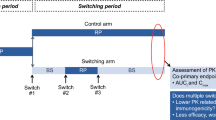
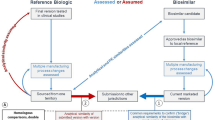
Abstract
The manufacture of biologics is a complex process involving numerous steps. Over time, differences may arise as a result of planned changes to the manufacturing processes of a biologic from the same manufacturer. Comparability is the regulatory process that outlines the scope of an assessment required of an already licensed biologic after a manufacturing process change made by the same manufacturer. The aim of a comparability assessment is to demonstrate that any pre-manufacturing and post-manufacturing changes have no adverse impact on quality, safety, and efficacy of the biologic. A comparability assessment is distinct from a biosimilarity assessment, which involves extensive assessment of a biologic that is highly similar to the originator (reference product) in terms of quality, safety, and efficacy. The US Food and Drug Administration, European Medicines Agency, and World Health Organization have applied the fundamental comparability concepts into their respective biosimilarity guidance documents. In this review, we examine the rationale behind the distinct, highly regulated approval processes governing changes that may occur over time to an originator biologic due to planned manufacturing changes (as described by a comparability exercise) and those that outline the approval of a proposed biosimilar drug, based on its relationship with the reference product (biosimilarity evaluations).


Similar content being viewed by others
References
Morrow T, Felcone LH (2004) Defining the difference: what makes biologics unique. Biotechnol Healthc 1:24–29
Kuhlmann M, Covic A (2006) The protein science of biosimilars. Nephrol Dial Transplant 21(Suppl 5):v4–8. doi:10.1093/ndt/gfl474
US Food and Drug Admiistration, Troy DE (2013) Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Amendments). US Department of Heralth & Human Services. http://www.fda.gov/newsevents/testimony/ucm115033.htm. Accessed 5 July 2016
US Food and Drug Administration (2011) Submission of summary bioequivalence data for ANDAs. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER). http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM134846.pdf. Accessed 5 July 2016
Morais JA, Lobato Mdo R (2010) The new European Medicines Agency guideline on the investigation of bioequivalence. Basic Clin Pharmacol Toxicol 106:221–225. doi:10.1111/j.1742-7843.2009.00518.x
Meyer MC (2001) United States Food and Drug Administration requirements for approval of generic drug products. J Clin Psychiatry 62(Suppl 5):4–9, discussion 23-24
Mellstedt H, Niederwieser D, Ludwig H (2008) The challenge of biosimilars. Ann Oncol 19:411–419. doi:10.1093/annonc/mdm345
Zelenetz AD, Ahmed I, Braud EL, Cross JD, Davenport-Ennis N, Dickinson BD, Goldberg SE, Gottlieb S, Johnson PE, Lyman GH, Markus R, Matulonis UA, Reinke D, Li EC, DeMartino J, Larsen JK, Hoffman JM (2011) NCCN biosimilars white paper: regulatory, scientific, and patient safety perspectives. J Natl Compr Canc Netw 9(Suppl 4):S1–22
Walsh G (2010) Biopharmaceutical benchmarks 2010. Nat Biotechnol 28:917–924. doi:10.1038/nbt0910-917
Committee for Medicinal Products for Human Use (CHMP) (2014) Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance: non-clinical and clinical issues. European Medicines Agency. http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2015/01/WC500180219.pdf. Accessed 5 July 2016
US Food and Drug Administration (2015) Scientific considerations in demonstrating bosimilarity to a reference product. Department of Health and Human Services, Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER). http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM291128.pdf. Accessed 5 July 2016
Garces S, Antunes M, Benito-Garcia E, da Silva JC, Aarden L, Demengeot J (2014) A preliminary algorithm introducing immunogenicity assessment in the management of patients with RA receiving tumour necrosis factor inhibitor therapies. Ann Rheum Dis 73:1138–1143. doi:10.1136/annrheumdis-2013-203296
Declerck PJ (2013) Biosimilar monoclonal antibodies: a science-based regulatory challenge. Expert Opin Biol Ther 13:153–156. doi:10.1517/14712598.2012.758710
Dorner T, Strand V, Castaneda-Hernandez G, Ferraccioli G, Isaacs JD, Kvien TK, Martin-Mola E, Mittendorf T, Smolen JS, Burmester GR (2013) The role of biosimilars in the treatment of rheumatic diseases. Ann Rheum Dis 72:322–328. doi:10.1136/annrheumdis-2012-202715
Graumann K, Premstaller A (2006) Manufacturing of recombinant therapeutic proteins in microbial systems. Biotechnol J 1:164–186. doi:10.1002/biot.200500051
Berger M, Kaup M, Blanchard V (2012) Protein glycosylation and its impact on biotechnology. Adv Biochem Eng Biotechnol 127:165–185. doi:10.1007/10_2011_101
Sarpatwari A, Avorn J, Kesselheim AS (2015) Progress and hurdles for follow-on biologics. N Engl J Med 372:2380–2382. doi:10.1056/NEJMp1504672
Nowicki M (2007) Basic facts about biosimilars. Kidney Blood Press Res 30:267–272. doi:10.1159/000105133
Lee JF, Litten JB, Grampp G (2012) Comparability and biosimilarity: considerations for the healthcare provider. Curr Med Res Opin 28:1053–1058. doi:10.1185/03007995.2012.686902
Ramanan S, Grampp G (2014) Drift, evolution, and divergence in biologics and biosimilars manufacturing. BioDrugs 28:363–372. doi:10.1007/s40259-014-0088-z
Kay J, Smolen JS (2013) Biosimilars to treat inflammatory arthritis: the challenge of proving identity. Ann Rheum Dis 72:1589–1593. doi:10.1136/annrheumdis-2012-203198
International Federation of Pharmaceutical Manufacturers & Associations (IFPMA) (2014) Policy statement. Non-comparable biotherapeutic products. IFPMA. http://www.ifpma.org/wp-content/uploads/2016/02/Non-comparable_Biotherapeutic_Products__English__02.pdf. Accessed 5 July 2016
World Health Organization (2010) Guidelines on evaluation of similar biotherapeutic products (SBPs). World Health Organization. http://www.who.int/biologicals/areas/biological_therapeutics/BIOTHERAPEUTICS_FOR_WEB_22APRIL2010.pdf. Accessed 5 July 2016
International Conference on Harmonisation (ICH), ICH Expert Working Group (2009) ICH harmonised tripartite gudieline: pharmaceutical development Q8 (R2): current Step 4 version. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q8_R1/Step4/Q8_R2_Guideline.pdf. Accessed 5 July 2016
International Conference on Harmonisation (ICH), ICH Expert Working Group (2004) ICH harmonised tripartite guideline: comparability of biotechnological/biological products subject to changes in their manufacturing process. Q5E. International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use. http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q5E/Step4/Q5E_Guideline.pdf. Accessed 5 July 2016
Declerck P, Farouk-Rezk M, Rudd PM (2015) Biosimilarity versus manufacturing change: two distinct concepts. Pharm Res. doi:10.1007/s11095-015-1790-3
US Food and Drug Adminstration, Yao L (2008) Meeting of the Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) to evaluate myozyme (alglucosidase alfa) for the treatment of late onset Pompe disease. Clinical background materials. US Food and Drug Administrtion, Center for Drug Evaluation and Research (CDER). http://www.fda.gov/ohrms/dockets/ac/08/briefing/2008-4389b1-01-FDA.pdf. Accessed 5 July 2016
McCamish M, Woollett G (2011) Worldwide experience with biosimilar development. MAbs 3:209–217
Committee for Medicinal Products for Human Use (CHMP) (2016) European public assessment reports: biosimilars. European Medicines Agency. http://www.ema.europa.eu/ema/index.jsp?curl=pages%2Fmedicines%2Flanding%2Fepar_search.jsp&mid=WC0b01ac058001d125&searchTab=searchByAuthType&alreadyLoaded=true&isNewQuery=true&status=Authorised&status=Withdrawn&status=Suspended&status=Refused&keyword=Enter+keywords&searchType=name&taxonomyPath=&treeNumber=&searchGenericType=biosimilars&genericsKeywordSearch=Submit. Accessed 5 July 2016
de Mora F (2015) Biosimilar: what it is not. Br J Clin Pharmacol 80:949–956. doi:10.1111/bcp.12656
Colwell J (2015) FDA approves first biosimilar, Zarxio. Cancer Discov 5:460. doi:10.1158/2159-8290.CD-ND2015-002
US Food and Drug Admiistration (2016) FDA approves Inflectra, a biosimilar to Remicade [news release]. US Department of Health & Human Services. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm494227.htm. Accessed 5 July 2016
Martinusen DJ, Lo C, Marin JG, Tsao NW, Leung M (2014) Potential impact of subsequent entry biologics in nephrology practice in Canada. Can J Kidney Health Dis 1:32. doi:10.1186/s40697-014-0032-7
Castañeda-Hernández G, González-Ramírez R, Kay J, Scheinberg MA (2015) Biosimilars in rheumatology: what the clinician should know. RMD Open 1:e000010
Beck A (2011) Biosimilar, biobetter and next generation therapeutic antibodies. MAbs 3:107–110
Weise M, Bielsky MC, De Smet K, Ehmann F, Ekman N, Giezen TJ, Gravanis I, Heim HK, Heinonen E, Ho K, Moreau A, Narayanan G, Kruse NA, Reichmann G, Thorpe R, van Aerts L, Vleminckx C, Wadhwa M, Schneider CK (2012) Biosimilars: what clinicians should know. Blood 120:5111–5117. doi:10.1182/blood-2012-04-425744
Declerck PJ, Farouk-Rezk M, Rudd PM (2016) Biosimilarity versus manufacturing change: two distinct concepts. Pharm Res 33:261–268. doi:10.1007/s11095-015-1790-3
Acknowledgments
Medical writing support was provided by Neel Misra, MSc, CMPP, of Engage Scientific Solutions and was funded by Pfizer. The authors would like to thank Chee-Keng Ng of Pfizer for his contributions to the manuscript. The authors also wish to thank Shivanthy Arnold, Beverly Ingram, and Judith Macdonald, all of Pfizer, for their contributions to the regulatory sections of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Authors had control of all primary data. Valderilio Azevedo has received unrestricted grants or served on speakers bureaus for AbbVie, Bristol-Myers Squibb, Janssen, Novartis, Pfizer, Roche, Celltrion, UCB, and AstraZeneca. João Eurico Fonseca has received unrestricted research grants or served on speakers bureaus for AbbVie, Amgen, Bristol-Myers Squibb, Janssen, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, Roche, Servier, and UCB. Tatsuya Atsumi has accepted honoraria for educational meetings from Mitsubishi Tanabe Pharma, Chugai Pharmaceutical Co., Astellas Pharma Inc., Takeda Pharmaceutical Co., Pfizer Inc., AbbVie Inc., and Eisai and has received a research grant from Astellas Pharma Inc., Takeda Pharmaceutical Co., Mitsubishi Tanabe Pharma Corporation, Chugai Pharmaceutical Co., Daiichi-Sankyo, Otsuka Pharmaceutical, and Pfizer Inc. Brian Hassett, Javier Coindreau, Ira Jacobs, Ehab Mahgoub, Julie O’Brien, Ena Singh, Steven Vicik, and Brian Fitzpatrick are full-time employees of Pfizer.
Funding
This report is supported by Pfizer Inc.
Rights and permissions
About this article
Cite this article
Azevedo, V., Hassett, B., Fonseca, J.E. et al. Differentiating biosimilarity and comparability in biotherapeutics. Clin Rheumatol 35, 2877–2886 (2016). https://doi.org/10.1007/s10067-016-3427-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3427-2