Abstract
Objective
We aimed to characterize the alterations in the immune phenotypes and explore the potential relevance to pathogenesis in IgG4-RD.
Methods
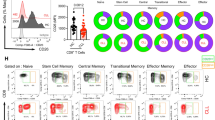
Forty-two IgG4-RD patients and thirty-eight healthy controls were recruited in this study. Peripheral immunocompetent cells including T cells, CD4 + T cells, CD8 + T cells, B cells, NK cells CD4 + CD45RA + T cells (naïve T cells), CD4 + CD25 − / + Foxp3 − T cells (Teff), CD4 + CD25hiCD127lowCD161 + T cells (CD161 + Treg), CD4 + CD25hiFoxp3 + T cells (Foxp3 + Treg), CD4 + CD4RA-CXCR5 + PD1 + CCR7low T cells (pTfh), T helper (Th) 1, Th2, and Th17 before and after treatment were immunophenotyped by flow cytometry.
Results
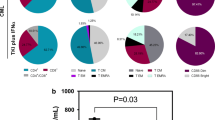
Compared with healthy controls, IgG4-RD patients showed higher proportions of NK (20.1% vs 13.6%, p < 0.01), Th1 (CD4 + IFN-γ + : 17.9% vs 14.2%, p = 0.061; TNF-α: 43.7% vs 36.7%, p < 0.05), Th2 (CD4 + IL-4 + : 2.4% vs 1.3%, p < 0.0001), CD161 + Treg (14.9% vs 11.6%, p < 0.01), pTfh (3.2% vs 2.4%, p < 0.05), and Foxp3 + Treg (8.3% vs 7.0%, p < 0.01) and lower proportions of B lymphocytes (8.4% vs 13.1%, p < 0.001), Teff (91.6% vs 92.6%, p < 0.01), and naïve Th cells (19.9% vs 32.1%, p < 0.01) before treatment. Foxp3 + Treg percentage decreased significantly after treatment (8.6% vs 6.9%, p < 0.05). Both serum C3 (r = − 0.6374, p < 0.01) and C4 (r = − 0.6174, p < 0.01) levels were in negative correlation with CD161 + Treg. The eosinophil percentage was positively correlated with Foxp3 + Treg (r = 0.5435, p < 0.05). Serum IgE level was positively correlated with Th2 (r = 0.5545, p < 0.05). There was a positive correlation between CD161 + Treg and pTfh (r = 0.4974, p < 0.05) while a negative correlation between Th2 and B cells (r = − 0.4925, p < 0.05).
Conclusion
Immune phenotypes were altered in IgG4-RD. Treg/Teff balance was shifted toward Treg in IgG4-RD. CD161 + Treg was likely to be involved in the pathogenesis of IgG4-RD.
Key Points •Immune phenotypes were altered in B cells, T cells, and NK cells in IgG4-RD. •Treg/Teff balance was shifted toward Treg in IgG4-RD. •CD161+ Treg maybe play a proinflammatory role in IgG4-RD |





Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Liu Y, Zhu L, Wang Z, Zeng Q, Yang F, Gao J et al (2020) Clinical features of IgG4-related retroperitoneal fibrosis among 407 patients with IgG4-related disease: a retrospective study. Rheumatology (Oxford, England). https://doi.org/10.1093/rheumatology/keaa411
Wallace ZS, Deshpande V, Mattoo H, Mahajan VS, Kulikova M, Pillai S et al (2015) IgG4-related disease: clinical and laboratory features in one hundred twenty-five patients. Arthritis Rheumatol (Hoboken, NJ) 67(9):2466–2475. https://doi.org/10.1002/art.39205
Inoue D, Yoshida K, Yoneda N, Ozaki K, Matsubara T, Nagai K et al (2015) IgG4-related disease: dataset of 235 consecutive patients. Medicine 94(15):e680. https://doi.org/10.1097/md.0000000000000680
Laidlaw BJ, Lu Y, Amezquita RA, Weinstein JS, Vander Heiden JA, Gupta NT et al (2017) Interleukin-10 from CD4(+) follicular regulatory T cells promotes the germinal center response. Sci Immunol 2(16):eaan4767. https://doi.org/10.1126/sciimmunol.aan4767
Lanzillotta M, Della-Torre E, Milani R, Bozzolo E, Bozzalla-Cassione E, Rovati L et al (2018) Increase of circulating memory B cells after glucocorticoid-induced remission identifies patients at risk of IgG4-related disease relapse. Arthritis Res Ther 20(1):222. https://doi.org/10.1186/s13075-018-1718-5
Moriyama M, Nakamura S (2017) Th1/Th2 immune balance and other T helper subsets in IgG4-related disease. Curr Top Microbiol Immunol 401:75–83. https://doi.org/10.1007/82_2016_40
Palomares O, Yaman G, Azkur AK, Akkoc T, Akdis M, Akdis CA (2010) Role of Treg in immune regulation of allergic diseases. Eur J Immunol 40(5):1232–1240. https://doi.org/10.1002/eji.200940045
Noval Rivas M, Chatila TA (2016) Regulatory T cells in allergic diseases. J Allergy Clin Immunol 138(3):639–652. https://doi.org/10.1016/j.jaci.2016.06.003
Uchida K, Okazaki K (2017) Roles of regulatory T and B cells in IgG4-related disease. Curr Top Microbiol Immunol 401:93–114. https://doi.org/10.1007/82_2016_41
Afzali B, Mitchell PJ, Edozie FC, Povoleri GAM, Dowson SE, Demandt L et al (2013) CD161 expression characterizes a subpopulation of human regulatory T cells that produces IL-17 in a STAT3-dependent manner. Eur J Immunol 43(8):2043–2054. https://doi.org/10.1002/eji.201243296
Duurland CL, Brown CC, O’Shaughnessy RF, Wedderburn LR (2017) CD161(+) Tconv and CD161(+) Treg share a transcriptional and functional phenotype despite limited overlap in TCRβ repertoire. Front Immunol 8:103. https://doi.org/10.3389/fimmu.2017.00103
Pesenacker AM, Bending D, Ursu S, Wu Q, Nistala K, Wedderburn LR (2013) CD161 defines the subset of FoxP3+ T cells capable of producing proinflammatory cytokines. Blood 121(14):2647–2658. https://doi.org/10.1182/blood-2012-08-443473
Gernez Y, Tirouvanziam R, Nguyen KD, Herzenberg LA, Krensky AM, Nadeau KC (2007) Altered phosphorylated signal transducer and activator of transcription profile of CD4+CD161+ T cells in asthma: modulation by allergic status and oral corticosteroids. J Allergy Clin Immunol 120(6):1441–1448. https://doi.org/10.1016/j.jaci.2007.08.012
Cosmi L, Cimaz R, Maggi L, Santarlasci V, Capone M, Borriello F et al (2011) Evidence of the transient nature of the Th17 phenotype of CD4+CD161+ T cells in the synovial fluid of patients with juvenile idiopathic arthritis. Arthritis Rheum 63(8):2504–2515. https://doi.org/10.1002/art.30332
Miao J, Geng J, Zhang K, Li X, Li Q, Li C et al (2014) Frequencies of circulating IL-17-producing CD4+CD161+ T cells and CD4+CD161+ T cells correlate with disease activity in rheumatoid arthritis. Mod Rheumatol 24(2):265–570. https://doi.org/10.3109/14397595.2013.854070
Umehara H, Okazaki K, Kawa S, Takahashi H, Goto H, Matsui S et al (2021) The 2020 revised comprehensive diagnostic (RCD) criteria for IgG4-RD. Mod Rheumatol 31(3):529–533. https://doi.org/10.1080/14397595.2020.1859710
Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, Saeki T et al (2012) Comprehensive diagnostic criteria for IgG4-related disease (IgG4-RD), 2011. Mod Rheumatol 22(1):21–30. https://doi.org/10.1007/s10165-011-0571-z
Kamekura R, Takahashi H, Ichimiya S (2019) New insights into IgG4-related disease: emerging new CD4+ T-cell subsets. Curr Opin Rheumatol 31(1):9–15. https://doi.org/10.1097/bor.0000000000000558
Li J, Liu Z, Zhang P, Lin W, Lu H, Peng Y et al (2021) Peripheral B-cell immunophenotyping identifies heterogeneity in IgG4-related disease. Front Immunol 12:747076. https://doi.org/10.3389/fimmu.2021.747076
Grados A, Ebbo M, Piperoglou C, Groh M, Regent A, Samson M et al (2017) T cell polarization toward TH2/TFH2 and TH17/TFH17 in patients with IgG4-related disease. Front Immunol 8:235. https://doi.org/10.3389/fimmu.2017.00235
Okazaki K, Uchida K, Ohana M, Nakase H, Uose S, Inai M et al (2000) Autoimmune-related pancreatitis is associated with autoantibodies and a Th1/Th2-type cellular immune response. Gastroenterology 118(3):573–581. https://doi.org/10.1016/s0016-5085(00)70264-2
Ohta N, Makihara S, Okano M, Kurakami K, Ishida A, Furukawa T et al (2012) Roles of IL-17, Th1, and Tc1 cells in patients with IgG4-related sclerosing sialadenitis. Laryngoscope 122(10):2169–2174. https://doi.org/10.1002/lary.23429
Takeuchi M, Sato Y, Ohno K, Tanaka S, Takata K, Gion Y et al (2014) T helper 2 and regulatory T-cell cytokine production by mast cells a key factor in the pathogenesis of IgG4-related disease. Modern Pathol 27(8):1126–36
Heeringa JJ, Karim AF, van Laar JAM, Verdijk RM, Paridaens D, van Hagen PM et al (2018) Expansion of blood IgG(4)(+) B, T(H)2, and regulatory T cells in patients with IgG(4)-related disease. J Allergy Clin Immunol 141(5):1831–43.e10. https://doi.org/10.1016/j.jaci.2017.07.024
Della Torre E, Mattoo H, Mahajan VS, Carruthers M, Pillai S, Stone JH (2014) Prevalence of atopy, eosinophilia, and IgE elevation in IgG4-related disease. Allergy 69(2):269–272. https://doi.org/10.1111/all.12320
Buckner JH (2010) Mechanisms of impaired regulation by CD4(+)CD25(+)FOXP3(+) regulatory T cells in human autoimmune diseases. Nat Rev Immunol 10(12):849–859. https://doi.org/10.1038/nri2889
Huang Z, Yang B, Shi Y, Cai B, Li Y, Feng W et al (2012) Anti-TNF-α therapy improves Treg and suppresses Teff in patients with rheumatoid arthritis. Cell Immunol 279(1):25–29. https://doi.org/10.1016/j.cellimm.2012.09.001
Machhi J, Yeapuri P, Lu Y, Foster E, Chikhale R, Herskovitz J et al (2021) CD4+ effector T cells accelerate Alzheimer’s disease in mice. J Neuroinflammation 18(1):272. https://doi.org/10.1186/s12974-021-02308-7
Chen Y, Li Z, Li H, Su W, Xie Y, Pan Y et al (2020) Apremilast regulates the Teff/Treg balance to ameliorate uveitis via PI3K/AKT/FoxO1 signaling pathway. Front Immunol 11:581673. https://doi.org/10.3389/fimmu.2020.581673
Culver EL, Sadler R, Bateman AC, Makuch M, Cargill T, Ferry B et al (2017) Increases in IgE, eosinophils, and mast cells can be used in diagnosis and to predict relapse of IgG4-related disease. Clin Gastroenterol Hepatol 15(9):1444–52.e6. https://doi.org/10.1016/j.cgh.2017.02.007
Takeuchi M, Ohno K, Takata K, Gion Y, Tachibana T, Orita Y et al (2015) Interleukin 13-positive mast cells are increased in immunoglobulin G4-related sialadenitis. Sci Rep 5:7696. https://doi.org/10.1038/srep07696
Mellor AL, Lemos H, Huang L (2017) Indoleamine 2,3-dioxygenase and tolerance: where are we now? Front Immunol 8:1360. https://doi.org/10.3389/fimmu.2017.01360
Funding
The work was supported by Capital’s Funds for Health Improvement and Research (No. 2022–2-2026) and National Natural Science Foundation of China (U1903210).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bian, W., Li, Y., Sun, F. et al. Immune phenotype changes in IgG4-related disease: CD161 + Treg and Foxp3 + Treg. Clin Rheumatol 42, 1113–1124 (2023). https://doi.org/10.1007/s10067-022-06445-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06445-z