Abstract
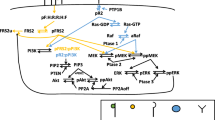
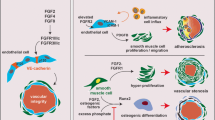
Basic fibroblast growth factor (bFGF) plays an important role in angiogenesis. However, the underlying mechanisms are not clear. Mg2+ is the most abundant intracellular divalent cation in the body and plays critical roles in many cell functions. We investigated the effect of bFGF on the intracellular Mg2+ concentration ([Mg2+]i) in human umbilical vein endothelial cells (HUVECs). bFGF increased [Mg2+]i in a dose-dependent manner, independent of extracellular Mg2+. This bFGF-induced [Mg2+]i increase was blocked by tyrosine kinase inhibitors (tyrphostin A-23 and genistein), phosphatidylinositol 3-kinase (PI3K) inhibitors (wortmannin and LY294002) and a phospholipase Cγ (PLCγ) inhibitor (U73122). In contrast, mitogen-activated protein kinase inhibitors (SB202190 and PD98059) did not affect the bFGF-induced [Mg2+]i increase. These results suggest that bFGF increases the [Mg2+]i from the intracellular Mg2+ stores through the tyrosine kinase/PI3K/PLCγ-dependent signaling pathways.
Similar content being viewed by others
References
Bussolino, F., Mantovani, A., and Persico, G. (1997). Molecular mechanisms of blood vessel formation. Trends Biochem. Sci. 22, 251–256.
Cines, D.B., Pollak, E.S., Buck, C.A., Loscalzo, J., Zimmerman, G.A., McEver, R.P., Pober, J.S., Wick, T.M., Konkle, B.A., Schwartz, B.S., et al. (1998). Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 91, 3527–3561.
Folkman, J., and Shing, Y. (1992). Angiogenesis. J. Biol. Chem. 267, 10931–10934.
Giavazzi, R., Giuliani, R., Coltrini, D., Bani, M.R., Ferri, C., Sennino, B., Tosatti, M.P., Stoppacciaro, A., and Presta, M. (2001). Modulation of tumor angiogenesis by conditional expression of fibroblast growth factor-2 affects early but not established tumors. Cancer Res. 61, 309–317.
Gunther, T. (1986). Functional compartmentation of intracellular magnesium. Magnesium 5, 53–59.
Hackett, S.F., Friedman, Z., and Campochiaro, P.A. (1987). Cyclic 3′,5′-adenosine monophosphate modulates vascular endothelial cell migration in vitro. Cell Biol. Int. Rep. 11, 279–287.
Hong, B.Z., Kang, H.S., So, J.N., Kim, H.N., Park, S.A., Kim, S.J., Kim, K.R., and Kwak, Y.G. (2006). Vascular endothelial growth factor increases the intracellular magnesium. Biochem. Biophys. Res. Commun. 347, 496–501.
Ishijima, S., and Tatibana, M. (1994). Rapid mobilization of intracellular Mg2+ by bombesin in Swiss 3T3 cells: mobilization through external Ca(2+)- and tyrosine kinase-dependent mechanisms. J. Biochem. 115, 730–737.
Kim, S.J., Kang, H.S., Kang, M.S., Yu, X., Park, S.Y., Kim, I.S., Kim, N.S., Kim, S.Z., Kwak, Y.G., and Kim, J.S. (2005). alpha(1)-Agonists-induced Mg(2+) efflux is related to MAP kinase activation in the heart. Biochem. Biophys. Res. Commun. 333, 1132–1138.
Klint, P., and Claesson-Welsh, L. (1999). Signal transduction by fibroblast growth factor receptors. Front Biosci. 4, D165–177.
Kurosawa, M., Ishizuka, T., and Shimizu, Y. (1993). Formation of phosphatidylinositol-4-phosphate in human peripheral blood eosinophils. Clin. Exp. Allergy 23, 770–776.
Laham, R.J., Chronos, N.A., Pike, M., Leimbach, M.E., Udelson, J.E., Pearlman, J.D., Pettigrew, R.I., Whitehouse, M.J., Yoshizawa, C., and Simons, M. (2000). Intracoronary basic fibroblast growth factor (FGF-2) in patients with severe ischemic heart disease: results of a phase I open-label dose escalation study. J. Am. Coll. Cardiol. 36, 2132–2139.
Lapidos, K.A., Woodhouse, E.C., Kohn, E.C., and Masiero, L. (2001). Mg(++)-induced endothelial cell migration: substratum selectivity and receptor-involvement. Angiogenesis 4, 21–28.
Lazarous, D.F., Unger, E.F., Epstein, S.E., Stine, A., Arevalo, J.L., Chew, E.Y., and Quyyumi, A.A. (2000). Basic fibroblast growth factor in patients with intermittent claudication: results of a phase I trial. J. Am. Coll. Cardiol. 36, 1239–1244.
Liao, F., Folsom, A.R., and Brancati, F.L. (1998). Is low magnesium concentration a risk factor for coronary heart disease? The Atherosclerosis Risk in Communities (ARIC) Study. Am. Heart J. 136, 480–490.
Maier, J.A., Bernardini, D., Rayssiguier, Y., and Mazur, A. (2004)a. High concentrations of magnesium modulate vascular endothelial cell behaviour in vitro. Biochim. Biophys. Acta 1689, 6–12.
Maier, J.A., Malpuech-Brugere, C., Zimowska, W., Rayssiguier, Y., and Mazur, A. (2004b). Low magnesium promotes endothelial cell dysfunction: implications for atherosclerosis, inflammation and thrombosis. Biochim. Biophys. Acta 1689, 13–21.
Mussoni, L., Sironi, L., Tedeschi, L., Calvio, A.M., Colli, S., and Tremoli, E. (2001). Magnesium inhibits arterial thrombi after vascular injury in rat: in vitro impairment of coagulation. Thromb. Haemost. 86, 1292–1295.
Ornitz, D.M., and Itoh, N. (2001). Fibroblast growth factors. Genome Biol. 2, REVIEWS3005.
Peacock, J.M., Folsom, A.R., Arnett, D.K., Eckfeldt, J.H., and Szklo, M. (1999). Relationship of serum and dietary magnesium to incident hypertension: the atherosclerosis risk in communities (ARIC) study. Ann. Epidemiol. 9, 159–165.
Powers, C.J., McLeskey, S.W., and Wellstein, A. (2000). Fibroblast growth factors, their receptors and signaling. Endocr. Relat. Cancer 7, 165–197.
Quamme, G.A., and Rabkin, S.W. (1990). Cytosolic free magnesium in cardiac myocytes: identification of a Mg2+ influx pathway. Biochem. Biophys. Res. Commun. 167, 1406–1412.
Raju, B., Murphy, E., Levy, L.A., Hall, R.D., and London, R.E. (1989). A fluorescent indicator for measuring cytosolic free magnesium. Am. J. Physiol. 256, C540–548.
Romani, A.M., and Scarpa, A. (2000). Regulation of cellular magnesium. Front Biosci. 5, D720–734.
Romani, A., Marfella, C., and Scarpa, A. (1992). Regulation of Mg2+ uptake in isolated rat myocytes and hepatocytes by protein kinase C. FEBS Lett. 296, 135–140.
Romani, A., Marfella, C., and Scarpa, A. (1993). Cell magnesium transport and homeostasis: role of intracellular compartments. Miner. Electrolyte Metab. 19, 282–289.
Scarpa, A., and Brinley, F.J. (1981). In situ measurements of free cytosolic magnesium ions. Fed. Proc. 40, 2646–2652.
Schlessinger, J. (2000). Cell signaling by receptor tyrosine kinases. Cell 103, 211–225.
Schweigel, M., Park, H.S., Etschmann, B., and Martens, H. (2006). Characterization of the Na+-dependent Mg2+ transport in sheep ruminal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G56–65.
Shi, B., Heavner, J.E., Boylan, L.M., Wang, M.J., and Spallholz, J.E. (1995). Dietary magnesium deficiency increases Gi alpha levels in the rat heart after myocardial infarction. Cardiovasc. Res. 30, 923–929.
Tashiro, M., Tursun, P., and Konishi, M. (2005). Intracellular and extracellular concentrations of Na+ modulate Mg2+ transport in rat ventricular myocytes. Biophys. J. 89, 3235–3247.
Watanabe, J., Nakayama, S., Matsubara, T., and Hotta, N. (1998). Regulation of intracellular free Mg2+ concentration in isolated rat hearts via beta-adrenergic and muscarinic receptors. J. Mol. Cell Cardiol. 30, 2307–2318.
Webber, C.A., Chen, Y.Y., Hehr, C. L., Johnston, J., and McFarlane, S. (2005). Multiple signaling pathways regulate FGF-2-induced retinal ganglion cell neurite extension and growth cone guidance. Mol. Cell. Neurosci. 30, 37–47.
Wolf, F.I., and Cittadini, A. (1999). Magne sium in cell proliferation and differentiation. Front Biosci. 4, D607–617.
Wolf, F.I., and Cittadini, A. (2003). Chemistry and biochemistry of magnesium. Mol. Aspects Med. 24, 3–9.
Wolf, F.I., Torsello, A., Fasanella, S., and Cittadini, A. (2003). Cell physiology of magnesium. Mol. Aspects Med. 24, 11–26.
Zhang, G.H., and Melvin, J.E. (1996). Na+-dependent release of Mg2+ from an intracellular pool in rat sublingual mucous acini. J. Biol. Chem. 271, 29067–29072.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hong, BZ., Park, SA., Kim, HN. et al. Basic fibroblast growth factor increases intracellular magnesium concentration through the specific signaling pathways. Mol Cells 28, 13–17 (2009). https://doi.org/10.1007/s10059-009-0103-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0103-2