Abstract
Objective
Systemic lupus erythematosus (SLE) is a heterogeneous disease with multiple clinical manifestations, which causes a significant deterioration in the quality of life (QoL). The Systemic Lupus Erythematosus Quality of Life Questionnaire (L-QoL) is a lupus-specific measure used to determine the burden of the disease and it applies the need-based model of QoL. Our aim was to produce the first successfully validated foreign language version of the questionnaire.
Methods
The development of the Bulgarian version involved three stages: translation, field testing and psychometric evaluation. Translation was conducted by an expert linguist working with a developer of the original L-QoL, followed by interviews with monolingual lay individuals. Face and content validity of the translation were assessed by cognitive debriefing interviews with Bulgarian SLE patients. Finally, the L-QoL was validated by administering the questionnaire to a random sample of SLE patients on two occasions, 2 weeks apart to evaluate its reliability and validity.
Results
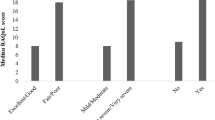
In the validation survey, the new Bulgarian version demonstrated high internal consistency (the Cronbach’s alpha coefficient was 0.92), and test–retest reliability (0.97). Additionally, scores on the L-QoL were correlated with those on the SF-36 sections to determine convergent validity and the strongest correlation was observed between L-QoL scores and the social functioning section of the SF-36. Known group validity was established by testing the ability of the Bulgarian L-QoL to distinguish between subgroups of patients from the study pool.
Conclusions
The demonstrated excellent psychometric properties ensure that the Bulgarian L-QoL accurately captures the impact of SLE on the quality of life.
Key points • The Bulgarian version of the L-QoL is a valid and reliable measure of QoL in lupus patients. • The Bulgarian version of the L-QoL can be used as an outcome measure in research, clinical trials and routine clinical practice. |


Similar content being viewed by others
References
Kaul A, Gordon C, Crow MK, Touma Z, Urowitz MB, van Vollenhoven R, Ruiz-Irastorza G, Hughes G (2016) Systemic lupus erythematosus. Nat Rev Dis Primers 2:16039. https://doi.org/10.1038/nrdp.2016.39
Houssiau FA, Vasconcelos C, D'Cruz D, Sebastiani GD, de Ramon GE, Danieli MG, Abramovicz D, Blockmans D, Cauli A, Direskeneli H, Galeazzi M, Gül A, Levy Y, Petera P, Popovic R, Petrovic R, Sinico RA, Cattaneo R, Font J et al (2010) The 10-year follow-up data of the Euro-Lupus Nephritis Trial comparing low-dose and high-dose intravenous cyclophosphamide. Ann Rheum Dis 69(1):61–64. https://doi.org/10.1136/ard.2008.102533
van Vollenhoven RF, Bertsias G, Doria A et al (2021) 2021 DORIS definition of remission in SLE: final recommendations from an international task force Lupus. Sci Med 8:e000538. https://doi.org/10.1136/lupus-2021-000538
Franklyn K, Lau CS, Navarra SV, Louthrenoo W, Lateef A, Hamijoyo L, Wahono CS, Chen SL, Jin O, Morton S, Hoi A, Huq M, Nikpour M, Morand EF (2016) Definition and initial validation of a Lupus Low Disease Activity State (LLDAS). Ann Rheum Dis 75(9):1615–1621. https://doi.org/10.1136/annrheumdis-2015-207726
Mosca M, Tani C, Aringer M, Bombardieri S, Boumpas D, Brey R, Cervera R, Doria A, Jayne D, Khamashta MA, Kuhn A, Gordon C, Petri M, Rekvig OP, Schneider M, Sherer Y, Shoenfeld Y, Smolen JS, Talarico R et al (2010) European League Against Rheumatism recommendations for monitoring patients with systemic lupus erythematosus in clinical practice and in observational studies. Ann Rheum Dis 69(7):1269–1274. https://doi.org/10.1136/ard.2009.117200
Strand V, Gladman D, Isenberg D, Petri M, Smolen J, Tugwell P (2000) Endpoints: consensus recommendations from OMERACT IV. Lupus 9(5):322–327. https://doi.org/10.1191/096120300678828424
Parra Sánchez AR, Voskuyl AE, van Vollenhoven RF (2022) Treat-to-target in systemic lupus erythematosus: advancing towards its implementation. Nat Rev Rheumatol 18(3):146–157. https://doi.org/10.1038/s41584-021-00739-3
Pisetsky DS, Clowse MEB, Criscione-Schreiber LG, Rogers JL (2019) A novel system to categorize the symptoms of systemic lupus erythematosus arthritis. Care Res 71(6):735–741. https://doi.org/10.1002/acr.23794
Ware JE Jr (2000) SF-36 health survey update. Spine 25:3130–3139. https://doi.org/10.1097/00007632-200012150-00008
Gordon C, Amissah-Arthur MB, Gayed M, Brown S, Bruce IN, D'Cruz D, Empson B, Griffiths B, Jayne D, Khamashta M, Lightstone L, Norton P, Norton Y, Schreiber K, Isenberg D, British Society for Rheumatology Standards, Audit and Guidelines Working Group (2018) The British Society for Rheumatology guideline for the management of systemic lupus erythematosus in adults. Rheumatology 57(1):e1–e45. https://doi.org/10.1093/rheumatology/kex286
McElhone K, Abbott J, Shelmerdine J, Bruce IN, Ahmad Y, Gordon C, Peers K, Isenberg D, Ferenkeh-Koroma A, Griffiths B, Akil M, Maddison P, Teh LS (2007) Development and validation of a disease-specific health-related quality of life measure, the LupusQol, for adults with systemic lupus erythematosus. Arthritis Rheum 57(6):972–979. https://doi.org/10.1002/art.22881
Leong KP, Kong KO, Thong BY, Koh ET, Lian TY, Teh CL, Cheng YK, Chng HH, Badsha H, Law WG, Lau TC, Chew LC, Ho HJ, Pong LY, Hoi LS, Sangeetha N, Chan SP, Howe HS (2005) Development and preliminary validation of a systemic lupus erythematosus-specific quality-of-life instrument (SLEQOL). Rheumatology 44(10):1267–1276. https://doi.org/10.1093/rheumatology/keh605
Jolly M, Pickard AS, Block JA, Kumar RB, Mikolaitis RA, Wilke CT, Rodby RA, Fogg L, Sequeira W, Utset TO, Cash TF, Moldovan I, Katsaros E, Nicassio P, Ishimori ML, Kosinsky M, Merrill JT, Weisman MH, Wallace DJ (2012) Disease-specific patient reported outcome tools for systemic lupus erythematosus. Semin Arthritis Rheum 42(1):56–65. https://doi.org/10.1016/j.semarthrit.2011.12.005
Doward LC, McKenna SP, Whalley D, Tennant A, Griffiths B, Emery P, Veale DJ (2009) The development of the L-QoL: a quality-of-life instrument specific to systemic lupus erythematosus. Ann Rheum Dis 68(2):196–200. https://doi.org/10.1136/ard.2007.086009
Hunt SM, McKenna SP (1992) The QLDS: a scale for the measurement of quality of life in depression. Health Policy 22:307–319. https://doi.org/10.1016/0168-8510(92)90004-u
Whalley D, McKenna SP, de Jong Z, van der Heijde D (1997) Quality of life in rheumatoid arthritis. Br J Rheumatol 1997 36(8):884–888. https://doi.org/10.1093/rheumatology/36.8.884
de Jong Z, van der Heijde D, McKenna SP, Whalley D (1997) The reliability and construct validity of the RAQoL: a rheumatoid arthritis-specific quality of life instrument. Br J Rheumatol 36(8):878–883. https://doi.org/10.1093/rheumatology/36.8.878
McKenna SP, Doward LC, Whalley D, Tennant A, Emery P, Veale DJ (2004) Development of the PsAQoL: a quality of life instrument specific to psoriatic arthritis. Ann Rheum Dis 63(2):162–169. https://doi.org/10.1136/ard.2003.006296
Doward LC, Spoorenberg A, Cook SA, Whalley D, Helliwell PS, Kay LJ, McKenna SP, Tennant A, van der Heijde D, Chamberlain MA (2003) Development of the ASQoL: a quality of life instrument specific to ankylosing spondylitis. Ann Rheum Dis 62(1):20–26. https://doi.org/10.1136/ard.62.1.20
Keenan AM, McKenna SP, Doward LC, Conaghan PG, Emery P, Tennant A (2008) Development and validation of a needs-based quality of life instrument for osteoarthritis. Arthritis Rheum 59(6):841–848. https://doi.org/10.1002/art.23714
Streiner DL, Norman GR (1995) Health measurement scales: a practical guide to their development and use, 2nd edn. Oxford University Press, New York
Weiner EA, Stewart BJ (1984) Assessing individuals Little Brown, Boston
Hunt SM, Alonso J, Bucquet D, Niero M, Wiklund I, McKenna S (1991) Cross-cultural adaptation of health measures. Eur Group Health Manag Qual Life Assess Health Policy 19(1):33–44. https://doi.org/10.1016/0168-8510(91)90072-6
Hunt SM, McKenna SP, McEwen J, Williams J, Papp E (1981) The Nottingham Health profile: subjective health status and medical consultations. Soc Sci Med A 15(3 Pt 1):221–229. https://doi.org/10.1016/0271-7123(81)90005-5
Mahieu M, Yount S, Ramsey-Goldman R (2016) Patient-reported outcomes in systemic lupus erythematosus. Rheum Dis Clin North Am 42:253–263. https://doi.org/10.1016/j.rdc.2016.01.001
Olesinska M, Saletra A (2018) Quality of life in systemic lupus erythematosus and its measurement. Reumatologia. 56:45–54. https://doi.org/10.5114/reum.2018.74750
Dua AB, Touma Z, Toloza S, Jolly M (2013) Top 10 recent developments in health-related quality of life in patients with systemic lupus erythematosus. Curr Rheumatol Rep 15(12):380. https://doi.org/10.1007/s11926-013-0380-9
Acknowledgements
The authors would like to thank all participants in the bilingual and lay translation panels and all the patients for their participation. The authors would also like to thank Alice Heaney and Evdokiya Sotirova for their contributions to the study.
Funding
None.
Author information
Authors and Affiliations
Contributions
Study concept and design: SPM and AC. Data collection: DTP and SVM. Statistical analysis and data interpretation: SPM and AC. DTP prepared the first version of the manuscript and all authors contributed to critical revision of the manuscript. All authors read and approved the final version.
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pencheva, D.T., Coffey, A., McKenna, S.P. et al. Adaptation and validation of the Bulgarian version of the Systemic Lupus Erythematosus Quality of Life Questionnaire (L-QoL). Clin Rheumatol 42, 1811–1817 (2023). https://doi.org/10.1007/s10067-023-06523-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-023-06523-w