Abstract
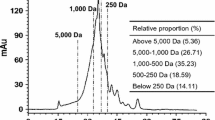
A novel polysaccharide (AGP-32) from the gonad of Haliotis discus hannai Ino was isolated using a protease-assisted process and successive ion-exchange and gel-filtration chromatography. The backbone of AGP-32 was determined using hydrolysis with trifluoroacetic acid. FTIR, NMR, and methylation analysis, and periodate oxidation and Smith degradation analysis revealed that the AGP-32 backbone mainly consisted of (1→6)-linked mannose, (1→3)-linked galactose, and (1→3)-linked glucose in a proportion of 2:3:1. An in vitro cell assay indicated that AGP-32 promoted mice splenic lymphocyte proliferation by 26% at a concentration of 50 μg/mL. AGP-32 had an effect on immune protection and is a candidate for consideration as a functional food.
Similar content being viewed by others
References
Vinh T D, Kirsten B, Peter S. In vitro antiviral activity against herpes simplex virus in the abalone Haliotis laevigata. J. Med. Virol. 92: 627–637 (2011)
Minguez L, Pierre MLH, Costil K, Bureau R, Lebel JM, Serpentini A. Assessment of cytotoxic and immunomodulatory properties of four antidepressants on primary cultures of abalone hemocytes (Haliotis tuberculata). Aquat. Toxicol. 153: 3–11 (2014)
Zhou DY, Zhu BW, Qiao L, Wu HT, Li DM, Yang JF, Murata Y. In vitro antioxidant activity of enzymatic hydrolysates prepared from abalone (Haliotis discus hannai Ino) viscera. Food Bioprod. Process. 90: 148–154 (2012)
Guo LL, Zhu BW, Sun LM, Niu HL, QIN L, Liu JR. Antithrombotic and antifatigue activities of polysaccharide conjugates extracted from abalone gonad. J. Dalian Polytech. Univ. 30: 250–252 (2011)
Wong CK, Leung KN, Fung KP, Choy YM. Immunomodulatory and anti-tumor polysaccharides from medicinal plants. J. Int. Med. Res. 22: 229–312 (1994)
Lowry OH, Rosebrough NJ, Farr AL. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275 (1951)
Rao P, Pattabiraman TN. Reevaluation of the phenol-sulphuric acid reaction for the estimation of hexoses and pentoses. Anal. Biochem. 181: 18–22 (1989)
Filisetti-Cozzi TM, Carpita NC. Measurement of uronic acids without interference from neutral sugars. Anal. Chem. 197: 157–162 (1991)
Therho TT, Hartiala K. Method for determination of the sulfate cont of glycosamino glycans. Anal. Biochem. 41: 471–476 (1971)
Staub AM. Removal of proteins. Sevag method. Vol. 5, pp. 5–6. In: Methods in Carbohydrate Chemistry. Whistler RL (ed). Academic Press, Inc., New York, NY, USA (1965)
Strydom DJJ. Chromatographic separation of 1-phenyl-3-methyl-5-pyrazolone-derivatized neutral, acidic and basic aldoses. J. Chromatogr. A 678: 17–23 (1994)
Yang JF, Zhu BW, Zheng J, Sun LM, Zhou DY, Dong XP, Yu CX. Stimulation of lymphocyte proliferation by oyster glycogen sulfated at C-6 position. Carbohyd. Polym. 94: 301–308 (2013)
Needs PW, Selvendran RR. Avoiding oxidative degradation during sodium hydroxide methyl iodide-mediated carbohydrate methylation in dimethyl sulfoxide. Carbohyd. Res. 245: 1–10 (1993)
Yang JF, Zhou DY, Liang ZY. A new polysaccharide from leaf of Ginkgo biloba L. Fitoterapia 80: 43–47 (2009)
Sun LM, Zhu BW, Li DM, Wang LS, Dong XP, Murata Y, Xing R, Dong Y. Purification and bioactivity of a sulphated polysaccharide conjugate from viscera of abalone Haliotis discus hannai Ino. Food Agr. Immunol. 21: 15–26 (2010)
Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 65: 55–63 (1983)
Liu CH, Lin QX, Gao Y, Ye L, Xing YY, Xi T. Characterization and antitumor activity of a polysaccharide from Strongylocentrotus nudus eggs. Carbohyd. Polym. 67: 313–318 (2007)
Lloyd AG, Dodgson KS, Price RB, Rose FA. Infrared studies of sulphate esters. I. Polysaccharide sulphates. Biochim. Biophys. Acta 46: 108–115 (1961)
Hoffman M, Jia ZH, Peña MJ, Cash M, Harper A, Blackburn AR, Darvill A, York WS. Structural analysis of xyloglucans in the primary cell walls of plants in the subclass Asteridae. Carbohyd. Res. 340: 1826–1840 (2005)
Cao W, Li XQ, Liu L, Yang TH, Li C, Fan HT, Jia M, Lv ZG, Mei QB. Structure of an antitumor polysaccharide from Angelica sinensis (Oliv.) Diels. Carbohyd. Polym. 66: 149–159 (2006)
Uzochukwu S, Balogh E, Loeer RT, Ngoddy PO. Structural analysis by 13C-nuclear magnetic resonance spectroscopy of glucan extracted from natural palm wine. Food Chem. 76: 287–291 (2002)
Seymour FR, Knapp RD, Bishop SH, Jeans A. High temperature enhancement of 13 C-N.M.R. chemical-shifts of unusual dextrans, and correlation with methylation structural analysis. Carbohyd. Res. 68: 123–140 (1979)
Dorries R, Schimpl A, Wecker E. Action of dextran sulfate as a direct and general B cell mitogen. Eur. J. Immunol. 4: 230–233 (1974)
Leung MY, Liu C, Zhu LF, Hui YZ, Yu B, Fung KP. Chemical and biological characterization of a polysaccharide biological response modifier from Aloe vera L. var. chinensis (Haw.) Berg. Glycobiology 14: 501–510 (2004)
Anderson B, Blomgren H. Evidence of thymus-independent humoral antibody production in mice against polyvinylpyrrolidone and E. coli lipopolysaccharide. Cell Immunol. 2: 411–424 (1971)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Jf., Li, Yh., Zhao, J. et al. Isolation, structural characterization, and lymphopoiesis stimulant activity of a polysaccharide from the abalone gonad. Food Sci Biotechnol 24, 23–30 (2015). https://doi.org/10.1007/s10068-015-0004-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-015-0004-4