Abstract
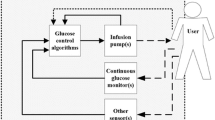
Artificial pancreas technology, involving “closed-loop” controls with real-time blood glucose monitoring, has been increasing in reliability as its potential for clinical use and application grows. One such device, based on this technology, is the STG-22 (Nikkiso Co., Ltd., Tokyo, Japan) artificial pancreas apparatus. In order to assess the reliability and accuracy of the device for measuring blood glucose, it is important to compare its readings to those obtained using a ‘gold standard’ method, such as the hexokinase method. Therefore, in the present study, canine blood [glucose] measurements using the STG-22 were compared to those obtained using a previously established commercial reagent, Quickauto-Neo GLU-HK. Furthermore, two different sample types (whole blood versus plasma constituent) were compared to determine which sample type results in more accurate and optimal readings with the STG-22. Given that the STG-22 was not primarily designed for canine blood samples, results for canine blood samples were not accurate. Measurements performed by the STG-22 with whole blood were significantly lower than reference [glucose] counterparts. Alternatively, an opposite trend was observed with plasma measurements that were significantly higher. A conversion format using the following formula, Hexokinase [glucose] = STG-22 [glucose] × 1.407 + 1.532, was observed with canine samples in our study.



Similar content being viewed by others
References
Okabayashi T, Hanazaki K, Nishimori I, Sugimoto T, Maeda H, Yatabe T, Dabanaka K, Kobayashi M, Yamashita K. Continuous post-operative blood glucose monitoring and control using a closed-loop system in patients undergoing hepatic resection. Dig Dis Sci. 2008;53:1405–10.
Kono T, Hanazaki K, Yazawa K, Ashizawa S, Fisher WE, Wang X-P, Nose Y, Brunicaidi FC. Pancreatic polypeptide administration reduces insulin requirements of artificial pancreas in pancreatectomized dogs. Artif Organs. 2005;29:83–9.
Hiramatsu S, Sako Y, Mimura K, Iwashige K, Taniuchi S, Umeda F, Nawata H. Impaired feedback inhibition of insulin secretion by hyperinsulinemia in patients with insulinoma. Endocrine J. 1995;42:39–42.
Suehiro T, Osaki F, Ikeda Y, Arii K, Nakayama F, Ota K, Hashimoto K. Type 1 diabetes developed in a type 2 diabetic patient with severe insulin resistance. Diabetes Res Clin Pract. 2005;70:298–302.
Heinemann L, Linkeschova R, Rave K, Hompesch B, Sedlak M, Heise T. Time-action profile of the long-acting insulin analog insulin glargine (HOE901) in comparison with those of NPH insulin and placebo. Diabetes Care. 2000;23:644–9.
Heise T, Nosek L, Ronn BB, Endahl L, Heinemann L, Kapitza C, Draeger E. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes. 2004;53:1614–20.
Madarame H, Kayanuma H, Shida T, Tsuchiya R. Retrospective study of canine insulinomas: eight cases (2005–2008). J Vet Med Sci. 2009;71:905–11.
Mori A, Lee P, Sako T, Mizutani H, Arai T. Successful intensive insulin treatment of type 1 diabetic dogs leads to restoration of peripheral leukocyte insulin signaling gene expression and enzyme activities. J Vet Med Sci. 2009;71:1017–26.
Sako T, Mori A, Lee P, Oda H, Saeki K, Miki Y, Kurishima M, Mimura K, Nozawa S, Mizutani H, Makino Y, Ishioka K, Arai T. Time-action profiles of insulin detemir in normal and diabetic dogs. Res Vet Sci. 2010 (in press). doi:10.1016/j.rvsc.2010.07.001.
Holford AL, Tobias KM, Bartges JW, Johnson BM. Adrenal response to adrenocorticotropic hormone in dogs before and after surgical attenuation of a single congenital portosystemic shunt. J Vet Intern Med. 2008;22:832–8.
Sakai M, Asano K, Nakata M, Takahashi T, Koie H, Yamaya Y, Watari T, Shibuya H, Sato T, Tokuriki M, Hasegawa A. Diabetes mellitus after resection of hepatocellular carcinoma with hypoglycemia in a dog. J Vet Med Sci. 2006;68:765–7.
Barman BN, Ashwood ER, Giddings JC. Separation and size distribution of red blood cells of diverse size, shape, and origin by flow/hyperlayer field-flow fractionation. Anal Biochem. 1993;212:35–42.
Hawkey CM, Dennett TB. Normal and abnormal red cells. In: A color atlas of comparative veterinary haematology. London: Wolfe Medical Publications Ltd; 1989. p. 9–57.
Higgins PJ, Garlick RL, Bunn HF. Glycosylated hemoglobin in human and animal red cells. Role of glucose permeability. Diabetes. 1982;31:743–8.
Rendell M, Stephen PM, Paulsen R, Valentine JL, Rasbold K, Hestorff T, Eastberg S, Shint DC. An interspecies comparison of normal levels of glycosylated hemoglobin and glycosylated albumin. Comp Biochem Physiol B. 1985;81:819–22.
Kadish AH, Hall DA. A new method for the continuous monitoring of blood glucose by measurement of dissolved oxygen. Clin Chem. 1965;11:869–75.
Yamashita K, Yatabe T. Intraoperative glycemic control procedures and the use of an artificial pancreas. World J Gastroenterol. 2009;15:4126–31.
Yamashita K, Okabayashi T, Yokoyama T, Yatabe T, Maeda H, Manabe M, Hanazaki K. Accuracy and reliability of continuous blood glucose monitor in post-surgical patients. Acta Anaesthesiol Scand. 2009;53:66–71.
Acknowledgments
This work was supported in part by the MEXT 2008 Strategic Research Base Development Program for Private Universities.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Mori, A., Lee, P., Yokoyama, T. et al. Evaluation of artificial pancreas technology for continuous blood glucose monitoring in dogs. J Artif Organs 14, 133–139 (2011). https://doi.org/10.1007/s10047-011-0560-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-011-0560-1