Abstract
Introduction
Objectives
Our aim was to analyze the link between hyperandrogenism and early clinical manifestations of osteoarthritis (OA), knee cartilage thickness, and serum cartilage oligomeric matrix protein (sCOMP) levels in patients with polycystic ovary syndrome (PCOS) and to compare them with healthy volunteers.
Methods
Fifty-four PCOS patients who met the Rotterdam criteria with phenotypes A, B, and C were included. They were compared with 26 age- and body mass index (BMI)-matched controls. Detailed anthropometric measurements and clinical evaluation for hyperandrogenism were performed for all participants who also filled in the Knee Injury and Osteoarthritis Outcome Score (KOOS) questionnaire. Furthermore, laboratory tests including sCOMP and hormone quantification were performed in a fasting stage. Finally, an ultrasound assessment was carried out in randomly selected 56 study participants.
Results
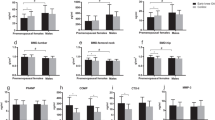
PCOS women reported more prominent knee-related symptoms (p = 0.035) and more impaired activities of daily living (ADL) (p = 0.001) than controls. Cartilage thickness of the left and right medial condyle and left lateral condyle was significantly greater in PCOS group (n = 41) than in control group (n = 15) (p = 0.05, p = 0.006, and p = 0.036, respectively). COMP correlated significantly and negatively with testosterone levels (p = 0.029, r = − 0.297) in women with PCOS and the correlation remained significant after controlling for BMI.
Conclusions
Women with PCOS may experience knee-related symptoms and impaired ADL. They had greater knee femoral cartilage thickness. Although sCOMP levels did not significantly differ between the groups, lower levels of sCOMP may be inherent to PCOS patients with higher testosterone levels.
Key Points • Although PCOS patients may experience more prominent knee related symptoms, their femoral cartilage of the knee joint is found thicker than controls. • PCOS patients did not have significantly elevated levels of sCOMP. • Lower sCOMP levels were related to higher testosterone levels. |

Similar content being viewed by others
Data availability
Data or materials are available upon reasonable request.
References
Bijlsma JWJ, Berenbaum F, Lafeber FPJG (2011) Osteoarthritis: an update with relevance for clinical practice. Lancet 377:2115–2126. https://doi.org/10.1016/S0140-6736(11)60243-2
Sowers MR, McConnell D, Jannausch M, Buyuktur AG, Hochberg M, Jamadar DA (2006) Estradiol and its metabolites and their association with knee osteoarthritis. Arthritis Rheum 54:2481–2487. https://doi.org/10.1002/art.22005
Szoeke CEI, Cicuttini FM, Guthrie JR, Clark MS, Dennerstein L (2006) Factors affecting the prevalence of osteoarthritis in healthy middle-aged women: data from the longitudinal Melbourne Women’s Midlife Health Project. Bone 39:1149–1155. https://doi.org/10.1016/j.bone.2006.05.016
Nadkar MY, Samant RS, Vaidya SS, Borges NE (1999) Relationship between osteoarthritis of knee and menopause. J Assoc Physicians India 47:1161–1163
Bergink AP, van Meurs JB, Loughlin J, Arp PP, Fang Y, Hofman A, van Leeuwen JPTM, van Duijn CM, Uitterlinden AG, Pols HAP (2003) Estrogen receptor alpha gene haplotype is associated with radiographic osteoarthritis of the knee in elderly men and women. Arthritis Rheum 48:1913–1922. https://doi.org/10.1002/art.11046
Herrero-Beaumont G, Roman-Blas JA, Castañeda S, Jimenez SA (2009) Primary osteoarthritis no longer primary: three subsets with distinct etiological, clinical, and therapeutic characteristics. Semin Arthritis Rheum 39:71–80. https://doi.org/10.1016/j.semarthrit.2009.03.006
Azziz R (2018) Polycystic ovary syndrome. Obstet Gynecol 132:321–336. https://doi.org/10.1097/AOG.0000000000002698
Azzini GOM, Santos GS, Visoni SBC, Azzini VOM, Santos RGD, Huber SC, Lana JF (2020) Metabolic syndrome and subchondral bone alterations: the rise of osteoarthritis - a review. J Clin Orthop Trauma 11:S849–S855. https://doi.org/10.1016/j.jcot.2020.06.021
Xie Y, Zhou W, Zhong Z, Zhao Z, Yu H, Huang Y, Zhang P (2021) Metabolic syndrome, hypertension, and hyperglycemia were positively associated with knee osteoarthritis, while dyslipidemia showed no association with knee osteoarthritis. Clin Rheumatol 40:711–724. https://doi.org/10.1007/s10067-020-05216-y
Senol O, Gundogdu G, Gundogdu K, Miloglu FD (2019) Investigation of the relationships between knee osteoarthritis and obesity via untargeted metabolomics analysis. Clin Rheumatol 38:1351–1360. https://doi.org/10.1007/s10067-019-04428-1
Ramly B, Afiqah-Aleng N, Mohamed-Hussein Z-A (2019) Protein-protein interaction network analysis reveals several diseases highly associated with polycystic ovarian syndrome. Int J Mol Sci. https://doi.org/10.3390/ijms20122959
Sanchez-Santos MT, Rubin KH, Glintborg D, Andersen MS, Kluzek S, Abrahamsen B (2018) Prevalence and incidence of clinically diagnosed knee, hip and hand osteoarthritis in women with polycystic ovary syndrome: a National Register-Based Study. Osteoarthritis Cartilage 26:S200. https://doi.org/10.1016/j.joca.2018.02.425
Kluzek S, Rubin KH, Sanchez-Santos M, O’Hanlon MS, Andersen M, Glintborg D, Abrahamsen B (2021) Accelerated osteoarthritis in women with polycystic ovary syndrome: a prospective nationwide registry-based cohort study. https://doi.org/10.21203/rs.3.rs-156809/v1
Eryılmaz ÖG, Kara M, Tiftik T, Aksakal FN, Uzunlar Ö, Su FA, Mollamahmutoğlu L, Özçakar L (2012) Ultrasonographic measurement of the femoral cartilage thickness in patients with polycystic ovary syndrome. Fertil Steril 97:235–237. https://doi.org/10.1016/j.fertnstert.2011.10.024
Shane Anderson A, Loeser RF (2010) Why is osteoarthritis an age-related disease? Best Pract Res Clin Rheumatol 24:15–26. https://doi.org/10.1016/j.berh.2009.08.006
Georgiev T (2020) Multimodal approach to intraarticular drug delivery in knee osteoarthritis. Rheumatol Int 40:1763–1769. https://doi.org/10.1007/s00296-020-04681-7
Lotz M, Martel-Pelletier J, Christiansen C et al (2013) Value of biomarkers in osteoarthritis: current status and perspectives. Ann Rheum Dis 72:1756–1763. https://doi.org/10.1136/annrheumdis-2013-203726
Wisłowska M, Jabłońska B (2005) Serum cartilage oligomeric matrix protein (COMP) in rheumatoid arthritis and knee osteoarthritis. Clin Rheumatol 24:278–284. https://doi.org/10.1007/s10067-004-1000-x
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81:19–25. https://doi.org/10.1016/j.fertnstert.2003.10.004
Ibáñez L, Ong KK, López-Bermejo A, Dunger DB, de Zegher F (2014) Hyperinsulinaemic androgen excess in adolescent girls. Nat Rev Endocrinol 10:499–508. https://doi.org/10.1038/nrendo.2014.58
Ferriman D, Gallwey JD (1961) Clinical assessment of body hair growth in women. J Clin Endocrinol Metab 21:1440–1447. https://doi.org/10.1210/jcem-21-11-1440
Hatch R, Rosenfield RL, Kim MH, Tredway D (1981) Hirsutism: implications, etiology, and management. Am J Obstet Gynecol 140:815–830. https://doi.org/10.1016/0002-9378(81)90746-8
Lizneva D, Gavrilova-Jordan L, Walker W, Azziz R (2016) Androgen excess: investigations and management. Best Pract Res Clin Obstet Gynaecol 37:98–118. https://doi.org/10.1016/j.bpobgyn.2016.05.003
Georgiev T, Ivanova M, Kopchev A et al (2018) Cartilage oligomeric protein, matrix metalloproteinase-3, and Coll2-1 as serum biomarkers in knee osteoarthritis: a cross-sectional study. Rheumatol Int 38:821–830. https://doi.org/10.1007/s00296-017-3887-y
Laudon J, Whittaker JL, Ren G, Jaremko JL, Emery CA, Krawetz RJ (2019) Serum cartilage oligomeric matrix protein (COMP) expression in individuals who sustained a youth sport-related intra-articular knee injury 3–10 years previously and uninjured matched controls. Osteoarthritis Cartilage 27:286–293. https://doi.org/10.1016/j.joca.2018.09.011
Denning WM, Winward JG, Pardo MB, Hopkins JT, Seeley MK (2015) Body weight independently affects articular cartilage catabolism. J Sports Sci Med 14:290–296
Golightly YM, Marshall SW, Kraus VB, Renner JB, Villaveces A, Casteel C, Jordan JM (2011) Biomarkers of incident radiographic knee osteoarthritis: do they vary by chronic knee symptoms? Arthritis Rheum 63:2276–2283. https://doi.org/10.1002/art.30412
Da Silva JA, Larbre JP, Spector TD, Perry LA, Scott DL, Willoughby DA (1993) Protective effect of androgens against inflammation induced cartilage degradation in male rodents. Ann Rheum Dis 52:285–291. https://doi.org/10.1136/ard.52.4.285
Valdes AM, Van Oene M, Hart DJ, Surdulescu GL, Loughlin J, Doherty M, Spector TD (2006) Reproducible genetic associations between candidate genes and clinical knee osteoarthritis in men and women. Arthritis Rheum 54:533–539. https://doi.org/10.1002/art.21621
Jin S-Y, Hong S-J, Yang HI, Park S, Yoo M-C, Lee HJ et al (2004) Estrogen receptor-alpha gene haplotype is associated with primary knee osteoarthritis in Korean population. Arthritis Res Ther 6:R415–R421. https://doi.org/10.1186/ar1207
Cooper C, Egger P, Coggon D, Hart DJ, Masud T, Cicuttini F, Doyle DV, Spector TD (1996) Generalized osteoarthritis in women: pattern of joint involvement and approaches to definition for epidemiological studies. J Rheumatol 23:1938–1942
DeVane GW, Czekala NM, Judd HL, Yen SS (1975) Circulating gonadotropins, estrogens, and androgens in polycystic ovarian disease. Am J Obstet Gynecol 121:496–500. https://doi.org/10.1016/0002-9378(75)90081-2
Gambineri A, Pelusi C, Vicennati V, Pagotto U, Pasquali R (2002) Obesity and the polycystic ovary syndrome. Int J Obes Relat Metab Disord 26:883–896. https://doi.org/10.1038/sj.ijo.0801994
Gateva A, Kamenov Z (2012) Cardiovascular risk factors in Bulgarian patients with polycystic ovary syndrome and/or obesity. Obstet Gynecol Int 2012:306347. https://doi.org/10.1155/2012/306347
Barnard L, Ferriday D, Guenther N, Strauss B, Balen AH, Dye L (2007) Quality of life and psychological well being in polycystic ovary syndrome. Hum Reprod 22:2279–2286. https://doi.org/10.1093/humrep/dem108
Kara M, Okur A, Serin Hİ (2019) Ultrasonographic evaluation of femoral cartilage thickness in patients with polycystic ovary syndrome. Acta Clin Croat 58:103–106. https://doi.org/10.20471/acc.2019.58.01.13
Calvo E, Palacios I, Delgado E, Sanchez-Pernaute O, Largo R, Egido J, Herrero-Beaumont G (2004) Histopathological correlation of cartilage swelling detected by magnetic resonance imaging in early experimental osteoarthritis. Osteoarthritis Cartilage 12:878–886. https://doi.org/10.1016/j.joca.2004.07.007
Sakthiswary R, Rajalingam S, Hussein H, Sridharan R, Asrul AW (2017) Cartilage oligomeric matrix protein (COMP) in rheumatoid arthritis and its correlation with sonographic knee cartilage thickness and disease activity. Clin Rheumatol 36:2683–2688. https://doi.org/10.1007/s10067-017-3817-0
Kühne SA, Neidhart M, Everson MP, Häntzschel H, Fine PR, Gay S, Häuselmann HJ, Gay RE (1998) Persistent high serum levels of cartilage oligomeric matrix protein in a subgroup of patients with traumatic knee injury. Rheumatol Int 18:21–25. https://doi.org/10.1007/s002960050049
Funding
The study was performed with the financial support of Medical University-Sofia, Bulgaria, grant “Young Investigator” Project No 8378/20.11.2019, Contract D-85/24.06.2020.
Author information
Authors and Affiliations
Contributions
PK: Study design, data acquisition, data analysis, and data interpretation, critical revision, and final approval of the manuscript. TG: Study design, data interpretation, ultrasound assessment, critical revision, and final approval of the manuscript. JH: Data interpretation, immunological assay evaluation, critical revision, and final approval of the manuscript. AG: Data interpretation, critical revision, and final approval of the manuscript. ZK: Data interpretation, critical revision, and final approval of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study protocol was approved by the Research Ethics Committee of the Medical University of Sofia, Bulgaria with approval protocol number 05/28.02.2020.
Consent to participate
Written informed consent was obtained from all participants included in the study.
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kabakchieva, P., Georgiev, T., Gateva, A. et al. Polycystic ovary syndrome and (pre)osteoarthritis: assessing the link between hyperandrogenism in young women and cartilage oligomeric matrix protein as a marker of cartilage breakdown. Clin Rheumatol 40, 4217–4223 (2021). https://doi.org/10.1007/s10067-021-05753-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-021-05753-0