Abstract
Purpose
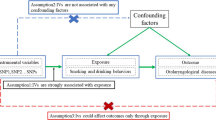
As the global population continues to age, there is a noticeable yearly rise in the incidence of hernias. Simultaneously, smoking, a widespread addictive behavior and a significant contributor to mortality, has evolved into a pervasive public health concern. Existing literature has already established a connection between smoking and an increased risk of postoperative recurrence and postoperative infections following hernia surgery. However, there remains a dearth of research exploring the association between smoking and hernia morbidity. In this study, our objective is to systematically evaluate the causal relationship between cigarette smoking behaviors and hernia morbidity using a Mendelian randomization (MR) approach.
Methods
Hernia-related data were sourced from the FinnGen Biobank database, while cigarette smoking behavior data were gathered from the GWAS and Sequencing Consortium of Alcohol and Nicotine Use. To assess the causal relationship, we employed five methods: the weighted median, the weighted mode the inverse variance weighted (IVW), MR-Egger, and the simple mode. Sensitivity analysis was conducted, incorporating Cochran's Q test, the MR-Egger intercept test, leave-one-out analysis, and funnel plot. The presentation of the causal relationship is expressed as an odds ratio (OR) along with their corresponding 95% confidence intervals (CI).
Results
Employing the IVW method as the reference standard, we found that smoking intensity is associated with an increased risk of diaphragmatic hernia (OR = 1.21, 95% CI 1.00–1.46, P = 0.047). These consistent findings were further corroborated by the weighted median and weighted mode methods (OR = 1.26, 95% CI 1.03–1.54, P = 0.026; OR = 1.25, 95% CI 1.02–1.52, P = 0.045). Conversely, when applying the IVW method, we identified no statistically significant causal relationship between smoking age, smoking initiation status, smoking cessation status, and the incidence of hernia.
Conclusions
Our MR study has uncovered genetic evidence linking smoking intensity and the occurrence of diaphragmatic hernia. The risk of developing diaphragmatic hernia rises in tandem with the intensity of smoking. This emphasizes the crucial role of regularly advising patients to cease smoking in clinical settings.




Similar content being viewed by others
Data availability
The outcome data used to conduct the analyses in the present study were obtained from FinnGen study https://www.finngen.fi/en (accessed on 11 September 2023). The exposure data used to conduct the analyses in the present study were obtained from GWAS and Sequencing Consortium of Alcohol and Nicotine Use https://conservancy.umn.edu/handle/11299/201564 (accessed on 11 September 2023). The data used to screen out SNPs were accessed from the human genotype–phenotype association database, PhenoScanner http://www.phenoscanner.medschl.cam.ac.uk (accessed on 11 September 2023).
References
Otto, J, Lindenau, T, Junge, K (2023). Hernia. In: Billmann, F, Keck, T (eds) Essentials of Visceral Surgery, Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-66735-4_13
Kingsnorth A, LeBlanc K (2003) Hernias: inguinal and incisional. Lancet 362(9395):1561–1571. https://doi.org/10.1016/S0140-6736(03)14746-0
Poulose BK, Shelton J, Phillips S, Moore D, Nealon W et al (2012) Epidemiology and cost of ventral hernia repair: making the case for hernia research. Hernia 16(2):179–183. https://doi.org/10.1007/s10029-011-0879-9
Abrahamson J (1998) Etiology and pathophysiology of primary and recurrent groin hernia formation. Surg Clin North Am 78(6):953–72, vi. https://doi.org/10.1016/S0039-6109(05)70364-9
Lau H, Fang C, Yuen WK, Patil NG (2007) Risk factors for inguinal hernia in adult males: a case-control study. Surgery 141(2):262–266. https://doi.org/10.1016/j.surg.2006.04.014
GBD (2019) Tobacco Collaborators (2021) Spatial, temporal, and demographic patterns in prevalence of smoking tobacco use and attributable disease burden in 204 countries and territories, 1990–2019: a systematic analysis from the Global Burden of Disease Study 2019. Lancet 397(10292):2337–2360. https://doi.org/10.1016/S0140-6736(21)01169-7
Gandini S, Botteri E, Iodice S, Boniol M, Lowenfels AB et al (2008) Tobacco smoking and cancer: a meta-analysis. Int J Cancer 122(1):155–164. https://doi.org/10.1002/ijc.23033
Parker SG, Mallett S, Quinn L, Wood CPJ, Boulton RW, et al (2021) Identifying predictors of ventral hernia recurrence: systematic review and meta-analysis. BJS Open 5(2):zraa071. https://doi.org/10.1093/bjsopen/zraa071
Park H, de Virgilio C, Kim DY, Shover AL, Moazzez A (2021) Effects of smoking and different BMI cutoff points on surgical site infection after elective open ventral hernia repair. Hernia 25(2):337–343. https://doi.org/10.1007/s10029-020-02190-x
Sekula P, Del Greco MF, Pattaro C, Köttgen A (2016) Mendelian randomization as an approach to assess causality using observational data. J Am Soc Nephrol 27(11):3253–3265. https://doi.org/10.1681/ASN.2016010098
Richmond RC, Davey Smith G (2022) Mendelian randomization: concepts and scope. Cold Spring Harb Perspect Med 12(1):a040501. https://doi.org/10.1101/cshperspect.a040501
Davey Smith G, Hemani G (2014) Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet 23(R1):R89-98. https://doi.org/10.1093/hmg/ddu328
Larsson SC, Burgess S, Michaëlsson K (2019) Smoking and stroke: a mendelian randomization study. Ann Neurol 86(3):468–471. https://doi.org/10.1002/ana.25534
Yuan S, Chen J, Ruan X, Sun Y, Zhang K et al (2023) Smoking, alcohol consumption, and 24 gastrointestinal diseases: mendelian randomization analysis. Elife 12:e84051. https://doi.org/10.7554/eLife.84051
Zhou W, Liu G, Hung RJ, Haycock PC, Aldrich MC et al (2021) Causal relationships between body mass index, smoking and lung cancer: univariable and multivariable Mendelian randomization. Int J Cancer 48(5):1077–1086. https://doi.org/10.1002/ijc.33292
Liu M, Jiang Y, Wedow R, Li Y, Brazel DM, et al (2019) Association studies of up to 1.2 million individuals yield new insights into the genetic etiology of tobacco and alcohol use. Nat Genet 51(2):237–244. https://doi.org/10.1038/s41588-018-0307-5
Kurki MI, Karjalainen J, Palta P, Sipilä TP, Kristiansson K et al (2023) FinnGen provides genetic insights from a well-phenotyped isolated population. Nature 613(7944):508–518. https://doi.org/10.1038/s41586-022-05473-8
Boef AG, Dekkers OM, le Cessie S (2015) Mendelian randomization studies: a review of the approaches used and the quality of reporting. Int J Epidemiol 44(2):496–511. https://doi.org/10.1093/ije/dyv071
Staley JR, Blackshaw J, Kamat MA, Ellis S, Surendran P, Sun BB (2016) PhenoScanner: a database of human genotype-phenotype associations. Bioinformatics 32(20):3207–3209. https://doi.org/10.1093/bioinformatics/btw373
Burgess S, Thompson SG, Genetics Collaboration CRPCHD (2011) Avoiding bias from weak instruments in Mendelian randomization studies. Int J Epidemiol 40(3):755–764. https://doi.org/10.1093/ije/dyr036
Burgess S, Dudbridge F, Thompson SG (2016) Combining information on multiple instrumental variables in Mendelian randomization: comparison of allele score and summarized data methods. Stat Med 35(11):1880–1906. https://doi.org/10.1002/sim.6835
Bowden J, Davey Smith G, Haycock PC, Burgess S (2016) Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol 40(4):304–314. https://doi.org/10.1002/gepi.21965
Bowden J, Davey Smith G, Burgess S (2015) Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol 44(2):512–525. https://doi.org/10.1093/ije/dyv080
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Bowden J, Spiller W, Del Greco MF, Sheehan N, Thompson J et al (2018) Improving the visualization, interpretation and analysis of two-sample summary data mendelian randomization via the radial plot and radial regression. Int J Epidemiol 47(4):1264–1278. https://doi.org/10.1093/ije/dyy101
Bowden J, Hemani G, Davey Smith G (2018) Invited commentary: detecting individual and global horizontal pleiotropy in mendelian randomization-a job for the humble heterogeneity statistic? Am J Epidemiol 187(12):2681–2685. https://doi.org/10.1093/aje/kwy185
Rees JMB, Wood AM, Burgess S (2017) Extending the MR-egger method for multivariable mendelian randomization to correct for both measured and unmeasured pleiotropy. Stat Med 36(29):4705–4718. https://doi.org/10.1002/sim.7492
Cheng H, Garrick DJ, Fernando RL (2017) Efficient strategies for leave-one-out cross validation for genomic best linear unbiased prediction. J Anim Sci Biotechnol 8:38. https://doi.org/10.1186/s40104-017-0164-6
Sorensen LT, Friis E, Jorgensen T, Vennits B, Andersen BR et al (2002) Smoking is a risk factor for recurrence of groin hernia. World J Surg 26(4):397–400. https://doi.org/10.1007/s00268-001-0238-6
Sørensen LT, Hemmingsen UB, Kirkeby LT, Kallehave F, Jørgensen LN (2005) Smoking is a risk factor for incisional hernia. Arch Surg 140(2):119–123. https://doi.org/10.1001/archsurg.140.2.119
Soppe S, Slieker S, Keerl A, Muller MK, Wirsching A et al (2022) Emergency repair and smoking predict recurrence in a large cohort of ventral hernia patients. Hernia 26(5):1337–1345. https://doi.org/10.1007/s10029-022-02672-0
Cannon DJ, Read RC (1981) Metastatic emphysema: a mechanism for acquiring inguinal herniation. Ann Surg 194(3):270–278. https://doi.org/10.1097/00000658-198109000-00004
Kim C, Ouyang W, Dass C, Zhao H, Criner GJ (2016) Hiatal Hernia on Chest High-Resolution Computed Tomography and Exacerbation Rates in COPD Individuals. Chronic Obstr Pulm Dis 3(2):570–579. https://doi.org/10.15326/jcopdf.3.2.2015.0158
Quiroga-Centeno AC, Quiroga-Centeno CA, Guerrero-Macías S, Navas-Quintero O, Gómez-Ochoa SA (2022) Systematic review and meta-analysis of risk factors for Mesh infection following Abdominal Wall Hernia Repair Surgery. Am J Surg 224(1 Pt A):239–246. https://doi.org/10.1016/j.amjsurg.2021.12.024
Jensen JA, Goodson WH, Hopf HW, Hunt TK (1991) Cigarette smoking decreases tissue oxygen. Arch Surg 126(9):1131–1134. https://doi.org/10.1001/archsurg.1991.01410330093013
Allen DB, Maguire JJ, Mahdavian M, Wicke C, Marcocci L et al (1997) Wound hypoxia and acidosis limit neutrophil bacterial killing mechanisms. Arch Surg 132(9):991–996. https://doi.org/10.1001/archsurg.1997.01430330057009
Thomsen T, Villebro N, Møller AM (2014) Interventions for preoperative smoking cessation. Cochrane Database Syst Rev 2014(3):CD002294. https://doi.org/10.1002/14651858.CD002294.pub4
de Goede B, Timmermans L, van Kempen BJ, van Rooij FJ, Kazemier G et al (2015) Risk factors for inguinal hernia in middle-aged and elderly men: results from the Rotterdam Study. Surgery 157(3):540–546. https://doi.org/10.1016/j.surg.2014.09.029
Ellis H (1986) Diaphragmatic hernia–a diagnostic challenge. Postgrad Med J 62(727):325–327. https://doi.org/10.1136/pgmj.62.727.325
Kahrilas PJ, Gupta RR (1990) Mechanisms of acid reflux associated with cigarette smoking. Gut 31(1):4–10. https://doi.org/10.1136/gut.31.1.4
Wu AH, Tseng CC, Bernstein L (2003) Hiatal hernia, reflux symptoms, body size, and risk of esophageal and gastric adenocarcinoma. Cancer 98(5):940–948. https://doi.org/10.1002/cncr.11568
Chen JX, Hsu SY, Lin MC, Shih PK (2022) Risk of diaphragmatic hernia in patients with spontaneous pneumothorax. BMC Pulm Med 22(1):347. https://doi.org/10.1186/s12890-022-02147-z
Henriksen NA (2016) Systemic and local collagen turnover in hernia patients. Dan Med J 63(7):B5265
Finn J, Suhl J, Kancherla V, Conway KM, Oleson J et al (2022) Maternal cigarette smoking and alcohol consumption and congenital diaphragmatic hernia. Birth Defects Res 114(13):746–758. https://doi.org/10.1002/bdr2.2059
Kardon G, Ackerman KG, McCulley DJ, Shen Y, Wynn J et al (2017) Congenital diaphragmatic hernias: from genes to mechanisms to therapies. Dis Model Mech 10(8):955–970. https://doi.org/10.1242/dmm.028365
Acknowledgements
We want to acknowledge the participants and investigators of the FinnGen Study and GWAS and Sequencing Consortium of Alcohol and Nicotine Use for providing the data publicly.
Funding
The present study received no funding.
Author information
Authors and Affiliations
Contributions
Conceptualization, QW and YG; formal analysis and investigation, QW and WD; writing—original draft preparation: QW, writing—review and editing: ZS and JY; resources: DY; supervision: YG, All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Institutional review board statement
Not applicable.
Ethical approval
Not applicable.
Human and animal rights
Not applicable.
Informed consent
Informed consent was obtained from all the subjects involved in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, Q., Yang, D., Dong, W. et al. Causal relationship between cigarette smoking behaviors and the risk of hernias: a Mendelian randomization study. Hernia 28, 435–446 (2024). https://doi.org/10.1007/s10029-023-02925-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-023-02925-6