Abstract
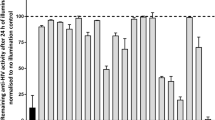
A total of 140,000 compounds were screened in a targetfree cell-based high throughput assay against HIV-1 infection, and a subset of 81 promising compounds was identified. Secondary screening of these 81 compounds revealed two putative human RNaseH2 inhibitors, RHI001 and RHI002, with IC50 value of 6.8 μM and 16 μM, respectively. RHI002 showed selective activity against human RNaseH2 while RHI001 inhibited HIV-RNaseH, E. coli RNaseH, and human RNaseH1 with IC50 value of 28.5 μM, 7.9 μM, and 31.7 μM, respectively. Kinetic analysis revealed that both inhibitors had non-competitive inhibitor-like properties. Because RNaseH2 is involved in the etiology of Aicardi-Goutier syndrome and has been suggested as an anticancer drug target, small molecule inhibitors modulating its activity would be useful for investigating the cellular function of this molecule.
Similar content being viewed by others
References
Aicardi, J., and Goutieres, F. (1984). A progressive familial encephalopathy in infancy with calcifications of the basal ganglia and chronic cerebrospinal fluid lymphocytosis. Ann. Neurol. 15, 49–54.
Chon, H., Vassilev, A., DePamphilis, M.L., Zhao, Y., Zhang, J., Burgers, P.M., Crouch, R.J., and Cerritelli, S.M. (2009). Contributions of the two accessory subunits, RNASEH2B and RNASEH2C, to the activity and properties of the human RNaseH2 complex. Nucleic Acids Res. 37, 96–110.
Copeland, R.A. (2003). Mechanistic considerations in high-throughput screening. Anal. Biochem. 320, 1–12.
Crow, Y.J., Leitch, A., Hayward, B.E., Garner, A., Parmar, R., Griffith, E., Ali, M., Semple, C., Aicardi, J., Babul-Hirji, R., et al. (2006). Mutations in genes encoding ribonuclease H2 subunits cause Aicardi-Goutieres syndrome and mimic congenital viral brain infection. Nat. Genet. 38, 910–916.
Flanagan, J.M., Funes, F.M., Henderson, S., Wild, L., Carey, N., and Boshoff, C. (2009). Genomics screen in transformed stem cells reveals RNASEH2A, PPAP2C, and ADARB1 as putative anticancer drug targets. Mol. Cancer Ther. 8, 249–260.
Gao, F., Li, Y., Decker, J.M., Peyerl, F.W., Bibollet-Ruche, F., Rodenburg, C.M., Chen, Y., Shaw, D.R., Allen, S., Musonda, R., et al. (2003). Codon usage optimization of HIV type 1 subtype C gag, pol, env, and nef genes: in vitro expression and immune responses in DNA-vaccinated mice. AIDS Res. Hum. Retroviruses 19, 817–823.
Genovesio, A., Kwon, Y.J., Windisch, M.P., Kim, N.Y., Choi, S.Y., Kim, H.C., Jung, S., Mammano, F., Perrin, V., Boese, A.S., et al. (2011). Automated genome wide profiling of cellular proteins involved in HIV infection. J. Biomol. Screen 16, 945–958.
Goutieres, F. (2005). Aicardi-Goutieres syndrome. Brain Dev. 27, 201–206.
Goutieres, F., Aicardi, J., Barth, P.G., and Lebon, P. (1998). Aicardi-Goutieres syndrome: an update and results of interferon-alpha studies. Ann. Neurol. 44, 900–907.
Hiller, B., Achleitner, M., Glage, S., Naumann, R., Behrendt, R., and Roers, A. (2012). Mammalian RNaseH2 removes ribonucleotides from DNA to maintain genome integrity. J. Exp. Med. 209, 1419–1426.
Reijns, M.A., Rabe, B., Rigby, R.E., Mill, P., Astell, K.R., Lettice, L.A., Boyle, S., Leitch, A., Keighren, M., Kilanowski, F., et al. (2012). Enzymatic removal of ribonucleotides from DNA is essential for mammalian genome integrity and development. Cell 149, 1008–1022.
Rice, G., Patrick, T., Parmar, R., Taylor, C.F., Aeby, A., Aicardi, J., Artuch, R., Montalto, S.A., Bacino, C.A., Barroso, B., et al. (2007). Clinical and molecular phenotype of Aicardi-Goutieres Syndrome. Am. J. Hum. Genet. 81, 713–725.
Sommer, P., Vartanian, J.P., Wachsmuth, M., Henry, M., Guetard, D., and Wain-Hobson, S. (2004). Anti-termination by SIV Tat requires flexibility of the nascent TAR structure. J. Mol. Biol. 344, 11–28.
Takeuchi, O., and Akira S. (2009). Innate immunity to virus infection. Immunol. Rev. 227, 75–86.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kim, J., Yoon, J., Ju, M. et al. Identification of two HIV inhibitors that also inhibit human RNaseH2. Mol Cells 36, 212–218 (2013). https://doi.org/10.1007/s10059-013-2348-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-2348-z