Abstract
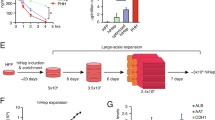
In recent years, studies on liver graft construction using the decellularized liver as a template for transplantation therapy have attracted much attention. However, the therapeutic effect of constructed liver grafts in hepatic failure has not been evaluated. Therefore, we aimed to develop a novel evaluation system demonstrating the curative effect of a constructed liver graft in animals with hepatic failure. First, we developed a highly reproducible rat model of hepatic failure by combining 80% partial hepatectomy with warm ischemia. In this model, severity could be controlled by the warm ischemic period. We also constructed a liver graft by recellularization of decellularized liver, and confirmed the ammonia metabolic function in the graft in vitro as one of the most important functions for recovery from hepatic failure. The graft was then applied to our developed hepatic failure rat model using a blood extracorporeal circulation system. In this application, the graft metabolized the ammonia in the blood of animals with hepatic failure and was thus suggested to be effective for the treatment of hepatic failure. In summary, a novel evaluation system for whole-organ-engineered liver graft as a preliminary stage of transplantation was developed. This system was expected to provide much information about the curative effect of a constructed liver graft.



Similar content being viewed by others
References
Kim WR, et al. OPTN/SRTR 2016 annual data report: liver. Am J Transplant. 2018;18(suppl 1):172–253.
Langer R, Vacanti JP. Tissue engineering. Science. 1993;260(5110):920–6.
Ohashi K, et al. Engineering functional two- and three-dimensional liver systems in vivo using hepatic tissue sheets. Nat Med. 2007;13:880–5.
Smith MK, et al. Delivery of hepatotrophic factors fails to enhance longer-term survival of subcutaneously transplanted hepatocytes. Tissue Eng. 2006;12(2):235–44.
Chen AA, et al. Humanized mice with ectopic artificial liver tissues. Proc Natl Acad Sci USA. 2011;108(29):11842–7.
Rotem A, et al. Oxygen uptake rates in cultured rat hepatocytes. Biotechnol Bioeng. 1992;40(10):1286–91.
Shirakigawa N, et al. Decellularized liver as a practical scaffold with a vascular network template for liver tissue engineering. J Biosci Bioeng. 2012;114(5):546–51.
Shirakigawa N, et al. Base structure consisting of an endothelialized vascular-tree network and hepatocytes for whole liver engineering. J Biosci Bioeng. 2013;116(6):740–5.
Uygun BE, et al. Organ reengineering through development of a transplantable recellularized liver graft using decellularized matrix. Nat Med. 2010;16(7):814–20.
Bao J, et al. Construction of a portal implantable functional tissue-engineered liver using perfusion-decellularized matrix and hepatocytes in rats. Cell Transplant. 2011;20(5):753–66.
Ko IK, et al. Bioengineered transplantable porcine livers with re-endothelialized vasculature. Biomaterials. 2015;40:72–9.
Yagi H, et al. Human-scale whole-organ bioengineering for liver transplantation: a regenerative medicine approach. Cell Transplant. 2013;22(2):231–42.
Meng F, et al. Whole liver engineering: a promising approach to develop functional liver surrogates. Liver Int. 2017;37(12):1759–72.
Seglen PO. Preparation of isolated rat liver cells. In: Prescott DM, editor. Methods of cell biology. New York: Academic Press; 1976. p. 29–83.
Ijima H, Kawakami K. Promotion of monolayer formation and high expression of ammonia metabolism of primary rat hepatocytes on arginine-glycine-aspartic acid-containing peptide-coated polystyrene dish. J Biosci Bioeng. 2005;100(1):62–6.
Bernal W, et al. Arterial ammonia and clinical risk factors for encephalopathy and intracranial hypertension in acute liver failure. Hepatology. 2007;46(6):1844–52.
Suh KS, et al. Bioartificial liver treatment in rats with fulminant hepatic failure: effect on DNA-binding activity of liver-enriched and growth-associated transcription factors. J Surg Res. 1999;85(2):243–50.
He Y, et al. A rat model for acute hepatic failure. Hepatobiliary Pancreat Dis Int. 2003;2(3):423–5.
Tuñón MJ, et al. An overview of animal models for investigating the pathogenesis and therapeutic strategies in acute hepatic failure. World J Gastroenterol. 2009;15(25):3086–98.
Eguchi S, et al. Fulminant hepatic failure in rats: survival and effect on blood chemistry and liver regeneration. Hepatology. 1996;24(6):1452–9.
Ijima H, et al. Evaluating the performance of a hybrid artificial liver support system with a recoverable hepatic failure rat model. Ann N Y Acad Sci. 2001;944:344–9.
Terblanche J, Hickman R. Animal models of fulminant hepatic failure. Dig Dis Sci. 1991;36(6):770–4.
Newsome PN, et al. Animal models of fulminant hepatic failure: a critical evaluation. Liver Transpl. 2000;6(1):21–31.
Takagi M, et al. Analysis of the ammonium metabolism of rat primary hepatocytes and a human hepatocyte cell line Huh 7. Cytotechnology. 2000;32(1):9–15.
Yamashita Y, et al. Efficacy of a larger version of the hybrid artificial liver support system using a polyurethane foam/spheroid packed-bed module in a warm ischemic liver failure pig model for preclinical experiments. Cell Transplant. 2003;12:101–7.
Acknowledgements
We are grateful to Prof. Aishima from Saga University for helpful discussions and valuable insights on histological evaluation. We would also like to thank Editage (www.editage.jp) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sakamoto, H., Shirakigawa, N., Bual, R.P. et al. A novel evaluation system for whole-organ-engineered liver graft by ex vivo application to a highly reproducible hepatic failure rat model. J Artif Organs 22, 222–229 (2019). https://doi.org/10.1007/s10047-019-01106-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-019-01106-6