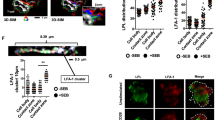
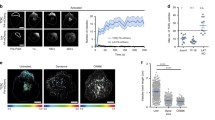
Abstract
Lck Interacting Membrane protein (LIME) was previously characterized as a transmembrane adaptor protein mediating TCR-dependent T cell activation. Here, we show that LIME associates with Vav in response to TCR stimulation and is required for Vav guanine nucleotide exchange factor (GEF) activity for Rac1. Consistent with this finding, actin polymerization at the immunological synapse (IS) was markedly enhanced by overexpression of LIME, but was reduced by expression of a LIME shRNA. Moreover, TCR-mediated cell adhesion to ICAM-1, laminin, or fibronectin was downregulated by expression of LIME shRNA. In addition, in the IS, LIME but not LAT was found to localize at the peripheral-supramolecular activation cluster (p-SMAC) where the integrins were previously shown to be localized. Together, these results establish LIME as a transmembrane adaptor protein linking TCR stimulation to IS formation and integrin activation through activation of Vav.
Similar content being viewed by others
References
Ahn, E., Lee, H., and Yun, Y. (2006). LIME acts as a transmembrane adapter mediating BCR-dependent B-cell activation. Blood 107, 1521–1527.
Ardouin, L., Bracke, M., Mathiot, A., Pagakis, S.N., Norton, T., Hogg, N., and Tybulewicz, V.L. (2003). Vav1 transduces TCR signals required for LFA-1 function and cell polarization at the immunological synapse. Eur. J. Immunol. 33, 790–797.
Benard, V., Bohl, B.P., and Bokoch, G.M. (1999). Characterization of rac and cdc42 activation in chemoattractant-stimulated human neutrophils using a novel assay for active GTPases. J. Biol. Chem. 274, 13198–13204.
Billadeau, D.D., Nolz, J.C., and Gomez, T.S. (2007). Regulation of T-cell activation by the cytoskeleton. Nat. Rev. Immunol. 7, 131–143.
Brdickova, N., Brdicka, T., Angelisova, P., Horvath, O., Spicka, J., Hilgert, I., Paces, J., Simeoni, L., Kliche, S., Merten, C., et al. (2003). LIME: a new membrane Raft-associated adaptor protein involved in CD4 and CD8 coreceptor signaling. J. Exp. Med. 198, 1453–1462.
Brummelkamp, T.R., Bernards, R., and Agami, R. (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296, 550–553.
Bunnell, S.C., Kapoor, V., Trible, R.P., Zhang, W., and Samelson, L.E. (2001). Dynamic actin polymerization drives T cell receptor-induced spreading: a role for the signal transduction adaptor LAT. Immunity 14, 315–329.
Burbach, B.J., Medeiros, R.B., Mueller, K.L., and Shimizu, Y. (2007). T-cell receptor signaling to integrins. Immunol. Rev. 218, 65–81.
Choi, Y.B., Kim, C.K., and Yun, Y. (1999). Lad, an adapter protein interacting with the SH2 domain of p56lck, is required for T cell activation. J. Immunol. 163, 5242–5249.
Crespo, P., Schuebel, K.E., Ostrom, A.A., Gutkind, J.S., and Bustelo, X.R. (1997). Phosphotyrosine-dependent activation of Rac-1 GDP/GTP exchange by the vav proto-oncogene product. Nature 385, 169–172.
Depoil, D., Fleire, S., Treanor, B.L., Weber, M., Harwood, N.E., Marchbank, K.L., Tybulewicz, V.L., and Batista, F.D. (2008). CD19 is essential for B cell activation by promoting B cell receptor-antigen microcluster formation in response to membrane-bound ligand. Nat. Immunol. 9, 63–72.
Fischer, K.D., Kong, Y.Y., Nishina, H., Tedford, K., Marengere, L.E., Kozieradzki, I., Sasaki, T., Starr, M., Chan, G., Gardener, S., et al. (1998). Vav is a regulator of cytoskeletal reorganization mediated by the T-cell receptor. Curr. Biol. 8, 554–562.
Gakidis, M.A., Cullere, X., Olson, T., Wilsbacher, J.L., Zhang, B., Moores, S.L., Ley, K., Swat, W., Mayadas, T., and Brugge, J.S. (2004). Vav GEFs are required for beta2 integrin-dependent functions of neutrophils. J. Cell Biol. 166, 273–282.
Gregoire, C., Simova, S., Wang, Y., Sansoni, A., Richelme, S., Schmidt-Giese, A., Simeoni, L., Angelisova, P., Reinhold, D., Schraven, B., et al. (2007). Deletion of the LIME adaptor protein minimally affects T and B cell development and function. Eur. J. Immunol. 37, 3259–3269.
Hashimoto-Tane, A., Yokosuka, T., Sakata-Sogawa, K., Sakuma, M., Ishihara, C., Tokunaga, M., and Saito, T. (2011). Dyneindriven transport of T cell receptor microclusters regulates immune synapse formation and T cell activation. Immunity 34, 919–931.
Horejsi, V. (2004). Transmembrane adaptor proteins in membrane microdomains: important regulators of immunoreceptor signaling. Immunol. Lett. 92, 43–49.
Hur, E.M., Son, M., Lee, O.H., Choi, Y.B., Park, C., Lee, H., and Yun, Y. (2003). LIME, a novel transmembrane adaptor protein, associates with p56lck and mediates T cell activation. J. Exp. Med. 198, 1463–1473.
Kaizuka, Y., Douglass, A.D., Varma, R., Dustin, M.L., and Vale, R.D. (2007). Mechanisms for segregating T cell receptor and adhesion molecules during immunological synapse formation in Jurkat T cells. Proc. Natl. Acad. Sci. USA 104, 20296–20301.
Kaminuma, O., Deckert, M., Elly, C., Liu, Y.C., and Altman, A. (2001). Vav-Rac1-mediated activation of the c-Jun N-terminal kinase/c-Jun/AP-1 pathway plays a major role in stimulation of the distal NFAT site in the interleukin-2 gene promoter. Mol. Cell. Biol. 21, 3126–3136.
Kikuchi, K., Kawasaki, Y., Ishii, N., Sasaki, Y., Asao, H., Takeshita, T., Miyoshi, I., Kasai, N., and Sugamura, K. (2001). Suppression of thymic development by the dominant-negative form of Gads. Int. Immunol. 13, 777–783.
Krawczyk, C., Oliveira-dos-Santos, A., Sasaki, T., Griffiths, E., Ohashi, P.S., Snapper, S., Alt, F., and Penninger, J.M. (2002). Vav1 controls integrin clustering and MHC/peptide-specific cell adhesion to antigen-presenting cells. Immunity 16, 331–343.
Lee, S.H., and Dominguez, R. (2010). Regulation of actin cytoskeleton dynamics in cells. Mol. Cells 29, 311–325.
Leo, A., Wienands, J., Baier, G., Horejsi, V., and Schraven, B. (2002). Adapters in lymphocyte signaling. J. Clin. Invest. 109, 301–309.
Monks, C.R., Freiberg, B.A., Kupfer, H., Sciaky, N., and Kupfer, A. (1998). Three-dimensional segregation of supramolecular activation clusters in T cells. Nature 395, 82–86.
Noguchi, T., Matozaki, T., Horita, K., Fujioka, Y., and Kasuga, M. (1994). Role of SH-PTP2, a protein-tyrosine phosphatase with Src homology 2 domains, in insulin-stimulated Ras activation. Mol. Cell. Biol. 14, 6674–6682.
Park, D., and Yun, Y. (2001). Tyrosine phosphorylation-dependent yeast two-hybrid system for the identification of the SH2 domain-binding proteins. Mol. Cells 12, 244–249.
Perez-Villar, J.J., Whitney, G.S., Sitnick, M.T., Dunn, R.J., Venkatesan, S., O’Day, K., Schieven, G.L., Lin, T.A., and Kanner, S.B. (2002). Phosphorylation of the linker for activation of T-cells by Itk promotes recruitment of Vav. Biochemistry 41, 10732–10740.
Saito, T., and Yokosuka, T. (2006). Immunological synapse and microclusters: the site for recognition and activation of T cells. Curr. Opin. Immunol. 18, 305–313.
Tuosto, L., Michel, F., and Acuto, O. (1996). p95vav associates with tyrosine-phosphorylated SLP-76 in antigen-stimulated T cells. J. Exp. Med. 184, 1161–1166.
Varma, R., Campi, G., Yokosuka, T., Saito, T., and Dustin, M.L. (2006). T cell receptor-proximal signals are sustained in peripheral microclusters and terminated in the central supramolecular activation cluster. Immunity 25, 117–127.
Wange, R.L. (2000). LAT, the linker for activation of T cells: a bridge between T cell-specific and general signaling pathways. Sci. STKE 2000, RE1.
Yokosuka, T., Sakata-Sogawa, K., Kobayashi, W., Hiroshima, M., Hashimoto-Tane, A., Tokunaga, M., Dustin, M.L., and Saito, T. (2005). Newly generated T cell receptor microclusters initiate and sustain T cell activation by recruitment of Zap70 and SLP-76. Nat. Immunol. 6, 1253–1262.
Yokosuka, T., Kobayashi, W., Sakata-Sogawa, K., Takamatsu, M., Hashimoto-Tane, A., Dustin, M.L., Tokunaga, M., and Saito, T. (2008). Spatiotemporal regulation of T cell costimulation by TCR-CD28 microclusters and protein kinase C theta translocation. Immunity 29, 589–601.
Yu, C.H., Wu, H.J., Kaizuka, Y., Vale, R.D., and Groves, J.T. (2010). Altered actin centripetal retrograde flow in physically restricted immunological synapses. PLoS One 5, e11878.
Zeng, R., Cannon, J.L., Abraham, R.T., Way, M., Billadeau, D.D., Bubeck-Wardenberg, J., and Burkhardt, J.K. (2003). SLP-76 coordinates Nck-dependent Wiskott-Aldrich syndrome protein recruitment with Vav-1/Cdc42-dependent Wiskott-Aldrich syndrome protein activation at the T cell-APC contact site. J. Immunol. 171, 1360–1368.
Zhang, W., Sloan-Lancaster, J., Kitchen, J., Trible, R.P., and Samelson, L.E. (1998). LAT: the ZAP-70 tyrosine kinase substrate that links T cell receptor to cellular activation. Cell 92, 83–92.
Zhang, W., Sommers, C.L., Burshtyn, D.N., Stebbins, C.C., DeJarnette, J.B., Trible, R.P., Grinberg, A., Tsay, H.C., Jacobs, H.M., Kessler, C.M., et al. (1999). Essential role of LAT in T cell development. Immunity 10, 323–332.
Zhang, W., Trible, R.P., Zhu, M., Liu, S.K., McGlade, C.J., and Samelson, L.E. (2000). Association of Grb2, Gads, and phospholipase C-gamma 1 with phosphorylated LAT tyrosine residues. Effect of LAT tyrosine mutations on T cell angigen receptor-mediated signaling. J. Biol. Chem. 275, 23355–23361.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Son, M., Park, I., Lee, OH. et al. LIME mediates immunological synapse formation through activation of VAV. Mol Cells 33, 407–414 (2012). https://doi.org/10.1007/s10059-012-0011-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-012-0011-8