Abstract
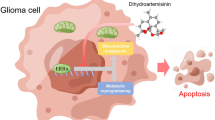
Dihydroartemisinin (DHA) is a semi-synthetic derivative of artemisinin, a well-tolerated and effective drug for malaria treatment, and has recently been shown to have antitumorigenic activity. However, the mechanistic basis of these activities in gliomas is unknown. The objective of this study was to evaluate whether DHA inhibits cell proliferation and invasion in glioma cells, and to elucidate the underlying mechanisms. The results demonstrate that DHA treatment significantly inhibited cell proliferation, migration and invasion, as determined using viability, transwell migration, and matrix penetration assays, respectively. Western blot analysis revealed that protein expression levels of a disintegrin and metalloproteinase 17 (ADAM17), and phosphorylated epidermal growth factor receptor and AKT (p-EGFR and p-AKT, respectively), were suppressed by DHA. EGFR and AKT phosphorylation was enhanced by stimulation with the ADAM17 agonist chemokine phorbol myristate acetate. These data suggest that DHA inhibits glioma proliferation and invasion through suppression of ADAM17 and downregulation of EGFR-PI3 K-AKT signaling.



Similar content being viewed by others
References
Arko L, Katsyv I, Park GE, Luan WP, Park JK (2010) Experimental approaches for the treatment of malignant gliomas. Pharmacol Ther 128(1):1–36
Mentlein R, Hattermann K, Held-Feindt J (2011) Lost in disruption: role of proteases in glioma invasion and progression. Biochim Biophys Acta 1825(2):178–185
Claes A, Idema AJ, Wesseling P (2007) Diffuse glioma growth: a guerilla war. Acta Neuropathol 114(5):443–458
Zheng X, Jiang F, Katakowski M, Zhang X, Jiang H, Zhang ZG, Chopp M (2008) Sensitization of cerebral tissue in nude mice with photodynamic therapy induces ADAM17/TACE and promotes glioma cell invasion. Cancer Lett 265(2):177–187
Zheng X, Jiang F, Katakowski M, Kalkanis SN, Hong X, Zhang X, Zhang ZG, Yang H, Chopp M (2007) Inhibition of ADAM17 reduces hypoxia-induced brain tumor cell invasiveness. Cancer Sci 98(5):674–684
Baumgart A, Seidl S, Vlachou P, Michel L, Mitova N, Schatz N, Specht K, Koch I, Schuster T, Grundler R (2010) ADAM17 regulates epidermal growth factor receptor expression through the activation of Notch1 in non-small cell lung cancer. Cancer Res 70(13):5368–5378
Chen X, Chen L, Zhang R, Yi Y, Ma Y, Yan K, Jiang X, Wang X (2013) ADAM17 regulates self-renewal and differentiation of U87 glioblastoma stem cells. Neurosci Lett 537:44–49
Asai M, Hattori C, Szabo B, Sasagawa N, Maruyama K, Tanuma S, Ishiura S (2003) Putative function of ADAM9, ADAM10, and ADAM17 as APP alpha-secretase. Biochem Biophys Res Commun 301(1):231–235
Bulstrode H, Jones LM, Siney EJ, Sampson JM, Ludwig A, Gray WP, Willaime-Morawek S (2012) A-Disintegrin and Metalloprotease (ADAM) 10 and 17 promote self-renewal of brain tumor sphere forming cells. Cancer Lett 326(1):79–87
Kenny PA (2007) TACE: a new target in epidermal growth factor receptor dependent tumors. Differentiation 75(9):800–808
Ludwig A, Hundhausen C, Lambert MH, Broadway N, Andrews RC, Bickett DM, Leesnitzer MA, Becherer JD (2005) Metalloproteinase inhibitors for the disintegrin-like metalloproteinases ADAM10 and ADAM17 that differentially block constitutive and phorbol ester-inducible shedding of cell surface molecules. Comb Chem High Throughput Screen 8(2):161–171
Scheller J, Chalaris A, Garbers C, Rose-John S (2011) ADAM17 a molecular switch to control inflammation and tissue regeneration. Trends Immunol 32(8):380–387
Katakowski M, Jiang F, Zheng X, Gutierrez JA, Szalad A, Chopp M (2009) Tumorigenicity of cortical astrocyte cell line induced by the protease ADAM17. Cancer Sci 100(9):1597–1604
Zheng X, Jiang F, Katakowski M, Lu Y, Chopp M (2012) ADAM17 promotes glioma cell malignant phenotype. Mol Carcinog 51(2):150–164
Houle CD (2011) Neuropathology standards: what constitutes an optimal histomorphologic evaluation of the nervous system in general toxicity studies. Toxicol Pathol 39(6):1010–1012
Meshnick SR (2002) Artemisinin: mechanisms of action, resistance and toxicity. Int J Parasitol 32(13):1655–1660
Zhou HJ, Wang WQ, Wu GD, Lee J, Li A (2007) Artesunate inhibits angiogenesis and downregulates vascular endothelial growth factor expression in chronic myeloid leukemia K562 cells. Vascul Pharmacol 47(2–3):131–138
He Q, Shi J, Shen XL, An J, Sun H, Wang L, Hu YJ, Sun Q, Fu LC, Sheikh MS (2010) Dihydroartemisinin upregulates death receptor 5 expression and cooperates with TRAIL to induce apoptosis in human prostate cancer cells. Cancer Biol Ther 9(10):819–824
Jiao Y, Ge CM, Meng QH, Cao JP, Tong J, Fan SJ (2007) Dihydroartemisinin is an inhibitor of ovarian cancer cell growth. Acta Pharmacol Sin 28(7):1045–1056
Du JH, Zhang HD, Ma ZJ, Ji KM (2010) Artesunate induces oncosis-like cell death in vitro and has antitumor activity against pancreatic cancer xenografts in vivo. Cancer Chemother Pharmacol 65(5):895–902
Lu YY, Chen TS, Qu JL, Pan WL, Sun L, Wei XB (2009) Dihydroartemisinin (DHA) induces caspase-3-dependent apoptosis in human lung adenocarcinoma ASTC-a-1 cells. J Biomed Sci 16:16
Zhao Y, Jiang W, Li B, Yao Q, Dong J, Cen Y, Pan X, Li J, Zheng J, Pang X (2011) Artesunate enhances radiosensitivity of human non-small cell lung cancer A549 cells via increasing NO production to induce cell cycle arrest at G2/M phase. Int Immunopharmacol 11(12):2039–2046
Chen XR, Lin H, Wen J, Rong QS, Xu WW, Wang XY (2012) Dihydroartemisinin suppresses cell proliferation, invasion, and angiogenesis in human glioma U87 cells. Afr J Pharm Pharmcol 6(32):2433–2440
Costa BM, Viana-Pereira M, Fernandes R, Costa S, Linhares P, Vaz R, Pinheiro C, Lima J, Soares P, Silva A (2011) Impact of EGFR genetic variants on glioma risk and patient outcome. Cancer Epidemiol Biomark Prev 20(12):2610–2617
Birner P, Toumangelova-Uzeir K, Natchev S, Guentchev M (2011) Expression of mutated isocitrate dehydrogenase-1 in gliomas is associated with p53 and EGFR expression. Folia Neuropathol 49(2):88–93
Zheng X, Jiang F, Katakowski M, Lu Y, Chopp M (2012) ADAM17 promotes glioma cell malignant phenotype. Mol Carcinog 51(2):150–164
Acknowledgments
This work was supported by grants from the funds for Youth scientific research support program of Fujian Province Hygienic Bureau (No. 2013-2-49) to Professor Xiangrong Chen and Fujian Province Natural Science Foundation to Professor Weipeng Hu (No. 2014J01408).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, J., Chen, X., Wang, F. et al. Dihydroartemisinin suppresses glioma proliferation and invasion via inhibition of the ADAM17 pathway. Neurol Sci 36, 435–440 (2015). https://doi.org/10.1007/s10072-014-1963-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-014-1963-6