Abstract
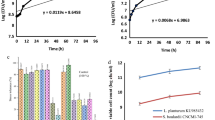
The best starter culture for fermented black soymilk was determined. Black soymilk was fermented using Lactobacillus acidophilus ATCC 4356 (LA), Lactobacillus plantarum P8 (LP), and Streptococcus thermophilus S10 (ST). An ST single culture exhibited higher β-glucosidase activities than LAST (LA:ST=1:1, v/v), LPST (LP:ST=1:1, v/v), and LALPST (LA:LP:ST=1:1:1, v/v) mixed cultures. The pH of fermented black soymilk was significantly (p<0.05) decreased during fermentation due to an increase in lactic acid formed by lactic acid bacteria. The β-glucosidase activity of the ST single culture was significantly (p<0.05) higher than for mixed cultures. Black soymilk fermented with ST alone showed significantly (p<0.05) more bioconversion of the isoflavone glycoside to aglycone, compared with controls. Black soymilk fermented with ST resulted in the best antioxidant effect. The ST single culture was best for production of health functional fermented black soymilk.
Similar content being viewed by others
References
Cederroth CR, Zimmermann C, Nef S. Soy, phytoestrogen, and their impact on reproductive health. Mol. Cell. Endocrinol. 355: 192–200 (2012)
Takahashi R, Ohmori R, Kiyose C, Momitama Y, Ohsuzu F, Kondo K. Antioxidant activities of black and yellow soybeans against low density lipoprotein oxidation. J. Agr. Food Chem. 53: 4578–4582 (2005)
Brouns F. Soya isoflavones: A new and promising ingredient for the health foods sector. Food Res. Int. 35: 187–193 (2002)
Cassidy A. Physiological effects of phyto-estrogens in relation to cancer and other human health risks. P. Nutr. Soc. 55: 399–417 (1996)
Adlercreutz H. Phyto-oestrogens and cancer. Lancet Oncol. 3: 364–373 (2002)
Messina M. Soyfoods and soybean phyto-oestrogens (isoflavones) as possible alternatives to hormone replacement therapy (HRT). Eur. J. Cancer 36: S71–S72 (2000)
Potter SM, Baum JA, Teng H, Stillman RJ, Shay NF. Erdman JW Jr.. Soy protein and isoflavones: Their effects on blood lipids and bone density in postmenopausal women. Am. J. Clin. Nutr. 68: 1375S–1379S (1998)
Scalabrini P, Rossi M, Spettoli P, Matteuzzi D. Characterization of Bifidobacterium strains for use in soymilk fermentation. Int. J. Food Microbiol. 39: 213–219 (1998)
Wang H, Murpy PA. Isoflavone content in commercial soybean foods. J. Agr. Food Chem. 42: 1666–1673 (1994)
Toda T, Sakamoto A, Takayanagi T, Yokotsuka K. Changes in isoflavone compositions of soybean Food during the cooking process. Food Sci. Technol. Res. 6: 314–319 (2000)
Izumi T, Piskula MK, Osawa S, Obata A, Tobe K, Saito M, Kataoka S, Kubota Y, Kikuchi M. Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J. Nutr. 130: 1695–1699 (2000)
Lee BH, Lo YH, Pan TM. Anti-obesity activity of Lactobacillus fermented soymilk products. J. Funct. Foods 5: 905–913 (2013)
Marazza JA, LeBlanc JG, de Giori GS, Garro MS. Soymilk fermented with Lactobacillus rhamnosus CRL981 ameliorates hyperglycemia, lipid profiles and increases antioxidant enzyme activities in diabetic mice. J. Funct. Foods 5: 1848–1853 (2013)
Shon MY, Seo KI, Lee SW, Choi SH, Sung NJ. Biological activities of chungkugjang prepared with black bean and changes in phytoestrogen content during fermentation. Korean J. Food Sci. Technol. 32: 936–941 (2000)
Hwang JS, Kim JY, Sung DI, Yi YS, Kim HB. Fermentation of black-soybean Chungkookjang using Bacillus licheniformis B1. Korean J. Microbiol. 48: 216–219 (2012)
Mital BK, Steinkraus KH, Naylor HB. Growth of lactic acid bacteria in soy milks. J. Food Sci. 39: 1018–1022 (1974)
Wang Z, Bao Y, Zhang Y, Zhang J, Yao G, Wang S, Zhang H. Effect of soymilk fermented with Lactobacillus plantarum P-8 on lipid metabolism and fecal microbiota in experimental hyperlipidemic rats. Food Biophys. 8: 43–49 (2013)
Hong GE, Mandal PK, Lim KW, Lee CH. Fermentation increased isoflavone aglycone contents in black soybean pulp. Asian J. Anim. Vet. Adv. 7: 502–511 (2012)
AOAC. Official method of analysis AOAC Intl. 17th ed. Method 947.05. Association of Official Analytical Communities, Arlington, VA, USA (2000)
AOAC. Official method of analysis AOAC Intl. 17th ed. Method 942.05 Association of Official Analytical Communities, Arlington, VA, USA (2000)
AOAC. Official method of analysis AOAC Intl. 17th ed. Method 920.39. Association of Official Analytical Communities, Arlington, VA, USA (2000)
AOAC. Official method of analysis AOAC Intl. 17th ed. Method 984.13. Association of Official Analytical Communities, Arlington, VA, USA (2000)
Matsuura M, Sasaki J, Murao S. Studies on β-glucosidase from soybeans that hydrolyze daidzin and genistin: Isolation and characterization of an isozyme. Biosci. Biotech. Bioch. 59: 1623–1627 (1995)
Chung WY, Kim SK, Son JY. Isoflavone contents and physiological activities of soybeans fermented with Aspergillus oryzae or Bacillus natto. J. Korean Soc. Food Sci. Nutr. 37: 141–147 (2008)
Singleton VL, Rossi Jr. JA. Colorimetry of total phenolics with phosphomolybic-phosphotungstic acid reagents. Am. J. Enol. Viticult. 16: 144–158 (1965)
Blois MS. Antioxidant determinations by the use of a stable free radical. Nature 181: 1198–1200 (1958)
Oyaizu M. Studies on products of browning reactions: Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. Diet. 44: 307–315 (1986)
Angeles AG, Marth EH. Growth and activity of lactic acid bacteria in soymilk. 1. Growth and acid production. J. Milk Food Technol. 34: 30–36 (1971)
Pinthong R, Macrae R, Dick J. The development of a soya-based yogurt. J. Food Technol. 15: 647–667 (1980)
Chien HL, Huang HY, Chou CC. Transformation of isoflavone phytoestrogens during the fermentation of soymilk with lactic acid bacteria and bifidobacteria. Food Microbiol. 23: 772–778 (2006)
Matsuura M, Obata A. β-Glucosidase from soybeans hydrolyze daidzin and genistin. J. Food Sci. 58: 144–147 (1993)
Tsangalis D, Ashton JF, Mcgill AEJ, Shah NP. Biotransformation of isoflavone by bifidobacteria in fermented soymilk supplemented with d-glucose and l-cystein. J. Food Sci. 68: 623–631 (2003)
Onozawa M, Fukuda K, Ohtani M, Akaza H, Sugimura T, Wakabayashi K. Effects of soybean isoflavones on cell growth and apoptosis of the human prostate cancer cell line LNCaP. Jpn. J. Clin. Oncol. 28: 360–363 (1998)
Xu X, Wang HJ, Murphy PA, Cook L, Hendrich S. Daidzein is a more bioavailable soymilk isoflavone than is genistein in adult woman. J. Nutr. 124: 825–832 (1994)
Park JW, Lee YJ, Yoon S. Total flavonoids and phenolics in fermented soy products and their effects on antioxidant activities determined by different assays. Korean J. Food Culture 22: 353–358 (2007)
Pratt DE, Birac PM. Source of antioxidant activity of soybeans and products. J. Food Sci. 44: 1720–1722 (1979)
Meir S, Kanner J, Akiri B, Hadas SP. Determination and involvement of aqueous reducing compounds in oxidative defense system of various senescing leaves. J. Agr. Food Chem. 43: 1813–1819 (1995)
Pyo YH, Lee TC, Lee YC. Effect of lactic acid fermentation on enrichment of antioxidant properties and bioactive isoflavones in soybean. J. Food Sci. 70: S215–S220 (2005)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, M., Hong, GE., Zhang, H. et al. Production of the isoflavone aglycone and antioxidant activities in black soymilk using fermentation with Streptococcus thermophilus S10. Food Sci Biotechnol 24, 537–544 (2015). https://doi.org/10.1007/s10068-015-0070-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-015-0070-7