Abstract
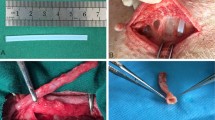
This study aims at the evaluation of blood vessel reconstruction process of decellularized small diameter vessels prepared by a hyperosmotic electrolyte solution treatment not only histologically but also physiologically in rat transplantation model. Complete cell removal by a hyperosmotic electrolyte solution treatment was confirmed by hematoxylin/eosin staining and scanning electron microscopic observation. All acellular vessels transplanted into the rat abdominal aorta were patent up to 14 months. One week post-transplantation, the vWF-positive cells were observed on the luminal surface but the layer formation did not complete. Five weeks following transplantation, the vWF-positive endothelial cells were located on the intima consistent with intact endothelial cells. Beneath the endothelial cells, α-SMA-positive smooth muscle cells were distributed. The harvested vessels displayed formation of tunica intima (endothelial cells) and tunica medulla (smooth muscle cell) layers. We also examined the physiological properties of the vessels 12 months post-transplantation using a wire myograph system. The transplanted vessels contracted upon addition of norepinephrine and relaxed upon addition of sodium nitroprusside as well as the native vessels. In conclusion, the acellular vessels prepared with hyperosmotic electrolytic solution showed excellent and long-term patency, which may be related to the successful preservation of vascular ECM. In addition, the acellular vessels revealed the intima/medulla regeneration with the physiological contraction–relaxation functions in response to the each substance.




Similar content being viewed by others
References
Bosiers M, Torsello G, Gissler HM, Ruef J, Muller-Hulsbeck S, Jahnke T, et al. Nitinol stent implantation in long superficial femoral artery lesions: 12-month results of the DURABILITY I study. J Endovasc Ther. 2009;16:261–9.
Chlupac J, Filova E, Bacakova L. Blood vessel replacement: 50 years of development and tissue engineering paradigms in vascular surgery. Physiol Res. 2009;58:S119–39.
Schaner PJ, Martin ND, Tulenko TN, Shapiro IM, Tarola NA, Leichter RF, et al. Decellularized vein as a potential scaffold for vascular tissue engineering. J Vasc Surg. 2004;40:146–53.
Lin PH, Chen C, Bush RL, Yao Q, Lumsden AB, Hanson SR. Small-caliber heparin-coated ePTFE grafts reduce platelet deposition and neointimal hyperplasia in a baboon model. J Vasc Surg. 2004;39:1322–8.
Scott EC, Glickman MH. Conduits for hemodialysis access. Semin Vasc Surg. 2007;20:158–63.
Matsumoto H, Hasegawa T, Fuse K, Yamamoto M, Saigusa M. A new vascular prosthesis for a small caliber artery. Surgery. 1973;74:519–23.
Daenens K, Schepers S, Fourneau I, Houthoofd S, Nevelsteen A. Heparin-bonded ePTFE grafts compared with vein grafts in femoropopliteal and femorocrural bypasses: 1- and 2-year results. J Vasc Surg. 2009;49:1210–6.
Heyligers JM, Verhagen HJ, Rotmans JI, Weeterings C, de Groot PG, Moll FL, et al. Heparin immobilization reduces thrombogenicity of small-caliber expanded polytetrafluoroethylene grafts. J Vasc Surg. 2006;43:587–91.
Hugl B, Nevelsteen A, Daenens K, Perez MA, Heider P, Railo M, et al. PEPE II—a multicenter study with an end-point heparin-bonded expanded polytetrafluoroethylene vascular graft for above and below knee bypass surgery: determinants of patency. J Cardiovasc Surg (Torino). 2009;50:195–203.
Bader A, Schilling T, Teebken OE, Brandes G, Herden T, Steinhoff G, et al. Tissue engineering of heart valves—human endothelial cell seeding of detergent acellularized porcine valves. Eur J Cardiothorac Surg. 1998;14:279–84.
Yeh HI, Lu SK, Tian TY, Hong RC, Lee WH, Tsai CH. Comparison of endothelial cells grown on different stent materials. J Biomed Mater Res A. 2006;76:835–41.
Sreerekha PR, Krishnan LK. Cultivation of endothelial progenitor cells on fibrin matrix and layering on dacron/polytetrafluoroethylene vascular grafts. Artif Organs. 2006;30:242–9.
Cho SW, Jeon O, Lim JE, Gwak SJ, Kim SS, Choi CY, et al. Preliminary experience with tissue engineering of a venous vascular patch by using bone marrow-derived cells and a hybrid biodegradable polymer scaffold. J Vasc Surg. 2006;44:1329–40.
Hoerstrup SP, Zund G, Sodian R, Schnell AM, Grunenfelder J, Turina MI. Tissue engineering of small caliber vascular grafts. Eur J Cardiothorac Surg. 2001;20:164–9.
Quint C, Kondo Y, Manson RJ, Lawson JH, Dardik A, Niklason LE. Decellularized tissue-engineered blood vessel as an arterial conduit. Proc Natl Acad Sci USA. 2011;108:9214–9.
Wu HC, Wang TW, Kang PL, Tsuang YH, Sun JS, Lin FH. Coculture of endothelial and smooth muscle cells on a collagen membrane in the development of a small-diameter vascular graft. Biomaterials. 2007;28:1385–92.
Haruguchi H, Teraoka S. Intimal hyperplasia and hemodynamic factors in arterial bypass and arteriovenous grafts: a review. J Artif Organs. 2003;6:227–35.
Lovett M, Eng G, Kluge JA, Cannizzaro C, Vunjak-Novakovic G, Kaplan DL. Tubular silk scaffolds for small diameter vascular grafts. Organogenesis. 2010;6:217–24.
Iwasaki K, Kojima K, Kodama S, Paz AC, Chambers M, Umezu M, et al. Bioengineered three-layered robust and elastic artery using hemodynamically-equivalent pulsatile bioreactor. Circulation. 2008;118:S52–7.
Enomoto S, Sumi M, Kajimoto K, Nakazawa Y, Takahashi R, Takabayashi C, et al. Long-term patency of small-diameter vascular graft made from fibroin, a silk-based biodegradable material. J Vasc Surg. 2010;51:155–64.
Nemcova S, Noel AA, Jost CJ, Gloviczki P, Miller VM, Brockbank KG. Evaluation of a xenogeneic acellular collagen matrix as a small-diameter vascular graft in dogs—preliminary observations. J Invest Surg. 2001;14:321–30.
Isenburg JC, Simionescu DT, Vyavahare NR. Elastin stabilization in cardiovascular implants: improved resistance to enzymatic degradation by treatment with tannic acid. Biomaterials. 2004;25:3293–302.
Malone JM, Brendel K, Duhamel RC, Reinert RL. Detergent-extracted small-diameter vascular prostheses. J Vasc Surg. 1984;1:181–91.
Schmidt CE, Baier JM. Acellular vascular tissues: natural biomaterials for tissue repair and tissue engineering. Biomaterials. 2000;21:2215–31.
Teebken OE, Bader A, Steinhoff G, Haverich A. Tissue engineering of vascular grafts: human cell seeding of decellularised porcine matrix. Eur J Vasc Endovasc Surg. 2000;19:381–6.
Wilson GJ, Yeger H, Klement P, Lee JM, Courtman DW. Acellular matrix allograft small caliber vascular prostheses. ASAIO Trans. 1990;36:M340–3.
Yang D, Guo T, Nie C, Morris SF. Tissue-engineered blood vessel graft produced by self-derived cells and allogenic acellular matrix: a functional performance and histologic study. Ann Plast Surg. 2009;62:297–303.
Lalka SG, Oelker LM, Malone JM, Duhamel RC, Kevorkian MA, Raper BA, et al. Acellular vascular matrix: a natural endothelial cell substrate. Ann Vasc Surg. 1989;3:108–17.
Ahn JM, Kim SJ, Kim H, Park C, Kim WH, Park JH. Triton X-100 induces apoptosis in human hepatoma cell lines. Yonsei Med J. 1997;38:52–9.
Ehashi T, Nishigaito A, Fujisato T, Moritan Y, Yamaoka T. Peripheral nerve regeneration and electrophysiological recovery with CIP-treated allogeneic acellular nerves. J Biomater Sci Polym Ed. 2011;22:627–40.
Veith FJ, Moss CM, Sprayregen S, Montefusco C. Preoperative saphenous venography in arterial reconstructive surgery of the lower extremity. Surgery. 1979;85:253–6.
Kawada H, Fujita J, Kinjo K, Matsuzaki Y, Tsuma M, Miyatake H, et al. Nonhematopoietic mesenchymal stem cells can be mobilized and differentiate into cardiomyocytes after myocardial infarction. Blood. 2004;104:3581–7.
Krenning G, Moonen JR, van Luyn MJ, Harmsen MC. Generating new blood flow: integrating developmental biology and tissue engineering. Trends Cardiovasc Med. 2008;18:312–23.
Saiura A, Sata M, Hirata Y, Nagai R, Makuuchi M. Circulating smooth muscle progenitor cells contribute to atherosclerosis. Nat Med. 2001;7:382–3.
Haruna Y, Morita Y, Komai N, Yada T, Sakuta T, Tomita N, et al. Endothelial dysfunction in rat adjuvant-induced arthritis: vascular superoxide production by NAD(P)H oxidase and uncoupled endothelial nitric oxide synthase. Arthritis Rheum. 2006;54:1847–55.
Tanaka Y, Mochizuki Y, Tanaka H, Shigenobu K. Significant role of neuronal non-N-type calcium channels in the sympathetic neurogenic contraction of rat mesenteric artery. Br J Pharmacol. 1999;128:1602–8.
Sbitany H, Serletti JM. Acellular dermis-assisted prosthetic breast reconstruction: a systematic and critical review of efficacy and associated morbidity. Plast Reconstr Surg. 2011;128:1162–9.
Glasberg SB, Light D. AlloDerm and Strattice in breast reconstruction: a comparison and techniques for optimizing outcomes. Plast Reconstr Surg. 2012;129:1223–33.
Meyer SR, Chiu B, Churchill TA, Zhu L, Lakey JR, Ross DB. Comparison of aortic valve allograft decellularization techniques in the rat. J Biomed Mater Res A. 2006;79:254–62.
Acknowledgments
This study was funded by JSPS KAKENHI (Grant Number 22791722). The funding agency received no commercial benefit from this study.
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakakibara, S., Ishida, Y., Hashikawa, K. et al. Intima/medulla reconstruction and vascular contraction–relaxation recovery for acellular small diameter vessels prepared by hyperosmotic electrolyte solution treatment. J Artif Organs 17, 169–177 (2014). https://doi.org/10.1007/s10047-014-0760-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-014-0760-6