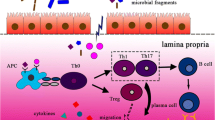
Abstract
Objective rheumatism refers to a large group of diseases with different etiology, mainly characterized by autoimmune disorder. Intestinal flora combines with the digestive organs of the human body to synthesize and secrete the key substances of growth. Several studies have reported abnormal intestinal flora in rheumatic diseases. The purposes of this research were to review the primary studies and figure out the relationship between intestinal flora and rheumatic disease activity. The article search was based on the database of PubMed (MEDLINE), EMBASE, Cochrane to collect English language studies that were published from 1985 to 2019. The articles concerning the intestinal flora and disease activities of rheumatic diseases were classified by disease types, and the relationship between disease activities and intestinal flora was summarized. Eight rheumatic diseases were included in the study. It was found that the changes of intestinal flora were significantly correlated with the activities of rheumatic diseases. There were significant differences in the classification of disease activity and the composition of intestinal flora. Interfering with the composition of intestinal flora can apparently modulate the development of disease. But how to apply such findings is rarely reported. The study finds out that intestinal flora disorder is linked to the activity of rheumatic diseases. But which specific gut flora is connected to the disease activity needs further researches. More discussion is needed on how to apply the results to clinical treatment.
Similar content being viewed by others
References
Ostrov BE, Amsterdam D (2017) Immunomodulatory interplay of the microbiome and therapy of rheumatic diseases. Immunol Investig 46(8):769–792. https://doi.org/10.1080/08820139.2017.1373828
Yakimenko E, Zakatova L, Tbilieli V, Antipova N, Kolomiets S, Tikhonchuk N, Bondar V, Klochko V (2019) Current trends in the prevention, diagnosis and treatment of rheumatic fever and rheumatic heart disease (Review). Georgian Med News (297):88–94
Koren O, Goodrich JK, Cullender TC, Spor A, Laitinen K, Backhed HK, Gonzalez A, Werner JJ, Angenent LT, Knight R, Backhed F, Isolauri E, Salminen S, Ley RE (2012) Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell 150(3):470–480. https://doi.org/10.1016/j.cell.2012.07.008
Dubois G, Girard C, Lapointe FJ, Shapiro BJ (2017) The Inuit gut microbiome is dynamic over time and shaped by traditional foods. Microbiome 5(1):151. https://doi.org/10.1186/s40168-017-0370-7
Jha AR, Davenport ER, Gautam Y, Bhandari D, Tandukar S, Ng KM, Fragiadakis GK, Holmes S, Gautam GP, Leach J, Sherchand JB, Bustamante CD, Sonnenburg JL (2018) Gut microbiome transition across a lifestyle gradient in Himalaya. PLoS Biol 16(11):e2005396. https://doi.org/10.1371/journal.pbio.2005396
Iizumi T, Battaglia T, Ruiz V, Perez Perez GI (2017) Gut microbiome and antibiotics. Arch Med Res 48(8):727–734. https://doi.org/10.1016/j.arcmed.2017.11.004
Galland L (2014) The gut microbiome and the brain. J Med Food 17(12):1261–1272. https://doi.org/10.1089/jmf.2014.7000
Swidsinski A, Loening-Baucke V, Lochs H, Hale LP (2005) Spatial organization of bacterial flora in normal and inflamed intestine: a fluorescence in situ hybridization study in mice. World J Gastroenterol 11(8):1131–1140. https://doi.org/10.3748/wjg.v11.i8.1131
Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, Magris M, Hidalgo G, Baldassano RN, Anokhin AP, Heath AC, Warner B, Reeder J, Kuczynski J, Caporaso JG, Lozupone CA, Lauber C, Clemente JC, Knights D, Knight R, Gordon JI (2012) Human gut microbiome viewed across age and geography. Nature 486(7402):222–227. https://doi.org/10.1038/nature11053
Kau AL, Ahern PP, Griffin NW, Goodman AL, Gordon JI (2011) Human nutrition, the gut microbiome and the immune system. Nature 474(7351):327–336. https://doi.org/10.1038/nature10213
De Luca F, Shoenfeld Y (2019) The microbiome in autoimmune diseases. Clin Exp Immunol 195(1):74–85. https://doi.org/10.1111/cei.13158
Rosser EC, Mauri C (2016) A clinical update on the significance of the gut microbiota in systemic autoimmunity. J Autoimmun 74:85–93. https://doi.org/10.1016/j.jaut.2016.06.009
Chu F, Shi M, Lang Y, Shen D, Jin T, Zhu J, Cui L (2018) Gut microbiota in multiple sclerosis and experimental autoimmune encephalomyelitis: current applications and future perspectives. Mediat Inflamm 2018:8168717–8168717. https://doi.org/10.1155/2018/8168717
He Z, Shao T, Li H, Xie Z, Wen C (2016) Alterations of the gut microbiome in Chinese patients with systemic lupus erythematosus. Gut pathogens 8:64. https://doi.org/10.1186/s13099-016-0146-9
Li Y, Wang H, Li X, Li H, Zhang Q, Zhou H, He Y, Li P, Fu C, Zhang X, Qiu Y, Li JL (2019) Disordered intestinal microbes are associated with the activity of systemic lupus erythematosus. Clin Sci 133:821–838. https://doi.org/10.1042/CS20180841
Katz-Agranov N, Zandman-Goddard G (2017) The microbiome and systemic lupus erythematosus. Immunol Res 65(2):432–437. https://doi.org/10.1007/s12026-017-8906-2
Silverman GJ (2019) The microbiome in SLE pathogenesis. Nat Rev Rheumatol 15(2):72–74. https://doi.org/10.1038/s41584-018-0152-z
Azzouz D, Omarbekova A, Heguy A, Schwudke D, Gisch N, Rovin BH, Caricchio R, Buyon JP, Alekseyenko AV, Silverman GJ (2019) Lupus nephritis is linked to disease-activity associated expansions and immunity to a gut commensal. Ann Rheum Dis 78(7):947–956. https://doi.org/10.1136/annrheumdis-2018-214856
Hevia A, Milani C, Lopez P, Cuervo A, Arboleya S, Duranti S, Turroni F, Gonzalez S, Suarez A, Gueimonde M, Ventura M, Sanchez B, Margolles A (2014) Intestinal dysbiosis associated with systemic lupus erythematosus. mBio 5(5):e01548–e01514. https://doi.org/10.1128/mBio.01548-14
Luo XM, Edwards MR, Mu Q, Yu Y, Vieson MD, Reilly CM, Ahmed SA, Bankole AA (2018) Gut microbiota in human systemic lupus erythematosus and a mouse model of lupus. Appl Environ Microbiol 84(4). https://doi.org/10.1128/AEM.02288-17
Picchianti-Diamanti A, Panebianco C, Salemi S, Sorgi ML, Di Rosa R, Tropea A, Sgrulletti M, Salerno G, Terracciano F, D'Amelio R, Lagana B, Pazienza V (2018) Analysis of gut microbiota in rheumatoid arthritis patients: disease-related dysbiosis and modifications induced by etanercept. Int J Mol Sci 19(10). https://doi.org/10.3390/ijms19102938
Jeong Y, Kim JW, You HJ, Park SJ, Lee J, Ju JH, Park MS, Jin H, Cho ML, Kwon B, Park SH, Ji GE (2019) Gut microbial composition and function are altered in patients with early rheumatoid arthritis. J Clin Med 8(5). https://doi.org/10.3390/jcm8050693
Hanninen KK, Rauma AL, Nenonen M, Torronen R, Hakkinen AS, Adlercreutz H, Laakso J (2000) Antioxidants in vegan diet and rheumatic disorders. Toxicology 155(1–3):45–53. https://doi.org/10.1016/s0300-483x(00)00276-6
Toivanen P, Eerola E (2002) A vegan diet changes the intestinal flora. Rheumatology 41(8):950–951. https://doi.org/10.1093/rheumatology/41.8.950
Peltonen R, Kjeldsen-Kragh J, Haugen M, Tuominen J, Toivanen P, Forre O, Eerola E (1994) Changes of faecal flora in rheumatoid arthritis during fasting and one-year vegetarian diet. Br J Rheumatol 33(7):638–643. https://doi.org/10.1093/rheumatology/33.7.638
Kjeldsen-Kragh J (1999) Rheumatoid arthritis treated with vegetarian diets. Am J Clin Nutr 70(3 Suppl):594S–600S. https://doi.org/10.1093/ajcn/70.3.594s
Mandel DR, Eichas K, Holmes J (2010) Bacillus coagulans: a viable adjunct therapy for relieving symptoms of rheumatoid arthritis according to a randomized, controlled trial. BMC Complement Altern Med 10:1. https://doi.org/10.1186/1472-6882-10-1
Alipour B, Homayouni-Rad A, Vaghef-Mehrabany E, Sharif SK, Vaghef-Mehrabany L, Asghari-Jafarabadi M, Nakhjavani MR, Mohtadi-Nia J (2014) Effects of Lactobacillus casei supplementation on disease activity and inflammatory cytokines in rheumatoid arthritis patients: a randomized double-blind clinical trial. Int J Rheum Dis 17(5):519–527. https://doi.org/10.1111/1756-185X.12333
Zamani B, Golkar HR, Farshbaf S, Emadi-Baygi M, Tajabadi-Ebrahimi M, Jafari P, Akhavan R, Taghizadeh M, Memarzadeh MR, Asemi Z (2016) Clinical and metabolic response to probiotic supplementation in patients with rheumatoid arthritis: a randomized, double-blind, placebo-controlled trial. Int J Rheum Dis 19(9):869–879. https://doi.org/10.1111/1756-185X.12888
Yang L, Wang L, Wang X, Xian CJ, Lu H (2016) A possible role of intestinal microbiota in the pathogenesis of ankylosing spondylitis. Int J Mol Sci 17(12). https://doi.org/10.3390/ijms17122126
Breban M, Tap J, Leboime A, Said-Nahal R, Langella P, Chiocchia G, Furet JP, Sokol H (2017) Faecal microbiota study reveals specific dysbiosis in spondyloarthritis. Ann Rheum Dis 76(9):1614–1622. https://doi.org/10.1136/annrheumdis-2016-211064
Mielants H, Veys EM, Cuvelier C, De Vos M, Botelberghe L (1985) HLA-B27 related arthritis and bowel inflammation. Part 2. Ileocolonoscopy and bowel histology in patients with HLA-B27 related arthritis. J Rheumatol 12(2):294–298
Wen C, Zheng Z, Shao T, Liu L, Xie Z, Le Chatelier E, He Z, Zhong W, Fan Y, Zhang L, Li H, Wu C, Hu C, Xu Q, Zhou J, Cai S, Wang D, Huang Y, Breban M, Qin N, Ehrlich SD (2017) Quantitative metagenomics reveals unique gut microbiome biomarkers in ankylosing spondylitis. Genome Biol 18(1):142. https://doi.org/10.1186/s13059-017-1271-6
Scarpa R, Manguso F, D'Arienzo A, D'Armiento FP, Astarita C, Mazzacca G, Ayala F (2000) Microscopic inflammatory changes in colon of patients with both active psoriasis and psoriatic arthritis without bowel symptoms. J Rheumatol 27(5):1241–1246
Gill T, Asquith M, Rosenbaum JT, Colbert RA (2015) The intestinal microbiome in spondyloarthritis. Curr Opin Rheumatol 27(4):319–325. https://doi.org/10.1097/BOR.0000000000000187
Seror R, Ravaud P, Bowman SJ, Baron G, Tzioufas A, Theander E, Gottenberg JE, Bootsma H, Mariette X, Vitali C, Force ESsT (2010) EULAR Sjogren's syndrome disease activity index: development of a consensus systemic disease activity index for primary Sjogren's syndrome. Ann Rheum Dis 69(6):1103–1109. https://doi.org/10.1136/ard.2009.110619
Ramos-Casals M, Brito-Zeron P, Solans R, Camps MT, Casanovas A, Sopena B, Diaz-Lopez B, Rascon FJ, Qanneta R, Fraile G, Perez-Alvarez R, Callejas JL, Ripoll M, Pinilla B, Akasbi M, Fonseca E, Canora J, Nadal ME, de la Red G, Fernandez-Regal I, Jimenez-Heredia I, Bosch JA, Ayala MD, Morera-Morales L, Maure B, Mera A, Ramentol M, Retamozo S, Kostov B, Group SSS, Autoimmune Diseases Study Group of the Spanish Society of Internal M (2014) Systemic involvement in primary Sjogren's syndrome evaluated by the EULAR-SS disease activity index: analysis of 921 Spanish patients (GEAS-SS Registry). Rheumatology 53(2):321–331. https://doi.org/10.1093/rheumatology/ket349
Sheikh SH, Shaw-Stiffel TA (1995) The gastrointestinal manifestations of Sjogren's syndrome. Am J Gastroenterol 90(1):9–14
de Paiva CS, Jones DB, Stern ME, Bian F, Moore QL, Corbiere S, Streckfus CF, Hutchinson DS, Ajami NJ, Petrosino JF, Pflugfelder SC (2016) Altered mucosal microbiome diversity and disease severity in Sjogren syndrome. Sci Rep 6:23561. https://doi.org/10.1038/srep23561
Seror R, Meiners P, Baron G, Bootsma H, Bowman SJ, Vitali C, Gottenberg JE, Theander E, Tzioufas A, De Vita S, Ramos-Casals M, Dorner T, Quartuccio L, Ravaud P, Mariette X, Force EST (2016) Development of the ClinESSDAI: a clinical score without biological domain. A tool for biological studies. Ann Rheum Dis 75(11):1945–1950. https://doi.org/10.1136/annrheumdis-2015-208504
Seror R, Ravaud P, Mariette X, Bootsma H, Theander E, Hansen A, Ramos-Casals M, Dorner T, Bombardieri S, Hachulla E, Brun JG, Kruize AA, Praprotnik S, Tomsic M, Gottenberg JE, Devauchelle V, Devita S, Vollenweider C, Mandl T, Tzioufas A, Carsons S, Saraux A, Sutcliffe N, Vitali C, Bowman SJ, Force ESsT (2011) EULAR Sjogren's Syndrome Patient Reported Index (ESSPRI): development of a consensus patient index for primary Sjogren's syndrome. Ann Rheum Dis 70(6):968–972. https://doi.org/10.1136/ard.2010.143743
Mandl T, Marsal J, Olsson P, Ohlsson B, Andreasson K (2017) Severe intestinal dysbiosis is prevalent in primary Sjogren's syndrome and is associated with systemic disease activity. Arthritis Res Ther 19(1):237. https://doi.org/10.1186/s13075-017-1446-2
Kamei R, Yamaoka T, Ikinaga K, Murota H, Shimizu K, Katayama I (2017) Successful treatment of a refractory dysbiotic intestinal pseudo-obstruction in a patient with systemic sclerosis-polymyositis overlap syndrome by intravenous immunoglobulin administration possibly related to gut flora normalisation. Clin Exp Rheumatol 35 Suppl 106(4):214–215
Patrone V, Puglisi E, Cardinali M, Schnitzler TS, Svegliati S, Festa A, Gabrielli A, Morelli L (2017) Gut microbiota profile in systemic sclerosis patients with and without clinical evidence of gastrointestinal involvement. Sci Rep 7(1):14874. https://doi.org/10.1038/s41598-017-14889-6
Andreasson K, Alrawi Z, Persson A, Jonsson G, Marsal J (2016) Intestinal dysbiosis is common in systemic sclerosis and associated with gastrointestinal and extraintestinal features of disease. Arthritis Res Ther 18(1):278. https://doi.org/10.1186/s13075-016-1182-z
Volkmann ER (2017) Intestinal microbiome in scleroderma: recent progress. Curr Opin Rheumatol 29(6):553–560. https://doi.org/10.1097/BOR.0000000000000429
Bellocchi C, Fernandez-Ochoa A, Montanelli G, Vigone B, Santaniello A, Milani C, Quirantes-Pine R, Borras-Linares I, Ventura M, Segura-Carrettero A, Alarcon-Riquelme ME, Beretta L (2018) Microbial and metabolic multi-omic correlations in systemic sclerosis patients. Ann N Y Acad Sci 1421(1):97–109. https://doi.org/10.1111/nyas.13736
Sawadpanich K, Soison P, Chunlertrith K, Mairiang P, Sukeepaisarnjaroen W, Sangchan A, Suttichaimongkol T, Foocharoen C (2019) Prevalence and associated factors of small intestinal bacterial overgrowth among systemic sclerosis patients. Int J Rheum Dis 22(4):695–699. https://doi.org/10.1111/1756-185X.13495
Marighela TF, Arismendi MI, Marvulle V, Brunialti MKC, Salomao R, Kayser C (2019) Effect of probiotics on gastrointestinal symptoms and immune parameters in systemic sclerosis: a randomized placebo-controlled trial. Rheumatology. 58:1985–1990. https://doi.org/10.1093/rheumatology/kez160
Consolandi C, Turroni S, Emmi G, Severgnini M, Fiori J, Peano C, Biagi E, Grassi A, Rampelli S, Silvestri E, Centanni M, Cianchi F, Gotti R, Emmi L, Brigidi P, Bizzaro N, De Bellis G, Prisco D, Candela M, D'Elios MM (2015) Behcet's syndrome patients exhibit specific microbiome signature. Autoimmun Rev 14(4):269–276. https://doi.org/10.1016/j.autrev.2014.11.009
Ye Z, Zhang N, Wu C, Zhang X, Wang Q, Huang X, Du L, Cao Q, Tang J, Zhou C, Hou S, He Y, Xu Q, Xiong X, Kijlstra A, Qin N, Yang P (2018) A metagenomic study of the gut microbiome in Behcet's disease. Microbiome 6(1):135. https://doi.org/10.1186/s40168-018-0520-6
Kim SE (2019) Importance of nutritional therapy in the management of intestinal diseases: beyond energy and nutrient supply. Intest Res 17:443–454. https://doi.org/10.5217/ir.2019.00075
Hvas CL, Dahl Jorgensen SM, Jorgensen SP, Storgaard M, Lemming L, Hansen MM, Erikstrup C, Dahlerup JF (2019) Fecal microbiota transplantation is superior to Fidaxomicin for treatment of recurrent Clostridium difficile infection. Gastroenterology 156(5):1324–1332 e1323. https://doi.org/10.1053/j.gastro.2018.12.019
Abdelhamid L, Luo XM (2018) Retinoic acid, leaky gut, and autoimmune diseases. Nutrients 10(8). https://doi.org/10.3390/nu10081016
Kohler CA, Maes M, Slyepchenko A, Berk M, Solmi M, Lanctot KL, Carvalho AF (2016) The gut-brain Axis, including the microbiome, leaky gut and bacterial translocation: mechanisms and pathophysiological role in Alzheimer's disease. Curr Pharm Des 22(40):6152–6166. https://doi.org/10.2174/1381612822666160907093807
Funding
National Natural Science Foundation of China, Grant/Award Number: 81671616, 81871278, and 81801610.
Postgraduate Research & Practice Innovation Program of Jiangsu Province, Grant/Award Number: KYCX18_2410. Special project of clinical medicine of Nantong University: 2019LQ001
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bao, Y., Dong, C., Ji, J. et al. Dysregulation of gut microbiome is linked to disease activity of rheumatic diseases. Clin Rheumatol 39, 2523–2528 (2020). https://doi.org/10.1007/s10067-020-05170-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05170-9