Abstract
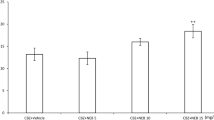
There have been conflicting reports regarding the role of ethanol in seizure. Another effect of ethanol is vascular damage in cerebral tissue. This study investigates the influence of ethanol on antiepileptic efficacy of valproic acid (VPA) and cerebral microvascular structure. In this study, four groups of mice (25–30 g) received pentylenetetrazole (PTZ) i.p. (37 mg/kg) every other day. Different groups of animals received an injection of saline, ethanol (1 g/kg), VPA (100 mg/kg), or VPA and ethanol 30 min before PTZ. Animals in groups 5 and 6 received only ethanol and saline, respectively. After recording seizure parameters, the animals were sacrificed under deep anesthesia and the brains of the animals were removed and fixed, thereafter coronal sections were prepared from cerebral cortex. Then, the cerebral microvessels were counted in microscopic sections after hematoxylin–eosin staining. Ethanol injection (1 g/kg) for 7 days decreased stage 4 duration and increased latency to the onset of stage 1 and stage 4 of seizure (p < 0.001). Concomitant injection of VPA (5 min before ethanol) and ethanol had significantly stronger anticonvulsant effects than VPA alone (p < 0.001). Furthermore, the findings showed that not only the cerebral microvessels increased significantly in ethanol group compared with saline group (p < 0.05), but also there were morphological changes in vascular endothelium in ethanol group. The obtained results show that short-term ethanol administration has anticonvulsant effects along with VPA, and enhances the anticonvulsant effects of VPA. Furthermore, it is possible that VPA leads to decreased ethanol-induced vascular damage.




Similar content being viewed by others
References
McNamara JO (1986) Kindling model of epilepsy. Adv Neurol 44:303–318
Delgado-Escueta AV, Wilson WA, Olsen RW, Porter RJ (1999) New waves of research in the epilepsies: crossing into the third millennium. Adv Neurol 79:3–58
Barkai E, Grossman Y, Gutnick MJ (1994) Long-term changes in neocortical activity after chemical kindling with systemic pentylenetetrazole: an in vitro study. J Neurophysiol 72(1):72–83
Goddard GV, McIntyre DC, Leech CK (1969) A permanent change in brain function resulting from daily electrical stimulation. Exp Neurol 25(3):295–330
Fischer W (2005) Influence of ethanol on the threshold for electroshock-induced seizures and electrically-evoked hippocampal afterdischarges. J Neural Transm 112(9):1149–1163
Kozan R, Ayyildiz M, Yildirim M, Agar E (2006) The effects of ethanol intake and withdrawal on penicillin-induced epileptiform activity in rats. Brain Res Bull 71(1–3):111–115
Town M, Naimi TS, Mokdad AH, Brewer RD (2006) Health care access among US adults who drink alcohol excessively: missed opportunities for prevention. Prev Chronic Dis 3(2):A53
Fried VM, Prager K, MacKay AP, Xia H (2003) Health, United States: With Chartbook on Trends in the Health of Americans. National Center for Health Statistics, Hyattsville
Fischer W, Kittner H (1998) Influence of ethanol on the pentylenetetrazol-induced kindling in rats. J Neural Transm 105(10–12):1129–1142
Hsieh CL, Chang CH, Chiang SY, Li TC, Tang NY, Pon CZ, Hsieh CT, Lin JG (2000) Anticonvulsive and free radical scavenging activities of vanillyl alcohol in ferric chloride-induced epileptic seizures in Sprague–Dawley rats. Life Sci 67(10):1185–1195
Scorza FA, Arida RM, Cysneiros RM, Priel MR, de Albuquerque M, Cavalheiro EA (2003) The effects of alcohol intake and withdrawal on the seizures frequency and hippocampal morphology in rats with epilepsy. Neurosci Res 47(3):323–328
Freeman FG (1978) Effects of alcohol on kindled seizure thresholds in rats. Pharmacol Biochem Behav 8(6):641–644
Hoppener RJ, Kuyer A, van der Lugt PJ (1983) Epilepsy and alcohol: the influence of social alcohol intake on seizures and treatment in epilepsy. Epilepsia 24(4):459–471
McQuarrie DG, Fingl E (1958) Effects of single doses and chronic administration of ethanol on experimental seizures in mice. J Pharmacol Exp Ther 124(3):264–271
Jaatinen P, Rintala J (2008) Mechanisms of ethanol-induced degeneration in the developing, mature, and aging cerebellum. Cerebellum (London, England) 7(3):332–347
Morleo M, Woolfall K, Dedman D, Mukherjee R, Bellis MA, Cook PA (2011) Under-reporting of foetal alcohol spectrum disorders: an analysis of hospital episode statistics. BMC Pediatr 11:14
Faria A, Pestana D, Teixeira D, Azevedo J, De Freitas V, Mateus N, Calhau C (2010) Flavonoid transport across RBE4 cells: a blood–brain barrier model. Cell Mol Biol Lett 15(2):234–241
Williams AJ, Tortella FC, Lu XM, Moreton JE, Hartings JA (2004) Antiepileptic drug treatment of nonconvulsive seizures induced by experimental focal brain ischemia. J Pharmacol Exp Ther 311(1):220–227
Go HS, Seo JE, Kim KC, Han SM, Kim P, Kang YS, Han SH, Shin CY, Ko KH (2011) Valproic acid inhibits neural progenitor cell death by activation of NF-kappaB signaling pathway and up-regulation of Bcl-XL. J Biomed Sci 18(1):48
Olfert ED, Cross BM, McWilliam AA (1993) Guide to the care and use of experimental animals, vol 1. Canadian Council on Animal Care, Ottawa
Yehuda S, Mostofsky DI (1993) Circadian effects of beta-endorphin, melatonin, DSIP, and amphetamine on pentylenetetrazol-induced seizures. Peptides 14(2):203–205
Pavlova MK, Shea SA, Bromfield EB (2004) Day/night patterns of focal seizures. Epilepsy Behav 5(1):44–49
Long MY, Li HH, Weng ZH (2008) Inhibitory effects of transfection of arresten gene on liver metastasis from colorectal cancer in nude mice. Chin J Cancer 27(10):312–315
Mladenovic D, Hrncic D, Vucevic D, Radosavljevic T, Loncar-Stevanovic H, Petrovic J, Susic V, Djuric D, Stanojlovic O (2007) Ethanol suppressed seizures in lindane-treated rats. Electroencephalographic and behavioral studies. J Physiol Pharmacol 58(4):641–656
Silverstone PH, Williams R, McMahon L, Fleming R, Fogarty S (2008) Alcohol significantly lowers the seizure threshold in mice when co-administered with bupropion hydrochloride. Ann Gen Psychiatry 7:11
Olsen RW (2002) GABA. In: Davis KL et al (eds) Neuropsychopharmacology: the fifth generation of Progress Lippincott Williams & Wilkins, Philadelphia, pp 159–168
Kleinrok Z, Dziki M, Janczarek T (1993) The influence of ethanol on pentetrazol-induced seizures and anticonvulsant activity of phenobarbital and valproate against maximal electroshock in mice. Pol J Pharmacol 45(4):361–368
Shiu C, Barbier E, Di Cello F, Choi HJ, Stins M (2007) HIV-1 gp120 as well as alcohol affect blood–brain barrier permeability and stress fiber formation: involvement of reactive oxygen species. Alcohol Clin Exp Res 31(1):130–137
Uchino H, Smith ML, Bengzon J, Lundgren J, Siesjö BK (1996) Characteristics of postischemic seizures in hyperglycemic rats. J Neurol Sci 139(1):21–27
Rigau V, Morin M, Rousset MC, de Bock F, Lebrun A, Coubes P et al (2007) Angiogenesis is associated with blood–brain barrier permeability in temporal lobe epilepsy. Brain 130(Pt 7):1942–1956
Haorah J, Knipe B, Leibhart J, Ghorpade A, Persidsky Y (2005) Alcohol-induced oxidative stress in brain endothelial cells causes blood–brain barrier dysfunction. J Leukoc Biol 78(6):1223–1232
Shang Y, Jiang YX, Ding ZJ, Shen AL, Xu SP, Yuan SY, Yao SL (2010) Valproic acid attenuates the multiple-organ dysfunction in a rat model of septic shock. Chin Med J 123(19):2682–2687
Hrebackova J, Hrabeta J, Eckschlager T (2010) Valproic acid in the complex therapy of malignant tumors. Curr Drug Targets 11(3):361–379
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Golmohammadi, R., Pejhan, A., Azhdari-Zarmehri, H. et al. The role of ethanol on the anticonvulsant effect of valproic acid and cortical microvascular changes after epileptogenesis in mice. Neurol Sci 34, 1125–1131 (2013). https://doi.org/10.1007/s10072-012-1190-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-012-1190-y