstein AL, Fross RD, Leimpeter A, Bloch DA et al (2003) Incidence of Parkinson’s disease: variation by age, gender, and race/ethnicity. Am J Epidemiol 157:1015–1022
Blanco-Rojo R, Toxqui L, López-Parra AM, Baeza-Richer C, Pérez-Granados AM, Arroyo-Pardo E et al (2014) Influence of diet, menstruation and genetic factors on iron status: a cross-sectional study in Spanish women of childbearing age. Int J Mol Sci 15:4077–4087
Coad J, Conlon C (2011) Iron deficiency in women: assessment, causes and consequences. Curr Opin Clin Nutr Metab Care 14:625–634
Noyce AJ, Bestwick JP, Silveira-Moriyama L, Hawkes CH, Giovannoni G, Lees AJ et al (2012) Meta-analysis of early nonmotor features and risk factors for Parkinson disease. Ann Neurol 72:893–901
Acknowledgments
We thank the scientific editors at Impactys (www.impactys.com) for editing and proofreading this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
P. F. Cheng and J. Yu contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Cheng, P., Yu, J., Huang, W. et al. Dietary intake of iron, zinc, copper, and risk of Parkinson’s disease: a meta-analysis. Neurol Sci 36, 2269–2275 (2015). https://doi.org/10.1007/s10072-015-2349-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-015-2349-0