Abstract
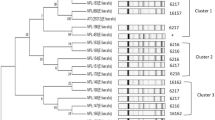
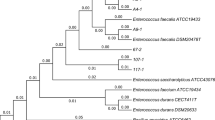
Enterococcus spp. from powdered infant formula and follow-on formulas were identified and characterized for antibiotic susceptibility to confirm the enterococcal safety. Seventy-three Enterococcus from 96 powdered infant formulas and 26 strains from 33 follow-on formulas were isolated. More than 90% of Enterococcus in the foods were E. faecium. E. casseliflavus, and E. faecalis were also isolated. All Enterococcus were sensitive to ampicillin, penicillin, tetracycline, and vancomycin. However, 2 E. casseliflavus showed low resistance to vacomycin by minimum inhibitory concentration 4.0 μg/mL. Multiplex PCR indicated no existence of highly hazardous vancomycinresistant vanA and vanB genes. The isolates also showed the broad susceptibilities to erythromycin, ripampin, and streptomycin. Major strains of about 60% were intermediately and highly resistant to erythromycin and streptomycin, respectively. However, Enterococcus resistances to those antibiotics were not high and similar to those of Enterococcus from the other evaluated foods. Therefore, it appeared that powdered infant formula and follow-on formula might be safe to Enterococcus with regard to vancomycin and the antibiotics.
Similar content being viewed by others
References
Nazarowec-White M. Enterobacter sakazakii: A review. Int. J. Food Microbiol. 34: 103–113 (1997)
Jung MK, Park JH. Prevalence and thermal stability of Enterobacter sakazakii from unprocessed ready-to-eat agricultural products and powdered infant formulas. Food Sci. Biotechnol. 15: 152–157 (2006)
Kandhai MC, Reij MW, van Puyveld K, Guillaume-Gentil O, Beumer RR, van Schothorst MA. Occurrence of Enterobacter sakazakii in food production environments and households. Lancet 363: 39–40 (2004)
Foulquié Moreno MR, Sarantinopoulos P, de Vuyst L. The role and application of enterococci in food and health. Int. J. Food Microbiol. 10: 1–24 (2006)
Fackalm RR, Collins MD. Identification of Enterococcus species isolated from human infections by a conventional test scheme. J. Clin. Microbiol. 27: 731–734 (1989)
Devriese LA, Pot B, Collins MD. Phenotypic identification of the genus Enterococcus and differentiation of phylogenetically distinct enterococcal species and species group. J. Appl. Bacteriol. 75: 399–408 (1993)
Moellering RC. Enterococcus species, Streptococcus bovis, and Leuconostoc. pp. 1826–1835. In: A Principles and Practice of Infectious Disease. 4th ed. Mandel GL, Bennett JE, Dolin R (eds). Churchill Living Stone, New York, NY, USA (1995)
Jett BD, Huycke MM, Gilnore MS. Virulence of enterococci. Clin. Microbiol. Rev. 7: 462–478 (1994)
Johnson AP, Uttley AHC, Woodford N, Geroge RC. Resistance to vancomycin and teicoplanin: An emerging clinical problem. Clin. Microbiol. Rev. 3: 280–291 (1990)
Schleifer KH, Kilpper-Balz R. Molecular and chemotaxonomic approach to the classification of streptococci, enterococci, and lactococci. Syst. Appl. Microbiol. 10: 1–18 (1987)
Anonymous. EEC council directive of 16 June 1992 on milk hygiene (92/46EEC). Off. J. Eur. Communities L268/1, 14.9. 1992 (1992)
Kang TM, Cho SK, Park JH. Antibiotic resistances of Enterococcus isolated from salad and sprout. Korean J. Microbiol. Biotechnol. 36: 142–148 (2008)
Kim SH, Kim JS, Park JH. Antibiotic resistance of Enterococcus isolated from the processed grain foods, saesik and sunsik. Food Sci. Biotechnol. 16: 470–476 (2007)
Sahm DF, Free L, Smith C, Eveland M, Mundy LM. Rapid characterization schemes for surveillance isolates of vancomycinresistant enterococci. J. Clin. Microbiol. 35: 2026–2030 (1997)
Danbing KE, Picard FJ, Martineau F, Menard C, Roy PH, Ouellette M, Bergeron MG. Development of a PCR assay for rapid detection of enterococci. J. Clin. Microbiol. 11: 3497–3503 (1999)
Zarazaga M, Saenz Y, Portillo A, Tenorio C, Ruiz-Larrea F, Campo RD, Baquero F, Torres C. In vitro activities of ketolide HMR 3647, mactolides, and other antibiotics against Lactobacillus, Leuconostoc, and Pediococcus isolates. Antimicrob. Agent Ch. 43: 3039–3041 (1999)
Clinical Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Test; Approved standards Method M2-A9, M7-A7. 8th ed. CLSI Document, Wayne, PA, USA (2008)
Dukta-Malen S, Evers S, Courvalin P. Detection of glycopeptide resistance genotypes and identification to the species level of clinically relevant enterococci by PCR. J. Clin. Micribiol. 33: 24–27 (1995)
Codex Alimentarius Commission. CAC/RCP-21, Alinorm 79: 38. Recommended International Code of Hygienic Practice for Foods for Infants and Children. Rome, Italy (1979)
Iversen C, Forsythe SJ. Risk profile of Enterobacter sakazakii, an emergent pathogen associated with infant milk formula. Trends Food Sci. Tech. 14: 443–454 (2003)
Korea Food & Drug Administration. Korea Food Code. http://www.kfda.go.kr. Accessed on Jan. 30, 2012.
Animal, Plant and Fisheries Quarantine and Inspection Agency. Korea Livestocks Code. http://www.qia.go.kr. Accessed Jan. 30, 2012.
Mundt OJ. Enterococci. Vol. 2, pp. 1063–1065. In: Bergey’s Manual of Systematic Bacteriology. Sneath PHA, Mair NS, Sharpe ME, Holt JG (eds). Williams and Wilkins, Baltimore, MD, USA (1986)
Klein G, Pack A, Reuter G. Antibiotic resistance patterns of enterococci and occurrence of vancomycin-resistant enterococci in raw minced beef and pork in Germany. Appl. Environ. Microb. 64: 1825–1830 (1998)
Stobbering E, van den Bogaard A, London N, Driessen C, Top J, Willens R. Enterococci with glycopeptide resistance in turkeys, turkey farmers, turkey slaughterers, and urban residents in the south of the Netherlands: Evidence for transmission of vancomycin resistance from animals to humans? Antimicrob. Agents Ch. 43: 2215–2221 (1999)
Devriese LA, Pot B, van Damme L, Kersters K, Haesebrouck F. Identification of Enterococcus species isolated from foods of animal origin. Int. Food Microbiol. 26: 187–197 (1995)
Cho YS, Lee HS, Kim JM, Ryu PD, Park YH, Yoo HS, Lee MH. Comparison of antimicrobial susceptibility of vancomycin resistant enterococci from animal and human. Korean J. Vet. Publ. Hlth. 27: 17–29 (2003)
Randazzo CL, Restuccia C, Romano AD, Caggia C. Lactobacillus casei, dominant species in naturally fermented Sicilian green olives. Int. J. Food Microbiol. 90: 9–14 (2004)
Klare I, Heier H, Claus H, Reissbrodt R, Witte W. vanA-mediated high-level glycopeptides resistance in Enterococcus faecium from animal husbandry. FEMS Microbiol. Lett. 125: 165–172 (1995)
Pavia M, Nobile CGA, Salpietro L, Angellillo IF. Vancomycin resustance and antibiotic susceptibility of enterococci in raw meat. J. Food Protect. 7: 912–915 (2000)
Chadwick PR, Woodford N, Kaczmarski EB, Gray S, Barrell RA, Oppenheim BA. Glycopeptide-resistant enterococci isolated from uncooked meat. J. Antimicrob. Ch. 38: 908–909 (1996)
Seo KS, Song DJ, Gwyther MM, Park YH. Development of multiplex PCR for detection of vancomycin resistant enterococci (VRE) and epidemiological application in Korea. Korean J. Vet. Res. 39: 343–352 (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, TM., Park, JH. Isolation of Enterococcus from powdered infant and follow-on formulas, and their antibiotic susceptibilites. Food Sci Biotechnol 21, 1113–1118 (2012). https://doi.org/10.1007/s10068-012-0145-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-012-0145-7