Abstract
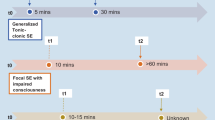
The terms developmental epileptic encephalopathy with spike-and-wave activation in sleep (DEE-SWAS) and epileptic encephalopathy with spike-and-wave activation in sleep (EE-SWAS) designate a spectrum of conditions that are typified by different combinations of motor, cognitive, language, and behavioral regression linked to robust spike-and-wave activity during sleep. In this study, we aimed at describing the clinical and molecular findings in “(developmental) epileptic encephalopathy with spike-and-wave activation in sleep” (D)EE-SWAS) patients as well as at contributing to the genetic etiologic spectrum of (D)EE-SWAS. Single nucleotide polymorphism (SNP) array and whole-exome sequencing (WES) techniques were used to determine the underlying genetic etiologies. Of the 24 patients included in the study, 8 (33%) were female and 16 (67%) were male. The median age at onset of the first seizure was 4 years and the median age at diagnosis of (D)EE-SWAS was 5 years. Of the 24 cases included in the study, 13 were compatible with the clinical diagnosis of DEE-SWAS and 11 were compatible with the clinical diagnosis of EE-SWAS. Abnormal perinatal history was present in four cases (17%), and two cases (8%) had a family history of epilepsy. Approximately two-thirds (63%) of all patients had abnormalities detected on brain computerized tomography/magnetic resonance (CT/MR) imaging. After SNP array and WES analysis, the genetic etiology was revealed in 7 out of 24 (29%) cases. Three of the variants detected were novel (SLC12A5, DLG4, SLC9A6). This study revealed for the first time that Smith-Magenis syndrome, SCN8A-related DEE type 13 and SLC12A5 gene variation are involved in the genetic etiology of (D)EE-SWAS. (D)EE-SWAS is a genetically diverse disorder with underlying copy number variations and single-gene abnormalities. In the current investigation, rare novel variations in genes known to be related to (D)EE-SWAS and not previously reported genes to be related to (D)EE-SWAS were discovered, adding to the molecular genetic spectrum. Molecular etiology enables the patient and family to receive thorough and accurate genetic counseling as well as a personalized medicine approach.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Rubboli G, Gardella E, Cantalupo G, Alberto Tassinari C (2023) Encephalopathy related to status epilepticus during slow sleep (ESES). Pathophysiological insights and nosological considerations. Epilepsy Behav 140:109105. https://doi.org/10.1016/j.yebeh.2023.109105
Specchio N, Wirrell EC, Scheffer IE, Nabbout R, Riney K, Samia P et al (2022) International League Against Epilepsy classification and definition of epilepsy syndromes with onset in childhood: Position paper by the ILAE Task Force on Nosology and Definitions. Epilepsia 63:1398–1442. https://doi.org/10.1111/epi.17241
Fernández IS, Chapman KE, Peters JM, Kothare SV, Nordli DR Jr, Jensen FE et al (2013) The tower of Babel: survey on concepts and terminology in electrical status epilepticus in sleep and continuous spikes and waves during sleep in North America. Epilepsia 54:741–750. https://doi.org/10.1111/epi.12039
Loddenkemper T, Fernández IS, Peters JM (2011) Continuous spike and waves during sleep and electrical status epilepticus in sleep. J Clin Neurophysiol 28:154–164. https://doi.org/10.1097/WNP.0b013e31821213eb
Tassinari CA, Rubboli G, Volpi L, Meletti S, d’Orsi G, Franca M et al (2000) Encephalopathy with electrical status epilepticus during slow sleep or ESES syndrome including the acquired aphasia. Clin Neurophysiol 111(Suppl 2):S94–S102. https://doi.org/10.1016/s1388-2457(00)00408-9
Arican P, Gencpinar P, OlgacDundar N, Tekgul H (2021) Electrical Status Epilepticus During Slow-wave Sleep (ESES): Current Perspectives. J Pediatr Neurosci 16:91–96. https://doi.org/10.4103/jpn.JPN_137_20
Galanopoulou AS, Bojko A, Lado F, Moshé SL (2000) The spectrum of neuropsychiatric abnormalities associated with electrical status epilepticus in sleep. Brain Dev 22:279–295. https://doi.org/10.1016/s0387-7604(00)00127-3
Eriksson MH, Baldeweg T, Pressler R, Boyd SG, Huber R, Cross JH et al (2023) Sleep homeostasis, seizures, and cognition in children with focal epilepsy. Dev Med Child Neurol 65:701–711. https://doi.org/10.1111/dmcn.15403
Monteiro JP, Roulet-Perez E, Davidoff V, Deonna T (2021) Primary neonatal thalamic haemorrhage and epilepsy with continuous spike-wave during sleep: a longitudinal follow-up of a possible significant relation. Eur J Paediatr Neurol 5:41–47. https://doi.org/10.1053/ejpn.2001.0403
Rubboli G, Huber R, Tononi G, Tassinari CA (2019) Encephalopathy related to Status Epilepticus during slow Sleep: a link with sleep homeostasis? Epileptic Disord 21:62–70. https://doi.org/10.1684/epd.2019.1059
Sánchez Fernández I, Takeoka M, Tas E, Peters JM, Prabhu SP, Stannard KM et al (2012) Early thalamic lesions in patients with sleep-potentiated epileptiform activity. Neurology 78:1721–1727. https://doi.org/10.1212/WNL.0b013e3182582ff8
Sánchez Fernández I, Loddenkemper T, Peters JM, Kothare SV (2012) Electrical status epilepticus in sleep: clinical presentation and pathophysiology. Pediatr Neurol 47:390–410. https://doi.org/10.1016/j.pediatrneurol.2012.06.016
van den Munckhof B, de Vries EE, Braun KP, Boss HM, Willemsen MA et al (2016) Serum inflammatory mediators correlate with disease activity in electrical status epilepticus in sleep (ESES) syndrome. Epilepsia 57:e45-50. https://doi.org/10.1111/epi.13274
Caraballo RH, Veggiotti P, Kaltenmeier MC, Piazza E, Gamboni B, Lopez Avaria MF et al (2013) Encephalopathy with status epilepticus during sleep or continuous spikes and waves during slow sleep syndrome: a multicenter, long-term follow-up study of 117 patients. Epilepsy Res 105:164–173. https://doi.org/10.1016/j.eplepsyres.2013.02.010
Gong P, Xue J, Jiao X, Zhang Y, Yang Z (2021) Genetic Etiologies in Developmental and/or Epileptic Encephalopathy With Electrical Status Epilepticus During Sleep: Cohort Study. Front Genet 12:607965. https://doi.org/10.3389/fgene.2021.607965
Kessi M, Peng J, Yang L, Xiong J, Duan H, Pang N et al (2018) Genetic etiologies of the electrical status epilepticus during slow wave sleep: systematic review. BMC Genet 19:40. https://doi.org/10.1186/s12863-018-0628-5
Alsini H, Alghamdi A, Alshafi S, Hundallah K, Almehmadi S, Alsowat D et al (2023) Developmental and/or epileptic encephalopathy with spike-and-wave activation in sleep in Saudi Arabia: Electroclinical, etiologic, genetic, and outcome multicenter study. Seizure 107:146–154. https://doi.org/10.1016/j.seizure.2023.04.013
Sonnek B, Döring JH, Mütze U, Schubert-Bast S, Bast T, Balke D et al (2021) Clinical spectrum and treatment outcome of 95 children with continuous spikes and waves during sleep (CSWS). Eur J Paediatr Neurol 30:121–127. https://doi.org/10.1016/j.ejpn.2020.10.010
Pavlidis E, Møller RS, Nikanorova M, Kölmel MS, Stendevad P, Beniczky S et al (2019) Idiopathic encephalopathy related to status epilepticus during slow sleep (ESES) as a “pure” model of epileptic encephalopathy. An electroclinical, genetic, and follow-up study. Epilepsy Behav 97:244–252. https://doi.org/10.1016/j.yebeh.2019.05.030
Kalia LV, Kalia SK, Salter MW (2008) NMDA receptors in clinical neurology: excitatory times ahead. Lancet Neurol 7:742–755. https://doi.org/10.1016/S1474-4422(08)70165-0
Bonanni P, Negrin S, Volzone A, Zanotta N, Epifanio R, Zucca C et al (2017) Electrical status epilepticus during sleep in Mowat-Wilson syndrome. Brain Dev 39:727–734. https://doi.org/10.1016/j.braindev.2017.04.013
Zanni G, Barresi S, Cohen R, Specchio N, Basel-Vanagaite L, Valente EM et al (2014) A novel mutation in the endosomal Na+/H+ exchanger NHE6 (SLC9A6) causes Christianson syndrome with electrical status epilepticus during slow-wave sleep (ESES). Epilepsy Res 108:811–815. https://doi.org/10.1016/j.eplepsyres.2014.02.009
Ergun-Longmire B, Nguyen MHN, Com G (2022) Electrical status epilepticus during sleep in a child with Prader-Willi syndrome: a case report. AME Case Rep 6:7. https://doi.org/10.21037/acr-21-34
Öktem F, Gençöz T, Erden G, Sezgin N, Uluç S (2013) Turkish version of Wechsler Intelligence Scale for Children-IV (WISC-IV) application and scoring handbook. Ankara Turkish Psychological Association Publications
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J et al (2015) ACMG Laboratory Quality Assurance Committee. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424. https://doi.org/10.1038/gim.2015.30
Fan X, Huang J, Jin X, Yan N (2023) Cryo-EM structure of human voltage-gated sodium channel Nav1.6. Proc Natl Acad Sci U S A 120:e2220578120. https://doi.org/10.1073/pnas.2220578120
Chi X, Li X, Chen Y, Zhang Y, Su Q, Zhou Q (2021) Cryo-EM structures of the full-length human KCC2 and KCC3 cation-chloride cotransporters. Cell Res 31:482–484. https://doi.org/10.1038/s41422-020-00437-x
Lomize MA, Pogozheva ID, Joo H, Mosberg HI, Lomize AL (2012) OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res 40:D370–D376. https://doi.org/10.1093/nar/gkr703
Floden EW, Tommaso PD, Chatzou M, Magis C, Notredame C, Chang JM (2016) PSI/TM-Coffee: a web server for fast and accurate multiple sequence alignments of regular and transmembrane proteins using homology extension on reduced databases. Nucleic Acids Res 44:W339–W343. https://doi.org/10.1093/nar/gkw300
Robert X, Gouet P (2014) Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res 42:W320–W324. https://doi.org/10.1093/nar/gku316
Kulandaisamy A, Zaucha J, Sakthivel R, Frishman D, Michael Gromiha M (2020) Pred-MutHTP: Prediction of disease-causing and neutral mutations in human transmembrane proteins. Hum Mutat 41:581–590. https://doi.org/10.1002/humu.23961
Pires DEV, Rodrigues CHM, Ascher DB (2020) mCSM-membrane: predicting the effects of mutations on transmembrane proteins. Nucleic Acids Res 48(W1):W147–W153. https://doi.org/10.1093/nar/gkaa416
Ittisoponpisan S, Islam SA, Khanna T, Alhuzimi E, David A, Sternberg MJE (2019) Can Predicted Protein 3D Structures Provide Reliable Insights into whether Missense Variants Are Disease Associated? J Mol Biol 431:2197–2212. https://doi.org/10.1016/j.jmb.2019.04.009
Sager G, Takis G, Vatansever Pinar Z, Duzkalir H, Turkyilmaz A, Çağ Y et al (2023) Evaluation of long-term neurocognitive functions in patients with epileptic encephalopathy with continuous spike-and-wave during sleep (CSWS)/epileptic encephalopathy with spike-and-wave activation in sleep (EE-SWAS). Neurophysiol Clin 53:102861. https://doi.org/10.1016/j.neucli.2023.102861
Sager SG, Turkyilmaz A, Gunbey HP, Karatoprak EY, Aslan ES, Akın Y (2023) A novel de novo TET3 loss-of-function variant in a Turkish boy presenting with neurodevelopmental delay and electrical status epilepticus during slow-wave sleep. Brain Dev 45:140–145. https://doi.org/10.1016/j.braindev.2022.09.004
Xie Y, Chang S, Zhao C et al (2020) Structures and an activation mechanism of human potassium-chloride cotransporters. Sci Adv 6:eabc5883. https://doi.org/10.1126/sciadv.abc5883
Stödberg T, McTague A, Ruiz AJ et al (2015) Mutations in SLC12A5 in epilepsy of infancy with migrating focal seizures. Nat Commun 6:8038. https://doi.org/10.1038/ncomms9038
Puskarjov M, Seja P, Heron SE, Williams TC, Ahmad F, Iona X et al (2014) A variant of KCC2 from patients with febrile seizures impairs neuronal Cl- extrusion and dendritic spine formation. EMBO Rep 15:723–729. https://doi.org/10.1002/embr.201438749
Tang BL (2020) The Expanding Therapeutic Potential of Neuronal KCC2. Cells 9:240. https://doi.org/10.3390/cells9010240
Moutton S, Bruel AL, Assoum M, Chevarin M, Sarrazin E, Goizet C et al (2018) Truncating variants of the DLG4 gene are responsible for intellectual disability with marfanoid features. Clin Genet 93:1172–1178. https://doi.org/10.1111/cge.13243
Rodríguez-Palmero A, Boerrigter MM, Gómez-Andrés D, Aldinger KA, Marcos-Alcalde Í, Popp B et al (2021) DLG4-related synaptopathy: a new rare brain disorder. Genet Med 23:888–899. https://doi.org/10.1038/s41436-020-01075-9
Kassabian B, Levy AM, Gardella E, Aledo-Serrano A, Ananth AL, Brea-Fernández et al (2023) Developmental epileptic encephalopathy in DLG4-related synaptopathy. Epilepsia https://doi.org/10.1111/epi.17876. Advance online publication. https://doi.org/10.1111/epi.17876
Goldman AM, Potocki L, Walz K, Lynch JK, Glaze DG, Lupski JR et al (2006) Epilepsy and chromosomal rearrangements in Smith-Magenis Syndrome [del(17)(p11.2p11.2)]. J Child Neurol 21:93–98. https://doi.org/10.1177/08830738060210021201
Rive Le Gouard N, Jacquinet A, Ruaud L, Deleersnyder H, Ageorges F, Gallard J et al (2021) Smith-Magenis syndrome: Clinical and behavioral characteristics in a large retrospective cohort. Clin Genet 99:519–528. https://doi.org/10.1111/cge.13906
Girirajan S, Vlangos CN, Szomju BB, Edelman E, Trevors CD, Dupuis L et al (2006) Genotype-phenotype correlation in Smith-Magenis syndrome: evidence that multiple genes in 17p11.2 contribute to the clinical spectrum. Genet Med 8:417–427. https://doi.org/10.1097/01.gim.0000228215.32110.89
Acknowledgements
We want to thank the children and the parents and the clinicians who made this study possible. Some of the data obtained in this study were presented by us as an oral presentation named “Genetic variants in the patients with developmental and/or epileptic encephalopathy with spike-and-wave activation in sleep” at the 15th European Paediatric Neurology Society Congress (Abstract no: EPNS2023-2212).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
AT and KT wrote the main manuscript text and prepared the figures and tables. AT, ET, SGS created the study design. SGS, ET performed the EEG evaluation. SGS, AT, ET, ME, YA, HD gathered and prepared patient data. All authors reviewed, edited, and approved the manuscript.
Corresponding author
Ethics declarations
Competing interest
All authors have agreed to the final version of the manuscript. There are no conflicts of interest and no sponsor have been involved in any part of this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Electrical status epileptic during sleep presenting as bilateral spikes and waves activated in sleep in patient 8, 12, 13, 16, 17 respectively. "(Calibration: vertical bar = 70 μV, horizontal bar = 1000 msec; Bandpass 0.5–70 Hz). (PDF 372 kb)
Supplementary Fig. 2
Electrical status epileptic during sleep presenting as bilateral spikes and waves activated in sleep in patient 8, 12, 13, 16, 17 respectively. "(Calibration: vertical bar = 70 μV, horizontal bar = 1000 msec; Bandpass 0.5–70 Hz). (PDF 362 kb)
Supplementary Fig. 3
Electrical status epileptic during sleep presenting as bilateral spikes and waves activated in sleep in patient 8, 12, 13, 16, 17 respectively. "(Calibration: vertical bar = 70 μV, horizontal bar = 1000 msec; Bandpass 0.5–70 Hz). (PDF 726 kb)
Supplementary Fig. 4
Electrical status epileptic during sleep presenting as bilateral spikes and waves activated in sleep in patient 8, 12, 13, 16, 17 respectively. "(Calibration: vertical bar = 70 μV, horizontal bar = 1000 msec; Bandpass 0.5–70 Hz). (PDF 478 kb)
Supplementary Fig. 5
Electrical status epileptic during sleep presenting as bilateral spikes and waves activated in sleep in patient 8, 12, 13, 16, 17 respectively. "(Calibration: vertical bar = 70 μV, horizontal bar = 1000 msec; Bandpass 0.5–70 Hz). (PDF 434 kb)

Supplementary Fig. 6
Computational biochemical analysis of the human NaV1.6 Gly1626Leu mutant. (A) Cartoon representation of the wild-type NaV1.6 α-subunit complexed with the auxiliary β1-subunit (PDB ID: 8FHD). The structurally homologous domains I to IV are colored pale green, pale yellow, light blue and light pink, respectively. Visible portion of the NaV1.6 N-terminal domain is colored cherry red, and visible portion of the NaV1.6 C-terminal domain is colored olive green. The cytoplasmic interdomain linkers are colored black. Dashed lines indicate the invisible regions of the channel. (B) Combined cartoon and stick representation of a part of the wild-type NaV1.6 VSDIV. R1–R5 and K6 correspond to the gating charges, An1 and An2 correspond to the anionic centers, and HSC corresponds to the hydrophobic constriction site. (C) Combined cartoon and stick representation of a part of the mutant NaV1.6 VSDIV. The in silico introduced mutant Leu1626 residue is colored gray. All images were rendered with the PyMOL Molecular Graphics System, v18 (Schrödinger LLC, Portland, OR, USA). (PNG 6108 kb)

Supplementary Fig. 7
Computational biochemical analysis of the human KCC2 Gly549Ala (Gly526Ala) mutant. (A) Cartoon representation of the wild-type KCC2 homodimer complexed with the K+ and Cl– ions (PDB ID: 6M23). The two monomers (A and B) are colored pale cyan and wheat yellow, respectively. Dashed lines indicate the invisible regions of the cotransporter. (B) Combined cartoon and stick representation of a part of the mutant KCC2 intracellular loop interconnecting TM8 and TM9. The in silico introduced mutant Ala549 (Ala526) residue is colored gray. Both images were rendered with the PyMOL Molecular Graphics System, v18 (Schrödinger LLC, Portland, OR, USA). (C) Multiple sequence alignment of known electroneutral cation-coupled chloride cotransporters of the human SLC12 family (except CCC9). Uppercase letters denote identity, and lowercase letters denote a consensus level of greater than 0.5. The position of the affected glycine residue is marked by an arrow. The graphically enhanced alignment was prepared with ESPript 3.0. (PNG 6689 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Türkyılmaz, A., Sağer, S.G., Tekin, E. et al. Expanding the clinical and genetic landscape of (developmental) epileptic encephalopathy with spike-and-wave activation in sleep: results from studies of a Turkish cohort. Neurogenetics (2024). https://doi.org/10.1007/s10048-024-00751-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10048-024-00751-1