Abstract
A floodplain aquifer within an agricultural watershed near Madison, Wisconsin (USA), was studied to determine whether denitrification was occurring below the surface organic layer. Groundwater levels and concentrations of O2, Cl−, NO −3 , SO 2−4 , dissolved organic carbon (DOC), and major cations were monitored over a 1-year period along a 230-m transect between an agricultural field and a stream discharge point. Seventeen groundwater samples were analyzed for δ15NNO3 and δ18ONO3 composition. Samples in which NO −3 was too low for stable isotope analysis were analyzed for excess dissolved N2. Groundwater NO −3 concentrations declined between the agricultural field and the discharge point. Chloride and δ15NNO3/δ18ONO3 data indicated that the drop in NO −3 was caused primarily by dilution of shallow NO −3 -rich water with deeper, NO −3 -depleted groundwater. Two localized zones of denitrification were identified in the upland-wetland transition by their δ15NNO3 and δ18ONO3 signatures, and two in the stream hyporheic zone by the presence of excess dissolved N2. The combined stratigraphic, hydrologic, and geochemical data in these locations correspond to groundwater mixing zones where NO −3 is delivered to subsurface layers that support denitrification fueled by dissolved (e.g. DOC or dissolved Fe(II)) and/or solid-phase (e.g. particulate organic carbon, solid-associated Fe(II), or pyrite) electron donors.
Résumé
Un aquifère situé sous plaine inondable à l’intérieur d’un bassin versant agricole, près de Madison, Wisconsin (USA), a été étudié afin de déterminer si une dénitrification avait lieu sous la couche organique de surface. Le niveau des eaux souterraines et la concentration en O2, Cl−, NO −3 , SO 2−4 , carbone organique dissous et majeurs cationiques ont été contrôlés pendant une année, le long d’un transect de 230-m, entre un terrain agricole et le cours d’eau drainant. La teneur en δ15NNO3 and δ18ONO3 de dix sept échantillons d’eau souterraine a été mesurée. Pour les échantillons dans lesquels NO −3 était trop bas pour permettre une détermination des isotopes stables, l’analyse a porté sur le N2 dissous en excès. La concentration en NO −3 des eaux souterraines diminue entre le terrain agricole et le cours d’eau drainant. Les données sur les chlorures et le rapport δ15NNO3/δ18ONO3 indiquent que la baisse du NO −3 est due principalement à la dilution de l’eau peu profonde riche en cet élément par une eau souterraine plus profonde moins chargée. Deux zones localisées de dénitrification ont été identifiées dans le domaine de transition entre le plateau et la zone humide grâce à leur signature δ15NNO3 and δ18ONO3 et deux autres dans l’inféro-flux grâce à la présence de N2 dissous en excès. Les données conjuguées de la stratigraphie, de l’hydrologie et de la géochimie dans ces localisations délimitent les zones de mélange des eaux souterraines, où NO −3 est transféré aux couches de sub-surface, qui sont le siège d’une dénitrification alimentée par les donneurs d’électrons de la phase dissoute (par exemple le carbone organique dissous COD ou le Fe(II) dissous) et/ou de la phase solide (par exemple le carbone organique des particules, le Fe(II) associé à la phase solide ou la pyrite).
Resumen
Se estudió un acuífero de llanura aluvial dentro de una cuenca agrícola cerca de Madison, Wisconsin (EEUU), para determinar si la desnitrificación estaba ocurriendo debajo de la capa orgánica superficial. Se monitorearon los niveles de agua subterráneas y las concentraciones de O2, Cl−, NO −3 , SO 2−4 , carbono orgánico disuelto (DOC), y los cationes mayoritarios durante un período de un año a lo largo de una transecta de 230 m entre un campo agrícola y un punto de descarga de una corriente. Se analizaron diecisiete muestras de aguas subterránea para la composición de δ15NNO3 y δ18ONO3. Las muestras en las cuales NO −3 fue demasiado bajo para un análisis de isótopos estables fueron analizados para el exceso de N2.disuelto. Las concentraciones de NO −3 en el agua subterránea disminuyeron entre el campo agrícola y el punto de descarga. Los datos de cloruros y δ15NNO3/δ18ONO3 indicaron que la caída de nitrato fue causada principalmente por dilución de agua somera rica en NO −3 con agua subterránea extraída con aguas subterránea con nitrato más profunda. Se identificaron dos zonas de desnitrificación en la transición de la llanura alta y el humedal por sus trazas de δ15NNO3 y δ18ONO3, y dos en la zona hiporreica de la corriente por la presencia de exceso de N2.disuelto. Los datos geoquímicos, hidrológicos y estratigráficos combinados en estos sitios corresponden a zonas de mezcla de aguas subterráneas donde el NO −3 es entregado a las capas subsuperficiales que suportan la desnitrificación, alimentados por electrones donores de la fase disuelta (por ejemplo DOC o Fe(II)) y/o electrones de la fase sólida (por ejemplo carbono orgánico particulado, Fe (II) asociado a Fe(II), o pirita)
摘要
本文通过对美国威斯康星州麦迪逊市附近的一个农业流域的泛滥平原含水层的研究, 确定地表有机层以下反硝化作用是否发生。在农田和溪流排泄点之间一个230m长的横断面上检测了超过一年的地下水位, O2、Cl−、 NO −3 、 SO =4 的浓度, 溶解有机碳(DOC), 以及主要的阳离子。分析17个地下水样中NO −3 的δ15N和δ18O值, 并对NO −3 中稳定性同位素低的水样进一步分析过量的溶解N2。在农田和排泄点之间, 地下水中NO −3 的浓度降低。氯化物和NO −3 中δ15N和δ18O的比值表明NO −3 的降低主要由浅部NO −3 含量较高的地下水与深部NO −3 耗尽的地下水稀释引起的。通过NO −3 的δ15N和δ18O值的鲜明特征, 将山地-湿地过渡区分为两个反硝化的局部作用带, 并通过过多的溶解N2的存在, 划分两个底流区。这些区域地层的、水文的以及地球化学的数据与地下水混合区相一致。在该区NO −3 迁移到地下, 并以溶解的 (如DOC或溶解的Fe++) 和固体相的 (如微粒状的有机碳、固相的Fe++, 或者黄铁矿) 电子供体为原料进行反硝化作用。
Resumo
Estudou-se um aquífero de planície de inundação dentro de uma bacia hidrográfica agrícola perto de Madison, Wisconsin (EUA), para determinar se ocorria desnitrificação abaixo da camada orgânica superficial, Durante um ano monitorizaram-se os níveis piezométricos e as concentrações de O2, Cl−, NO −3 , SO 2−4 , carbono orgânico dissolvido (COD) e catiões maiores ao longo de um corte transversal de 230 m entre um campo agrícola e um ponto de descarga numa linha de água. Analisou-se a composição de δ15NNO3 e δ18ONO3 em 17 amostras de águas subterrâneas. Nas amostras em que o NO −3 era muito baixo para análise de isótopos estáveis foi analisado o N2 dissolvido em excesso. As concentrações de NO −3 das águas subterrâneas diminuíram entre o campo agrícola e o ponto de descarga. Os dados de cloreto e δ15NNO3/δ18ONO3 indicaram que a queda do NO −3 foi causada em primeiro lugar pela diluição de água subsuperficial rica em NO −3 com água subterrânea mais profunda empobrecida em NO −3 . Identificaram-se duas zonas localizadas de desnitrificação na transição entre a zona superior e a zona húmida pelas suas assinaturas de δ15NNO3 e δ18ONO3, e duas na zona de fluxo hiporreíco da linha de água através da presença de N2 dissolvido em excesso. As combinações de dados de estratigrafia, hidrologia e geoquímica nestes locais correspondem a zonas de mistura de águas subterrâneas onde o NO −3 é entregue às camadas subsuperficiais que sustentam a desnitrificação impulsionadas por dadores de electrões dissolvidos (e.g. COD ou Fe(II) dissolvido) e/ou de fase sólida (e.g. carbono orgânico particulado, Fe(II) associado a sólidos ou pirite)









Similar content being viewed by others
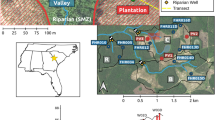
References
Anderson K (2002) Hydrogeologic controls on flow to Frederick Springs in the Pheasant Branch watershed, Middleton. Wisconsin, MSc Thesis, University of Wisconsin-Madison, USA
Böhlke J, Wanty R, Tuttle M, Delin G, Landon M (2002) Denitrification in the recharge area and discharge area of a transient agricultural nitrate plume in a glacial outwash sand aquifer, Minnesota. Water Resour Res 38:10.1–10.26
Bradbury K, Swanson S, Krohelski J, Fritz A (1999) Hydrogeology of Dane County, Wisconsin. Open-File Report 1999-04, Wisconsin Geological and Natural History Survey, University of Wisconsin Extension, Madison, WI
Brandes J, Devol A (1995) Simultaneous nitrate and oxygen respiration in coastal sediments: evidence for discrete diagenesis. J Mar Res 53:771–797
Browne B (2004) Pump-induced ebullition: a unified and simplified method for measuring multiple dissolved gases. Environ Sci Technol 38:5729–5736
Browne B, Kraft G, Bowling J, DeVita W, Mechenich D (2008) Collateral geochemical impacts of agricultural nitrogen enrichment from 1963 to 1985: a southern Wisconsin ground water depth profile. J Environ Qual 37:1456–1467
Cey E, Rudolph D, Aravena R, Parkin G (1999) Role of the riparian zone in controlling the distribution and fate of agricultural nitrogen near a small stream in southern Ontario. J Contam Hydrol 37:45–67
Cline D (1965) Geology and groundwater resources of Dane County, Wisconsin. US Geol Surv Water Suppl Pap 1779-U, 64 pp
Dane County (2005) Dane County Land Information Office and Fly Dane Partnership, 20060327, One-Foot Resolution Digital Orthophotography. Dane County Land Information Office, Madison, WI. http://dcimap.co.dane.wi.us/dcimap/index.htm. Cited November 2005
Dane County (2006) DCiMap. Dane County Land Information Office, Madison, WI. http://dcimap.co.dane.wi.us/dcimap/index.htm. Cited July 2006
Devito K, Fitzgerald D, Hill A, Aravena R (2000) Nitrate dynamics in relation to lithology and hydrologic flowpath in a river riparian zone. J Environ Qual 29:1075–1084
Ernstsen V (1996) Reduction of nitrate by Fe2+ in clay minerals. Clays Clay Miner 44:599–608
Ernstsen V, Binnerup S, Sorensen J (1998a) Reduction of nitrate in clayey subsoils controlled by geochemical and microbial barriers. Geomicrobiol J 15:195–207
Ernstsen V, Gates W, Stucki J (1998b) Microbial reduction of structural iron in clays: a renewable source of reduction capacity. J Environ Qual 27:761–766
Foster S (2000) Assessing and controlling the impacts of agriculture on groundwater: from barley barons to beef bans. Q J Eng Geol Hydrogeol 33:263–280
Fukada T, Hiscock K, Dennis P, Grischek T (2003) A dual isotope approach to identify denitrification in ground water at a river bank infiltration site. Water Res 37:3070–3078
Gillham R, Cherry J (1978) Field evidence of denitrification in shallow groundwater flow systems. Water Pollut Res J Canada 13:53–71
Haycock N, Burt T (1993) Role of floodplain sediments in reducing the nitrate concentration of substrate run-off: a case study in the Cotswolds, U.K. Hydrol Proc 7:287–295
Haycock N, Pinay G, Walker C (1993) Nitrogen retention in river corridors: European perspectives. Ambio 22:340–346
Hill A (1996) Nitrate removal in stream riparian zones. J Environ Qual 25:743–755
Hill A, Devito K, Campagnolo S, Samugadas K (2000) Subsurface denitrification in a forest riparian zone: interactions between hydrology and supplies of nitrate and organic carbon. Biogeochemistry 51:193–223
Hill A, Vidon P, Langat J (2004) Denitrification potential in relation to lithology in five headwater riparian zones. J Environ Qual 33:911–919
Howarth R, Marino R (2006) Nitrogen as the limiting nutrient for eutrophication in coastal marine ecosystems: evolving views over three decades. Limnol Oceanogr 51:364–376
Hunt R, Bradbury K, Krohelski J (2001) The effects of large-scale pumping and diversion on the water resources of Dane County, Wisconsin. US Geol Surv Fact Sheet FS-127-01. http://wi.water.usgs.gov/pubs/fs-127-01/fs-127-01.pdf. Cited February 2010
Jorgensen C, Jacobsen O, Elberling B, Aamand J (2009) Microbial oxidation of pyrite coupled to nitrate reduction in anoxic groundwater sediment. Environ Sci Technol 43:4851–4857
Keeney D (1986) Sources of nitrate to groundwater. Crit Rev Environ Control 16:257–304
Kendall C (1998) Tracing nitrogen sources and cycling in catchments. In: Kendall C, McDonnell JJ (eds) Isotope tracers in catchment hydrology. Elsevier, Amsterdam, pp 519–575
Kurtz A, Bahr J, Carpenter Q, Hunt R (2007) The importance of subsurface geology for water source and vegetation communities in Cherokee Marsh, Wisconsin. Wetlands 27:189–202
Lowrance R, Todd R, Asmussen L (1984) Nutrient cycling in an agricultural watershed. 1. Phreatic movement. J Environ Qual 13:22–27
Mariotti A, Germon J, Hubert P, Kaiser P, Letolle R, Tardieux A, Tardieux P (1981) Experimental determination of nitrogen kinetic isotope fractionation: some principles—illustration for the denitrification and nitrification process. Plant Soil 62:423–430
Mariotti A, Landreau A, Simon B (1988) 15N isotope biogeochemistry and natural denitrification process in groundwater: application to the chalk aquifer of northern France. Geochim Cosmochim Acta 52:1869–1878
Mayer B, Boyer E, Goodale C, Jaworski N, Breemen N, Howarth R, Seitzinger S, Billen G, Lajtha K, Nadelhoffer K, Dam D, Hetling L, Nosal M, Paustian K (2002) Sources of nitrate in rivers draining sixteen watersheds in the northeastern U.S.: isotopic constraints. Biogeochemistry 57(58):171–197
McClain M, Boyer E, Dent C, Gergel S, Grimm N, Groffman P, Hart S, Harvey J, Johnston C, Mayorga E, McDowell W, Pinay G (2003) Biogeochemical hot spots and hot moments at the interface of terrestrial and aquatic ecosystems. Ecosystems 6:301–312
Mengis M, Schiff S, Harris M, English M, Aravena R, Elgood R, MacLean A (1999) Multiple geochemical and isotopic approaches for assessing ground water NO −3 elimination in a riparian zone. Ground Water 37:448–457
Miller C (2009) Influence of wetland dynamics on microbial redox transformations of nitrate and iron. MSc Thesis, Department of Geology and Geophysics, University of Wisconsin-Madison, USA
Osborne L, Kovacic D (1993) Riparian vegetated buffer strips in water quality restoration and stream management. Freshwater Biol 29:243–258
Parry R (1998) Agricultural phosphate and water quality: a U.S. Environmental Protection Agency perspective. J Environ Qual 27:258–261
Peterjohn W, Correll D (1984) Nutrient dynamics in an agricultural watershed: observations on the role of a riparian forest. Ecology 65:1466–1475
Pfeiffer S, Bahr J, Beilfuss R (2006) Identification of groundwater flowpaths and denitrification zones in a dynamic floodplain aquifer. J Hydrol 325:262–272
Pinay G, Dechamps H (1988) The role of riparian woods in regulating nitrogen fluxes between the alluvial aquifer and surface water: a conceptual model. Reg Rivers Res Manage 2:507–516
Postma D, Boesen C, Kristiansen H, Larsen F (1991) Nitrate reduction in an unconfined sandy aquifer: water chemistry, reduction processes, and geochemical modeling. Water Resour Res 27:2027–2045
Rivett M, Buss S, Morgan P, Smith J, Bemment C (2008) Nitrate attenuation in groundwater: a review of biogeochemical controlling processes. Water Res 42:4215–4232
Rogers J (2006) Hydrologic and water quality characteristics of a small wetland: upper Dorn Creek wetland, Wisconsin. MSc Thesis, Department of Civil and Environmental Engineering, University of Wisconsin-Madison, USA
Schippers A, Jorgensen B (2001) Biogeochemistry of pyrite and iron sulfide oxidation in marine sediments. Geochim Cosmochim Acta 65:915–922
Seitzinger S, Harrison J, Böhlke J, Bouwman A, Lowrance R, Peterson B, Tobias C, Van Drecht G (2006) Denitrification across landscapes and waterscapes: a synthesis. Ecol App 16:2064–2090
Sexstone A, Revsbech N, Parkin T, Tiedje J (1985) Direct measurement of oxygen profiles and denitrification rates in soil aggregates. Soil Sci Soc Am J 49:645–651
Starr R, Gillham R (1993) Denitrification and organic carbon availability in two aquifers. Ground Water 31:934–947
Stites W, Chambers L (1991) A method for installing miniature multilevel sampling wells. Ground Water 29:430–432
Straub K, Benz M, Schink B, Widdel F (1996) Anaerobic, nitrate-dependent microbial oxidation of ferrous iron. Appl Environ Microbiol 62:1458–1460
Straub K, Schonhuber W, Buchholz-Cleven B, Schink B (2004) Diversity of ferrous iron-oxidizing, nitrate-reducing bacteria and their involvement in oxygen-independent iron cycling. Geomicrobiol J 21:371–378
Thayalakumaran T, Bristow K, Charlesworth P, Fass T (2008) Geochemical conditions in groundwater systems: implications for the attenuation of agricultural nitrate. Agric Water Manage 95:103–115
Triska F, Kennedy V, Avanzino R, Zellweyer G, Bencala K (1989) Retention and transport of nutrients in a third order stream in northwestern California: hyporheic processes. Ecology 70:1893–1905
Turner R, Rabalais N (1994) Coastal eutrophication near the Mississippi River Delta. Nature 368:619–621
US Geological Survey (1983) 7.5 minute quadrangle maps: Springfield Corners, WI, and Waunakee, WI. Wisconsin Geological and Natural History Survey, Madison, WI
Vidon P, Hill A (2004a) Landscape controls on nitrate removal in stream riparian zones. Water Resour Res 40:W03201
Vidon P, Hill A (2004b) Denitrification and patterns of electron donors and acceptors in eight riparian zones with contrasting hydrogeology. Biogeochemistry 71:259–283
Wassenaar L (1995) Evaluation of the origin and fate of nitrate in the Abbotsford Aquifer using the isotopes of 15N and 18O on NO −3 . Appl Geochem 10:391–405
Weber K, Picardal F, Roden E (2001) Microbially-catalyzed nitrate-dependent oxidation of biogenic solid-phase Fe(II) compounds. Environ Sci Technol 35:1644–1650
Weber K, Churchill P, Urrutia M, Kukkadapu R, Roden E (2006) Anaerobic redox cycling of iron by wetland sediment microorganisms. Environ Microbiol 8:100–113
Zhang Y, Slomp C, Broers H, Passier H, Van Cappellen P (2009) Denitrification coupled to pyrite oxidation and changes in groundwater quality in a shallow sandy aquifer. Geochim Cosmochim Acta 73:6716–6726
Acknowledgements
We thank R. Sharma, J. Dockery, C. Lowry, M. Cobb, and J. Wyss for field assistance. We also acknowledge the generous assistance from the U.S. Geological Survey Stable Isotope Laboratory in Reston, VA, and the University of Wisconsin-Stevens Point Dissolved Gas Laboratory. Funding was provided by the University of Wisconsin Department of Geology and Geophysics, with additional funding from a Brown and Caldwell Eckenfelder Scholarship, an Anna Grant Birge Memorial Award, and a Sigma Xi Grant-in-Aid of Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Craig, L., Bahr, J.M. & Roden, E.E. Localized zones of denitrification in a floodplain aquifer in southern Wisconsin, USA. Hydrogeol J 18, 1867–1879 (2010). https://doi.org/10.1007/s10040-010-0665-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10040-010-0665-2



