Abstract
Objectives
Our aim was to evaluate the association between salivary gland scintigraphy and the clinical parameters, including histological characteristics of salivary glands, in patients with primary Sjogren’s syndrome (pSS).
Methods
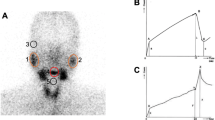
Forty-one pSS patients were included in the study. The patients who had received salivary gland scintigraphy and minor salivary gland biopsy were retrospectively analyzed. Salivary gland scintigraphy was interpreted via semi-quantitative methods obtained by calculating the peak uptake and washout of each gland using regions of interest. All specimens were examined by pathologists for focus scores and leukocyte common antigen (LCA) to determine the degree of inflammatory infiltration.
Results
The mean age of pSS patients was 46.4 years, 82.9% were female, and the mean duration of symptoms was 2.5 years. The focus score was negatively correlated to the mean peak uptake (r = ‒0.396; p = 0.019), mean uptake (r = ‒0.388; p = 0.021), and mean percentage washout (r = ‒0.391; p = 0.02). In addition, the focus score and number of LCA positive cells per mm2 were correlated with the clinical parameters including erythrocyte sedimentation rate, globulin, rheumatoid factor, unstimulated whole saliva, and stimulated whole saliva flow. The number of LCA positive cells per mm2 was negatively correlated to leukocytes and hemoglobin.
Conclusion
Although the diagnostic role of salivary gland biopsy is widely accepted and features in the classification criteria of Sjogren’s syndrome, salivary gland scintigraphy may be an acceptable alternative method especially if a non-invasive test is required.
Key Points • Salivary gland biopsy in patients with primary Sjogren’s syndrome (pSS) showed a significant correlation with several markers of both the parotid and submandibular glands in salivary gland scintigraphy, and with clinical features such as cytopenia, and lower unstimulated whole saliva and stimulated whole saliva flow rates. • Salivary gland scintigraphy might be a good alternative to minor salivary gland biopsy, and the two methods could be complementary to the clinical features in the diagnosis of pSS. |

Similar content being viewed by others
Data availability
All available data are reported in the manuscript and supplementary file.
References
Wang J, Zhou L, Liu B (2020) Update on disease pathogenesis, diagnosis, and management of primary Sjögren’s syndrome. Int J Rheum Dis 23:723–727
Psianou K, Panagoulias I, Papanastasiou AD et al (2018) Clinical and immunological parameters of Sjögren’s syndrome. Autoimmun Rev 17:1053–1064
Cafaro G, Croia C, Argyropoulou OD et al (2019) One year in review 2019: Sjögren’s syndrome. Clin Exp Rheumatol 37(Suppl 118):3–15
Negrini S, Emmi G, Greco M et al (2022) Sjögren’s syndrome: a systemic autoimmune disease. Clin Exp Med 22:9–25
Bloch KJ, Buchanan WW, Wohl MJ, Bunim JJ (1965) Sjoegren’s syndrome. A clinical, pathological, and serological study of sixty-two cases. Medicine (Baltimore) 44:187–231
Daniels TE, Silverman S Jr, Michalski JP, Greenspan JS, Sylvester RA, Talal N (1975) The oral component of Sjögren’s syndrome. Oral Surg Oral Med Oral Pathol 39:875–885
Manthorpe R, Frost-Larsen K, Isager H, Prause JU (1981) Sjögren’s syndrome. A review with emphasis on immunological features. Allergy 36:139–153
Fox RI, Robinson CA, Curd JG, Kozin F, Howell FV (1986) Sjögren’s syndrome Proposed criteria for classification. Arthritis Rheum 29:577–585
Skopouli FN, Drosos AA, Papaioannou T, Moutsopoulos HM (1986) Preliminary diagnostic criteria for Sjögren’s syndrome. Scand J Rheumatol Suppl 61:22–25
Vitali C, Bombardieri S, Moutsopoulos HM et al (1993) Preliminary criteria for the classification of Sjögren’s syndrome. Results of a prospective concerted action supported by the European Community. Arthritis Rheum 36:340–347
Fujibayashi T, Sugai S, Miyasaka N, Hayashi Y, Tsubota K (2004) Revised Japanese criteria for Sjögren’s syndrome (1999): availability and validity. Mod Rheumatol 14:425–434
Vitali C, Bombardieri S, Jonsson R et al (2002) Classification criteria for Sjögren’s syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann Rheum Dis 61:554–558
Shiboski SC, Shiboski CH, Criswell L et al (2012) American College of Rheumatology classification criteria for Sjögren’s syndrome: a data-driven, expert consensus approach in the Sjögren’s International Collaborative Clinical Alliance cohort. Arthritis Care Res (Hoboken) 64:475–487
Cornec D, Saraux A, Cochener B et al (2014) Level of agreement between 2002 American-European Consensus Group and 2012 American College of Rheumatology classification criteria for Sjögren’s syndrome and reasons for discrepancies. Arthritis Res Ther 16:R74
Shiboski CH, Shiboski SC, Seror R et al (2017) 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Ann Rheum Dis 76:9–16
Shiboski CH, Shiboski SC, Seror R et al (2017) 2016 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Primary Sjögren’s Syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol 69:35–45
Tsuboi H, Hagiwara S, Asashima H et al (2017) Comparison of performance of the 2016 ACR-EULAR classification criteria for primary Sjögren’s syndrome with other sets of criteria in Japanese patients. Ann Rheum Dis 76:1980–1985
Lee J, Koh JH, Kim JW et al (2018) Performance of the 2016 ACR-EULAR classification criteria for primary Sjogren’s syndrome in a Korean cohort. Rheumatol Int 38:1651–1660
Varoni EM, Villani G, Lombardi N et al (2020) Local complications associated with labial salivary gland biopsy for diagnosis of Sjögren’s syndrome: a retrospective cohort study. J Clin Exp Dent 2(12):e713–e718
Seror R, Ravaud P, Bowman SJ et al (2010) EULAR Sjogren’s syndrome disease activity index: development of a consensus systemic disease activity index for primary Sjogren’s syndrome. Ann Rheum Dis 69:1103–1109
Chisholm DM, Mason DK (1968) Labial salivary gland biopsy in Sjögren’s disease. J Clin Pathol 21:656–660
Costa S, Quintin-Roué I, Lesourd A et al (2015) Reliability of histopathological salivary gland biopsy assessment in Sjögren’s syndrome: a multicentre cohort study. Rheumatology (Oxford) 54:1056–1064
Bankhead P, Loughrey MB, Fernández JA et al (2017) QuPath: open source software for digital pathology image analysis. Sci Rep 7:16878
Min HK, Kim SH, Lee KA, et al (2021) Correlation between salivary gland ultrasonography and scintigraphy in primary Sjögren's Syndrome. Rheumatology (Oxford) keab881 (e-pub)
Milic V, Petrovic R, Boricic I et al (2012) Ultrasonography of major salivary glands could be an alternative tool to sialoscintigraphy in the American-European classification criteria for primary Sjogren’s syndrome. Rheumatology (Oxford) 51:1081–1085
Fana V, Dohn UM, Krabbe S, Terslev L (2021) Application of the OMERACT grey-scale ultrasound scoring system for salivary glands in a single-centre cohort of patients with suspected Sjögren’s syndrome. RMD Open 7:e001516
Giovannini I, Lorenzon M, Manfrè V et al (2022) Safety, patient acceptance and diagnostic accuracy of ultrasound core needle biopsy of parotid or submandibular glands in primary Sjögren’s syndrome with suspected salivary gland lymphoma. RMD Open. 8:e001901
Milic VD, Petrovic RR, Boricic IV et al (2009) Diagnostic value of salivary gland ultrasonographic scoring system in primary Sjogren’s syndrome: a comparison with scintigraphy and biopsy. J Rheumatol 36:1495–1500
Delli K, Dijkstra PU, Stel AJ, Bootsma H, Vissink A, Spijkervet FK (2015) Diagnostic properties of ultrasound of major salivary glands in Sjögren’s syndrome: a meta-analysis. Oral Dis 21:792–800
Vinagre F, Santos MJ, Prata A, da Silva JC, Santos AI (2009) Assessment of salivary gland function in Sjögren’s syndrome: the role of salivary gland scintigraphy. Autoimmun Rev 8:672–676
Adams BK, Al Attia HM, Parkar S (2003) Salivary gland scintigraphy in Sjögren’s syndrome: are quantitative indices the answer? Nucl Med Commun 24:1011–1016
Zou Q, Jiao J, Zou MH et al (2012) Semi-quantitative evaluation of salivary gland function in Sjögren’s syndrome using salivary gland scintigraphy. Clin Rheumatol 31:1699–1705
Dugonjić S, Stefanović D, Ethurović B, Spasić-Jokić V, Ajdinović B (2014) Evaluation of diagnostic parameters from parotid and submandibular dynamic salivary glands scintigraphy and unstimulated sialometry in Sjögren’s syndrome. Hell J Nucl Med 17:116–122
Tensing EK, Nordström DC, Solovieva S et al (2003) Salivary gland scintigraphy in Sjögren’s syndrome and patients with sicca symptoms but without Sjögren’s syndrome: the psychological profiles and predictors for salivary gland dysfunction. Ann Rheum Dis 62:964–968
Nadal M, Levy M, Bakhsh A et al (2018) Salivary scintigraphy for Sjögren’s syndrome in patients with xerostomia: a retrospective study. Oral Dis 24:552–560
Kim HA, Yoon SH, Yoon JK et al (2014) Salivary gland scintigraphy in Sjögren’s syndrome. Comparison of the diagnostic performance of visual and semiquantitative analysis. Nuklearmedizin 53:139–145
Kohn WG, Ship JA, Atkinson JC, Patton LL, Fox PC (1992) Salivary gland 99mTc-scintigraphy: a grading scale and correlation with major salivary gland flow rates. J Oral Pathol Med 21:70–74
Löfgren CD, Wickström C, Sonesson M, Lagunas PT, Christersson C (2012) A systematic review of methods to diagnose oral dryness and salivary gland function. BMC Oral Health 12:29
Pijpe J, Kalk WW, Bootsma H, Spijkervet FK, Kallenberg CG, Vissink A (2007) Progression of salivary gland dysfunction in patients with Sjogren’s syndrome. Ann Rheum Dis 66:107–112
Aksoy T, Kiratli PO, Erbas B (2012) Correlations between histopathologic and scintigraphic parameters of salivary glands in patients with Sjögren’s syndrome. Clin Rheumatol 31:1365–1370
Huang J, Wu J, Zhao L et al (2020) Quantitative evaluation of salivary gland scintigraphy in Sjögren’s syndrome: comparison of diagnostic efficacy and relationship with pathological features of the salivary glands. Ann Nucl Med 34:289–298
Güne S, Yilmaz S, Karalezli A, Aktaş A (2010) Quantitative and visual evaluation of salivary and thyroid glands in patients with primary Sjögren’s syndrome using salivary gland scintigraphy: relationship with clinicopathological features of salivary, lacrimal and thyroid glands. Nucl Med Commun 31:666–672
Wise CM, Agudelo CA, Semble EL, Stump TE, Woodruff RD (1988) Comparison of parotid and minor salivary gland biopsy specimens in the diagnosis of Sjögren’s syndrome. Arthritis Rheum 31:662–666
Delli K, Haacke EA, Kroese FG et al (2016) Towards personalised treatment in primary Sjögren’s syndrome: baseline parotid histopathology predicts responsiveness to rituximab treatment. Ann Rheum Dis 75:1933–1938
Verstappen GM, Kroese FGM, Bootsma H (2019) T cells in primary Sjögren’s syndrome: targets for early intervention. Rheumatology (Oxford) 60:3088–3098
Park Y, Lee J, Koh JH et al (2020) Positive histopathologic assessment in salivary glands shows little impact on clinical features of established primary Sjögren’s syndrome in a Korean population. Clin Exp Rheumatol 38(Suppl 126):158–165
Sebastian A, Woytala P, Madej M et al (2021) Is it possible to not perform salivary gland biopsy in targeted patients according to unstimulated salivary flow results in patients with suspected Sjögren’s syndrome? Rheumatol Int 41:1125–1131
Haga HJ (2002) Clinical and immunological factors associated with low lacrimal and salivary flow rate in patients with primary Sjögren’s syndrome. J Rheumatol 29:305–308
Henriksen AM, Nossent HC (2007) Quantitative salivary gland scintigraphy can distinguish patients with primary Sjøgren’s syndrome during the evaluation of sicca symptoms. Clin Rheumatol 26:1837–1841
Kaldeway HP, Ter Borg EJ, van de Garde EMW, Habraken JBA, van Buul MMC (2019) Validation of quantitative salivary gland scintigraphy in relation to the American-European concensus criteria for Sjögren’s syndrome. Nucl Med Commun 40:343–348
Bautista-Vargas M, Vivas AJ, Tobon GJ (2020) Minor salivary gland biopsy: its role in the classification and prognosis of Sjogren’s syndrome. Autoimmun Rev 19:102690
Nakamura T, Matsui M, Uchida K et al (2004) M(3) muscarinic acetylcholine receptor plays a critical role in parasympathetic control of salivation in mice. J Physiol 558:561–575
Chen YC, Chen HY, Hsu CH (2021) Recent advances in salivary scintigraphic evaluation of salivary gland function. Diagnostics (Basel). 11:1173
Funding
This work was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea [HI16C0992].
Author information
Authors and Affiliations
Contributions
JWK, RJ, JHH, JHK, JYJ, CHS, YSA, and HAK contributed to the study design and data collection, analysis, and interpretation. JWK, RJ, JHH, YSA, and HAK contributed to the data collection and/or data interpretation. All authors revised the manuscript and gave final approval for submission.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the institutional review board of Ajou University Hospital (AJIRB-BMR-MDB-22–031).
Consent to participate
The consent to participate was waived due to the study of retrospective nature.
Consent for publication
The consent for publication was approved.
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, JW., Jin, R., Han, J.H. et al. Correlations between salivary gland scintigraphy and histopathologic data of salivary glands in patients with primary Sjogren’s syndrome. Clin Rheumatol 41, 3083–3093 (2022). https://doi.org/10.1007/s10067-022-06269-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06269-x