Abstract
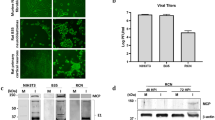
Gammaherpesvirus infection of the central nervous system (CNS) has been linked to various neurological diseases, including meningitis, encephalitis, and multiple sclerosis. However, little is known about the interactions between the virus and the CNS in vitro or in vivo. Murine gammaherpesvirus 68 (MHV-68 or γHV-68) is genetically related and biologically similar to human gammaherpesviruses, thereby providing a tractable animal model system in which to study both viral pathogenesis and replication. In the present study, we show the successful infection of cultured neuronal cells, microglia, and astrocytes with MHV-68 to various extents. Upon intracerebroventricular injection of a recombinant virus (MHV-68/LacZ) into 4–5-week-old and 9–10-week-old mice, the 4–5-week-old mice displayed high mortality within 5–7 days, while the majority of the 9–10-week-old mice survived until the end of the experimental period. Until a peak at 3–4 days post-infection, viral DNA replication and gene expression were similar in the brains of both mouse groups, but only the 9–10-week-old mice were able to subdue viral DNA replication and gene expression after 5 days post-infection. Pro-inflammatory cytokine mRNAs of tumor necrosis factor-α, interleukin 1β, and interleukin 6 were highly induced in the brains of the 4–5-week-old mice, suggesting their possible contributions as neurotoxic factors in the agedependent control of MHV-68 replication of the CNS.
Similar content being viewed by others
References
Ascherio, A., and Munch, M. (2000). Epstein-Barr virus and multiple sclerosis. Epidemiology 11, 220–224.
Ascherio, A., Munger, K.L., Lennette, E.T., Spiegelman, D., Hernan, M.A., Olek, M.J., Hankinson, S.E., and Hunter, D.J. (2001). Epstein-Barr virus antibodies and risk of multiple sclerosis: a prospective study. JAMA 286, 3083–3088.
Blaskovic, D., Stancekova, M., Svobodova, J., and Mistrikova, J. (1980). Isolation of five strains of herpesviruses from two species of free living small rodents. Acta Virol. 24, 468.
Blaskovic, D., Stanekova, D., and Rajcani, J. (1984). Experimental pathogenesis of murine herpesvirus in newborn mice. Acta Virol. 28, 225–231.
Bossolasco, S., Falk, K.I., Ponzoni, M., Ceserani, N., Crippa, F., Lazzarin, A., Linde, A., and Cinque, P. (2006). Ganciclovir is associated with low or undetectable Epstein-Barr virus DNA load in cerebrospinal fluid of patients with HIV-related primary central nervous system lymphoma. Clin. Infect. Dis. 42, e21–e25.
Brabers, N.A., and Nottet, H.S. (2006). Role of the pro-inflammatory cytokines TNF-alpha and IL-1beta in HIV-associated dementia. Eur. J. Clin. Invest. 36, 447–458.
Chan, P.K., Ng, H.K., Hui, M., Ip, M., Cheung, J.L., and Cheng, A.F. (1999). Presence of human herpesviruses 6, 7, and 8 DNA sequences in normal brain tissue. J. Med. Virol. 59, 491–495.
Chan, P.K., Ng, H.K., Cheung, J.L., and Cheng, A.F. (2000). Survey for the presence and distribution of human herpesvirus 8 in healthy brain. J. Clin. Microbiol. 38, 2772–2773.
Corbellino, M., Poirel, L., Bestetti, G., Pizzuto, M., Aubin, J.T., Capra, M., Bifulco, C., Berti, E., Agut, H., Rizzardini, G., et al. (1996). Restricted tissue distribution of extralesional Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS patients with Kaposi’s sarcoma. AIDS Res. Hum. Retroviruses 12, 651–657.
Hoover, S.E., Ross, J.P., and Cohen, J.I. (2004). Epstein-Barr virus, In Infections of the central nervous system. W.M. Scheld, R.J. Whitley, and C.M. Marra, eds. (Philadelphia: Lippincott Williams & Wilkins), pp. 175–183.
Karatas, H., Gurer, G., Pinar, A., Soylemezoglu, F., Tezel, G.G., Hascelik, G., Akalan, N., Tuncer, S., Ciger, A., and Saygi, S. (2008). Investigation of HSV-1, HSV-2, CMV, HHV-6 and HHV-8 DNA by real-time PCR in surgical resection materials of epilepsy patients with mesial temporal lobe sclerosis. J. Neurol. Sci. 264, 151–156.
Kwak, S.E., Kim, J.E., Kim, D.S., Jung, J.Y., Won, M.H., Kwon, O.S., Choi, S.Y., and Kang, T.C. (2005). Effects of GABAergic transmissions on the immunoreactivities of calcium binding proteins in the gerbil hippocampus. J. Comp. Neurol. 485, 153–164.
Kwon, H.J., Lee, K.W., Yu, S.H., Han, J.H., and Kim, D.S. (2003). NF-kappaB-dependent regulation of tumor necrosis factor-alpha gene expression by CpG-oligodeoxynucleotides. Biochem. Biophys. Res. Commun. 311, 129–138.
Lee, S., Cho, H.J., Park, J.J., Kim, Y.S., Hwang, S., Sun, R., and Song, M.J. (2007a). The ORF49 protein of murine gammaherpesvirus 68 cooperates with RTA in regulating virus replication. J. Virol. 81, 9870–9877.
Lee, J.Y., Kim, J.Y., Lee, Y.G., Shin, W.C., Chun, T., Rhee, M.H., and Cho, J.Y. (2007b). Hydroquinone, a reactive metabolite of benzene, reduces macrophage-mediated immune responses. Mol. Cells. 23, 198–206.
Lokensgard, J.R., Hu, S., Sheng, W., vanOijen, M., Cox, D., Cheeran, M.C., and Peterson, P.K. (2001). Robust expression of TNF-alpha, IL-1beta, RANTES, and IP-10 by human microglial cells during nonproductive infection with herpes simplex virus. J. Neurovirol. 7, 208–219.
Rajcani, J., Blaskovic, D., Svobodova, J., Ciampor, F., Huckova, D., and Stanekova, D. (1985). Pathogenesis of acute and persistent murine herpesvirus infection in mice. Acta Virol. 29, 51–60.
Said, J.W., Tasaka, T., de Vos, S., and Koeffler, H.P. (1997). Kaposi’s sarcoma-associated herpesvirus/human herpesvirus type 8 encephalitis in HIV-positive and -negative individuals. AIDS 11, 1119–1122.
Serafini, B., Rosicarelli, B., Franciotta, D., Magliozzi, R., Reynolds, R., Cinque, P., Andreoni, L., Trivedi, P., Salvetti, M., Faggioni, A., et al. (2007). Dysregulated Epstein-Barr virus infection in the multiple sclerosis brain. J. Exp. Med. 204, 2899–2912.
Sergerie, Y., Rivest, S., and Boivin, G. (2007). Tumor necrosis factor-alpha and interleukin-1 beta play a critical role in the resistance against lethal herpes simplex virus encephalitis. J. Infect. Dis. 196, 853–860.
Simas, J.P., and Efstathiou, S. (1998). Murine gammaherpesvirus 68: a model for the study of gammaherpesvirus pathogenesis. Trends Microbiol 6, 276–282.
Song, M.J., Hwang, S., Wong, W., Round, J., Martinez-Guzman, D., Turpaz, Y., Liang, J., Wong, B., Johnson, R.C., Carey, M., et al. (2004). The DNA architectural protein HMGB1 facilitates RTAmediated viral gene expression in gamma-2 herpesviruses. J. Virol. 78, 12940–12950.
Song, M.J., Hwang, S., Wong, W.H., Wu, T.T., Lee, S., Liao, H.I., and Sun, R. (2005). Identification of viral genes essential for replication of murine gamma-herpesvirus 68 using signaturetagged mutagenesis. Proc. Natl. Acad. Sci. USA 102, 3805–3810.
Stewart, J.P., Usherwood, E.J., Ross, A., Dyson, H., and Nash, T. (1998). Lung epithelial cells are a major site of murine gammaherpesvirus persistence. J. Exp. Med. 187, 1941–1951.
Sunil-Chandra, N.P., Efstathiou, S., Arno, J., and Nash, A.A. (1992a). Virological and pathological features of mice infected with murine gamma-herpesvirus 68. J. Gen. Virol. 73, 2347–2356.
Sunil-Chandra, N.P., Efstathiou, S., and Nash, A.A. (1992b). Murine gammaherpesvirus 68 establishes a latent infection in mouse B lymphocytes in vivo. J. Gen. Virol. 73, 3275–3279.
Sunil-Chandra, N.P., Arno, J., Fazakerley, J., and Nash, A.A. (1994). Lymphoproliferative disease in mice infected with murine gammaherpesvirus 68. Am. J. Pathol. 145, 818–826.
Taylor, W.R., Rasley, A., Bost, K.L., and Marriott, I. (2003). Murine gammaherpesvirus-68 infects microglia and induces high levels of pro-inflammatory cytokine production. J. Neuroimmunol. 136, 75–83.
Terry, L.A., Stewart, J.P., Nash, A.A., and Fazakerley, J.K. (2000). Murine gammaherpesvirus-68 infection of and persistence in the central nervous system. J. Gen. Virol. 81, 2635–2643.
Weck, K.E., Kim, S.S., Virgin, H.I., and Speck, S.H. (1999). Macrophages are the major reservoir of latent murine gammaherpesvirus 68 in peritoneal cells. J. Virol. 73, 3273–3283.
Weinberg, A., Bloch, K.C., Li, S., Tang, Y.W., Palmer, M., and Tyler, K.L. (2005). Dual infections of the central nervous system with Epstein-Barr virus. J. Infect. Dis. 191, 234–237.
Wu, T.T., Tong, L., Rickabaugh, T., Speck, S., and Sun, R. (2001). Function of Rta is essential for lytic replication of murine gammaherpesvirus 68. J. Virol. 75, 9262–9273.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Cho, HJ., Kim, S., Kwak, SE. et al. Age-dependent pathogenesis of murine gammaherpesvirus 68 infection of the central nervous system. Mol Cells 27, 105–111 (2009). https://doi.org/10.1007/s10059-009-0011-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-009-0011-5