Abstract
Introduction/objectives
The effects of biologic disease–modifying antirheumatic drugs (bDMARDs) in patients with rheumatoid arthritis (RA) and cancer are largely unknown. We examined overall survival (OS) in patients with RA and solid malignancies receiving bDMARDs.
Methods
We performed a retrospective cohort study of patients with RA and solid malignancies seen at MD Anderson Cancer Center between 2002 and 2014. Cox proportional hazard regression models, stratified by tumor type and stage, were fit evaluating use of bDMARDs as a time fixed and time varying covariate.
Results
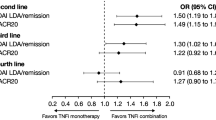
We identified 431 RA patients with solid malignancies: 111 (26%) received bDMARDs after their cancer diagnosis. Median OS from cancer diagnosis was 16.1 years. Of the patients receiving bDMARDs, most had localized disease, and only 14 (13%) had advanced cancer. In the stratified Cox models no statistically significant differences were observed between patients who received tumor necrosis factor inhibitors (TNFi) or patients who received nonTNFi, compared with those who did not receive bDMARDs (hazard ratio (HR), 0.67; 95% confidence interval (CI), 0.31, 1.44; HR, 1.10; 95% CI, 0.26, 4.60 respectively). In breast cancer patients, those receiving TNFi or nonTNFi had a numerically higher but statistically nonsignificant HR compared with those who did not receive bDMARD: HR, 1.40 (95% CI, 0.42, 4.73), and HR, 1.37 (95% CI, 0.22, 8.42) respectively.
Conclusion
No significant differences in OS were observed between patients who received bDMARDs and those who did not. Additional data is needed to evaluate other cancer outcomes such as recurrence and progression, and patients with advanced cancer.
Key Points •We found no statistically significant differences in OS between patients with RA and concomitant solid malignancies who received bDMARDs and those who did not. •Most patients who received bDMARDs had been diagnosed with early stage cancer •As few patients with advanced cancer received bDMARDs safety for this group cannot be established •No significant differences were observed between TNFi and nonTNFi, but the sample size was small |

Similar content being viewed by others
References
Balkwill F (2009) Tumour necrosis factor and cancer. Nat Rev Cancer 9:361–371
Lebrec H, Ponce R, Preston BD, Iles J, Born TL, Hooper M (2015) Tumor necrosis factor, tumor necrosis factor inhibition, and cancer risk. Curr Med Res Opin 31:557–574
Tarella C, Passera R, Magni M, Benedetti F, Rossi A, Gueli A, Patti C, Parvis G, Ciceri F, Gallamini A, Cortelazzo S, Zoli V, Corradini P, Carobbio A, Mulé A, Bosa M, Barbui A, di Nicola M, Sorio M, Caracciolo D, Gianni AM, Rambaldi A (2011) Risk factors for the development of secondary malignancy after high-dose chemotherapy and autograft, with or without rituximab: a 20-year retrospective follow-up study in patients with lymphoma. J Clin Oncol 29:814–824
Askling J, Bongartz T (2008) Malignancy and biologic therapy in rheumatoid arthritis. Curr Opin Rheumatol 20:334–339
Bongartz T, Sutton AJ, Sweeting MJ, Buchan I, Matteson EL, Montori V (2006) Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. Jama 295:2275–2285
Callegari PE, Schaible TF, Boscia JA (2006) Risk of serious infections and malignancies with anti-TNF antibody therapy in rheumatoid arthritis. Jama 296:2201–2204
Geborek P, Bladström A, Turesson C, Gulfe A, Petersson IF, Saxne T, Olsson H, Jacobsson LT (2005) Tumour necrosis factor blockers do not increase overall tumour risk in patients with rheumatoid arthritis, but may be associated with an increased risk of lymphomas. Ann Rheum Dis 64:699–703
Setoguchi S, Solomon DH, Weinblatt ME, Katz JN, Avorn J, Glynn RJ, Cook EF, Carney G, Schneeweiss S (2006) Tumor necrosis factor α antagonist use and cancer in patients with rheumatoid arthritis. Arthritis Rheum 54:2757–2764
Strangfeld A, Hierse F, Rau R, Burmester GR, Krummel-Lorenz B, Demary W, Listing J, Zink A (2010) Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther 12:R5
Raaschou P, Soderling J, Turesson C, Askling J, Group AS (2018) Tumor necrosis factor inhibitors and cancer recurrence in Swedish patients with rheumatoid arthritis: a nationwide population-based cohort study. Ann Intern Med 169:291–299
Dreyer L, Cordtz RL, Hansen IMJ, Kristensen LE, Hetland ML, Mellemkjaer L (2018) Risk of second malignant neoplasm and mortality in patients with rheumatoid arthritis treated with biological DMARDs: a Danish population-based cohort study. Ann Rheum Dis 77:510–514
Waljee AK, Higgins PDR, Jensen CB, Villumsen M, Cohen-Mekelburg SA, Wallace BI, Berinstein JA, Allin KH, Jess T (2020) Anti-tumour necrosis factor-alpha therapy and recurrent or new primary cancers in patients with inflammatory bowel disease, rheumatoid arthritis, or psoriasis and previous cancer in Denmark: a nationwide, population-based cohort study. Lancet Gastroenterol Hepatol 5(3):276–284
Singh JA, Furst DE, Bharat A, Curtis JR, Kavanaugh AF, Kremer JM, Moreland LW, O’Dell J, Winthrop KL, Beukelman T, Bridges SL Jr, Chatham WW, Paulus HE, Suarez-almazor M, Bombardier C, Dougados M, Khanna D, King CM, Leong AL, Matteson EL, Schousboe JT, Moynihan E, Kolba KS, Jain A, Volkmann ER, Agrawal H, Bae S, Mudano AS, Patkar NM, Saag KG (2012) 2012 update of the 2008 American College of Rheumatology recommendations for the use of disease-modifying antirheumatic drugs and biologic agents in the treatment of rheumatoid arthritis. Arthritis Care Res 64:625–639
Bombardier C, Hazlewood GS, Akhavan P et al (2012) Canadian rheumatology association recommendations for the pharmacological management of rheumatoid arthritis with traditional and biologic disease-modifying antirheumatic drugs: part II safety. J Rheumatol 39:1583–1602
Singh JA, Saag KG, Bridges SL Jr et al (2016) 2015 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheum 68:1–26
Lopez-Olivo MA, Colmegna I, Karpes AR et al (2020) Systematic review of recommendations on the use of disease-modifying antirheumatic drugs in patients with rheumatoid arthritis and cancer. Arthritis Care Res 72(3):309–318
Pappas DA, Rebello S, Liu M, Schenfeld J, Li YF, Collier DH, Accortt NA (2019) Therapy with biologic agents after diagnosis of solid malignancies: results from the Corrona Registry. J Rheumatol 46:1438–1444
Pundole X, Zamora NV, Siddhanamatha H, Lin H, Tayar J, Hong LC, Li L, Suarez-Almazor ME (2020) Utilization of biologic disease-modifying anti-rheumatic drugs in patients with rheumatoid arthritis and cancer. Clin Rheumatol 39(3):787–794
Mariette X, Matucci-Cerinic M, Pavelka K, Taylor P, van Vollenhoven R, Heatley R, Walsh C, Lawson R, Reynolds A, Emery P (2011) Malignancies associated with tumour necrosis factor inhibitors in registries and prospective observational studies: a systematic review and meta-analysis. Ann Rheum Dis 70:1895–1904
Bongartz T, Warren FC, Mines D, Matteson EL, Abrams KR, Sutton AJ (2009) Etanercept therapy in rheumatoid arthritis and the risk of malignancies: a systematic review and individual patient data meta-analysis of randomised controlled trials. Ann Rheum Dis 68:1177–1183
Xie W, Xiao S, Huang Y, Sun X, Gao D, Ji LL, Li G, Zhang Z (2020) A meta-analysis of biologic therapies on risk of new or recurrent cancer in patients with rheumatoid arthritis and a prior malignancy. Rheumatology (Oxford) 59(5):930–939
Howlader N, Noone A, Krapcho M, et al. (2019) SEER Cancer Statistics Review, 1975–2016, National Cancer Institute Bethesda, MD. National Cancer Institute 1423–37
Funding
This research was supported by the Advancing Science through Pfizer–Investigator Research Exchange program, a competitive grants program supported by Pfizer, Pfizer ASPIRE Rheumatology award #WI195021.
The statistical analysis was supported in part by the Cancer Center Support Grant (NCI Grant P30 CA016672).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Xerxes Pundole—none
Natalia V. Zamora—none
Harish Siddhanamatha—none
Heather Lin—none
Cheuk Hong Leung—none
Natalia V. Zamora—none.
Maria E. Suarez-Almazor—Dr. Suarez-Almazor has been a consultant for Pfizer, AbbVie, and Agile Pharmaceuticals.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Prior abstract publication
Part of the data of this project was presented as a poster at the 2018 American College of Rheumatology annual meeting.
Pundole X, Zamora N, Siddhanamatha H, Tayar J, Leung CH, Lin H, Suarez-Almazor M. Time Dependent Effect of Biologic Therapy on Overall Survival in Patients with Rheumatoid Arthritis and Cancer [abstract]. Arthritis Rheumatol. 2018; 70 (suppl 10). https://acrabstracts.org/abstract/time-dependent-effect-of-biologic-therapy-on-overall-survival-in-patients-with-rheumatoid-arthritis-and-cancer/. Accessed November 11, 2019
Electronic supplementary material
ESM 1
(DOCX 42 kb)
Rights and permissions
About this article
Cite this article
Pundole, X., Zamora, N.V., Siddhanamatha, H. et al. Overall survival in patients with rheumatoid arthritis and solid malignancies receiving biologic disease-modifying antirheumatic therapy. Clin Rheumatol 39, 2943–2950 (2020). https://doi.org/10.1007/s10067-020-05318-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05318-7