Abstract
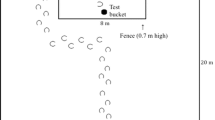
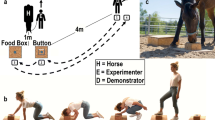
In species that move in cohesive groups, animals generally reach decisions through socially distributed processes, and individual knowledge is expected to influence collective decision making. Pooling of information should not be considered a general rule, however, since conflicts of interest may occur between group members. When resources are limited or highly attractive, higher-ranking individuals can prevent others from accessing food, and subordinates may have an interest in withholding information about its location. We investigated the role individual knowledge may play in recruitment processes in four groups of horses (Equus caballus). Animals were repeatedly released in a food search situation, in which one individual had been informed about the location of a preferred food, while another was a naïve control subject. Horses that were informed about the location of the feeding site were seen to approach the food source more steadily and were followed by a higher number of group members than their uninformed counterparts. Recruitment processes appeared mostly passive. Among the informed subjects, lower-ranking individuals were overall less followed than the higher-ranking ones. Most lower-ranking horses arrived alone at the feeding site. Non-followed informed subjects spent less time in active walk and used direct paths less frequently than followed animals, and they were joined by fewer partners at the attractive food source and spent more time feeding alone. This indicates that the influence of informed individuals on the behavior of other group members was a mixed process. Some horses brought nutritional benefits to their conspecifics by leading them to food supplies, whereas the behavior of others might be functionally deceptive.
Similar content being viewed by others
References
Albers P (2001) Elo-rating as a tool in the sequential estimation of dominance strengths. Anim Behav 61:489–495
Baragli P, Vitale V, Paoletti E, Sighieri C, Reddon AR (2011) Detour behaviour in horses (Equus caballus). J Ethol 29:227–234
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc B 57:289–300
Biro D, Sumpter DJT, Meade J, Guilford T (2006) From compromise to leadership in pigeon homing. Curr Biol 16:2123–2128
Bourjade M, Thierry B, Maumy M, Petit O (2009) Decision-making in Przewalski horses (Equus ferus przewalskii) is driven by the ecological contexts of collective movements. Ethology 115:321–330
Bourjade M, Thierry B, Hausberger M, Petit O (2015) Is leadership a reliable concept in animals? An empirical study in the horse. PLoS ONE 10:e0126344
Briard L, Dorn C, Petit O (2015) Personality and affinities play a key role in the organisation of collective movements in a group of domestic horses. Ethology 121:888–902
Chauvin C, Thierry B (2005) Tonkean macaques orient their food search from olfactory cues conveyed by group mates. Ethology 111:1–10
Conradt L, Roper TJ (2005) Consensus decision-making in animals. Trends Ecol Evol 20:449–456
Coussi-Korbel S (1994) Learning to outwit a competitor in mangabeys (Cercocebus torquatus torquatus). J Comp Psychol 108:164–171
Couzin ID, Krause J (2003) Self-organization and collective behavior in vertebrates. Adv Stud Behav 32:1–75
Couzin ID, Krause J, Franks NR, Levin SA (2005) Effective leadership and decision-making in animal groups on the move. Nature 433:513–516
Ducoing AM, Thierry B (2003) Withholding information in semifree-ranging Tonkean macaques (Macaca tonkeana). J Comp Psychol 117:67–75
Ducoing AM, Thierry B (2004) Following and joining the informed individual in semifree-ranging Tonkean macaques (Macaca tonkeana). J Comp Psychol 118:413–420
Elo AE (1978) The rating of chess players, past and present. Arco Pub, New York
Feh C (2005) Relationships and communication in socially natural horse herds. In: Mills DS, McDonnell SM (eds) The domestic horse: the origins, development and management of its behavior. Cambridge University Press, Cambridge, pp 83–91
Feist JD, McCullough DR (1976) Behavior patterns and communication in feral horses. Z Tierpsychol 41:337–341
Fischoff IR, Sundaresan SR, Cordingley J, Larkin HM, Sellier MJ, Rubenstein DI (2007) Social relationship and reproductive state influence leadership roles in movements of plain zebra Equus burchellii. Anim Behav 73:825–831
Flack A, Pettit B, Freeman R, Guilford T, Biro D (2012) What are leaders made of? The role of individual experience in determining leader–follower relations in homing pigeons. Anim Behav 83:703–709
Fureix C, Bourjade M, Henry C, Sankey C, Hausberger M (2012) Exploring aggression regulation in managed groups of horses Equus caballus. Appl Anim Behav Sci 138:216–228
Gabor G, Gerken M (2014) Shetland ponies (Equus caballus) show quantity discrimination in a matching-to-sample design. Anim Cogn 17:1233–1243
Galef BG, Giraldeau LA (2001) Social influences on foraging in vertebrates: causal mechanisms and adaptive functions. Anim Behav 61:3–15
Galef BG, Stein M (1985) Demonstrator influence on observer diet preference: analyses of critical social interactions and olfactory signals. Anim Learn Behav 13:31–38
Hare B, Call J, Tomasello M (2001) Do chimpanzees know what conspecifics know? Anim Behav 61:139–151
Held S, Mendl M, Devereux C, Byrne RW (2000) Social tactics of pigs in a competitive foraging task: the ‘informed forager’ paradigm. Anim Behav 59:569–576
Krueger K, Flauger B (2011) Olfactory recognition of individual competitors by means of faeces in horse (Equus caballus). Anim Cogn 14:245–257
Krueger K, Flauger K, Farmer K, Hemelrijk CK (2014) Movement initiation in groups of feral horses. Behav Proc 103:91–101
Kummer H (1968) Social organization of hamadryas baboons. Karger, Basel
Lemasson A, Boutin A, Boivin S, Blois-Heulin C, Hausberger M (2009) Horse (Equus caballus) whinnies: a source of social information. Anim Cogn 12:693–704
Menzel EW (1974) A group of young chimpanzees in a one-acre field. In: Schrier AM, Stollnitz F (eds) Behavior of non-human primates, vol 5. Academic Press, New York, pp 83–153
Meunier H, Deneubourg JL, Petit JL (2008) How many for dinner? Recruitment and monitoring by glances in capuchins. Primates 49:26–31
Miklósi A, Soproni K (2006) A comparative analysis of animals’ understanding of the human pointing gesture. Anim Cogn 9:81–93
Neumann C, Duboscq J, Dubuc C, Ginting A, Irwan AM, Agil M, Widdig A, Engelhardt A (2011) Assessing dominance hierarchies: validation and advantages of progressive evaluation with Elo-rating. Anim Behav 82:911–921
Osthaus B, Proops L, Hocking I, Burden F (2013) Spatial cognition and perseveration by horses, donkeys and mules in a simple A-not-B detour task. Anim Cogn 16:301–305
Petit O, Bon R (2010) Decision-making processes: the case of collective movements. Behav Proc 84:635–647
Pillot MH, Gautrais J, Gouello J, Michelena P, Sibbald A, Bon R (2010) Moving together: incidental leaders and naïve followers. Behav Proc 83:235–241
Proops L, McComb K (2010) Attributing attention: the use of human-given cues by domestic horses (Equus caballus). Anim Cogn 13:197–205
Reebs SG (2000) Can a minority of informed leaders determine the foraging movements of a fish shoal? Anim Behav 59:403–409
Schneider G, Krueger K (2012) Third-party interventions keep social partners from exchanging affiliative interactions with others. Anim Behav 83:377–387
Shrader AM, Kerley GIH, Kotler BP, Brown JS (2007) Social information, social feeding, and competition in group-living goats (Capra hircus). Behav Ecol 18:103–107
Sueur C, Petit O (2008) Shared or unshared consensus decision in macaques? Behav Proc 78:84–92
Taylor DB, Price IR, Brown WY, Hinch GN (2011) Effects of Merino flock size, paddocks complexity and time of day on response to trained leaders. Small Rumin Res 97:35–40
Townsend SW, Manser MB (2012) Functionally referential communication in mammals: the past, present and the future. Ethology 118:1–11
VanDierendonck MC, de Vries H, Schilder MBH, Colenbrander B, Þorhallsdóttir AG, Sigurjónsdóttir H (2009) Interventions in social behaviour in a herd of mares and geldings. Appl Anim Behav Sci 116:67e73
Waring GH (2003) Horse behavior. Noyes Publishing, Norwich
Wathan J, McComb K (2014) The eyes and ears are visual indicators of attention in domestic horses. Curr Biol 24:R677–R679
Whiten A, Byrne RW (1988) Tactical deception in primates. Behav Brain Sci 11:233–273
Wilkinson GS (1992) Information transfer at evening bat colonies. Anim Behav 44:501–518
Wolff A, Hausberger M (1996) Learning and memorisation of two different tasks in horses: the effects of age, sex and sire. Appl Anim Behav Sci 46:137–143
Wolter N, Pantel N, Stefanski V, Möstl E, Krueger K (2014) The role of an alpha animal in changing environmental conditions. Physiol Behav 133:236–243
Acknowledgments
We would like to thank Louis Menier and l’Élevage Kergane (Saint-Brieuc-de-Mauron) as well as Martine Clerc and Mireille Dumond at the Écurie Terre d’Illich (Mériadec, Plumergat) for their logistical and practical support. We are grateful to Hélène Thieltges for her valuable assistance in experiments. The study benefited from financial support from the Groupement de Recherche d’Ethologie (GDR 2822). The experiments complied with the current French laws governing animal research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Andrieu, J., Henry, S., Hausberger, M. et al. Informed horses are influential in group movements, but they may avoid leading. Anim Cogn 19, 451–458 (2016). https://doi.org/10.1007/s10071-015-0945-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-015-0945-2