Abstract
Purpose
Chronic post-operative inguinal pain (CPIP) following inguinal hernia repair has been a major sequela affecting 4000–48000 patients annually. Optimal management of CPIP has been a challenge, and pharmacological management particularly with opioids has shown unsatisfactory results. The main objective of this systematic review is to investigate the safety and efficacy of neuromodulation as an alternative intervention for the management of post-operative inguinal pain.
Methods
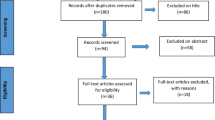
A literature search was conducted by three reviewers to identify all relevant studies on the use of neuromodulatory interventions for treating post-operative inguinal pain. Data on study characteristics, neuromodulatory modalities, and patient’s clinical data such as pre/post-interventional pain scores and analgesic requirements were extracted and reported.
Results
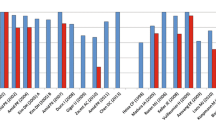
A total of 389 patients with 357 (95.9%) males and 15 (4.1%) females were evaluated. The mean age of study participants was 47.9 ± 10.4 years. There were 187 (48.1%) and 202 (51.9%) patients allocated to the control and trial groups, respectively. The most common neuromodulation modality was TENS (4, 36.4%), followed by SCS (3, 27.3%), PNS (3, 27.3%), and acupuncture-assisted (2, 18.2%). The overall mean follow-up duration of the entire cohort was 3.8 months. The mean difference between pre-operative and post-operative VAS scores in the trial groups was 4.65 (95% Confidence Interval [CI], 2.97, 6.33), which was statistically significant (P value < 0.05). Patient-reported outcome measures showed significant responsiveness toward their treatments.
Conclusion
Nerve stimulation, in its many forms, is a safe and feasible option for the management of post-operative inguinal pain.




Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article. SPSS data of the participant can be requested from the authors. Please write to the corresponding author if you are interested in such data.
References:
Lichtenstein IL, Shulman AG, Amid PK, Montllor MM (1989) The tension-free hernioplasty. Am J Surg 157(2):188–193. https://doi.org/10.1016/0002-9610(89)90526-6
EU Hernia Trialists Collaboration (2022) Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg 235(3):322. https://doi.org/10.1097/00000658-200203000-00003
Nguyen DK, Amid PK, Chen DC (2016) Groin pain after inguinal hernia repair. Adv Surg 50(1):203–220. https://doi.org/10.1016/j.yasu.2016.04.003
Landry M, Lewis R, Lew M, Forman B, Heidel E, Ramshaw B (2020) Evaluating effectiveness of cognitive behavioral therapy within multimodal treatment for chronic groin pain after inguinal hernia repair. Surg Endosc 34:3145–3152. https://doi.org/10.1007/s00464-019-07082-5
Loos MJA, Roumen RMH, Scheltinga MRM (2007) Classifying postherniorrhaphy pain syndromes following elective inguinal hernia repair. World J Surg 31(9):1760–1765. https://doi.org/10.1007/s00268-007-9121-4
Amid P (2004) Causes, prevention, and surgical treatment of postherniorrhaphy neuropathic inguinodynia: triple neurectomy with proximal end implantation. Hernia 8(4):343–349. https://doi.org/10.1007/s10029-004-0247-0
Treede RD, Jensen TS, Campbell JN et al (2008) Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology 70(18):1630–1635. https://doi.org/10.1212/01.wnl.0000282763.29778.59
Brummett CM, Waljee JF, Goesling J et al (2017) New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg 152(6):e170504–e170504. https://doi.org/10.1001/jamasurg.2017.0504
Oderda GM, Said Q, Evans RS et al (2007) Opioid-related adverse drug events in surgical hospitalizations: impact on costs and length of stay. Ann Pharmacother 41(3):400–407. https://doi.org/10.1345/aph.1H386
Urman RD, Seger DL, Fiskio JM et al (2021) The burden of opioid-related adverse drug events on hospitalized previously opioid-free surgical patients. J Patient Saf 17(2):e76–e83. https://doi.org/10.1097/PTS.0000000000000566
Melzack R, Wall PD (1965) Pain mechanisms: a new theory: a gate control system modulates sensory input from the skin before it evokes pain perception and response. Science 150(3699):971–979. https://doi.org/10.1126/science.150.3699.971
Bananzadeh A, Sohooli M, Shamsi T, Darabi M, Shahriarirad R, Shekouhi R (2022) Effects of neuromodulation on treatment of recurrent anal fissure: A systematic review. Int J Surg 102:106661. https://doi.org/10.1016/j.ijsu.2022.106661
HerniaSurge Group (2018) International guidelines for groin hernia management. Hernia 22(1):1–165. https://doi.org/10.1007/s10029-017-1668-x
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev 10(1):1–11. https://doi.org/10.1186/s13643-021-01626-4
Shea BJ, Reeves BC, Wells G et al (2017) AMSTAR: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. https://doi.org/10.1136/bmj.j4008
Health, N.I.O (2017) Quality assessment tool for case series studies. National Heart, Lung, and Blood Institute, Cham
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. https://doi.org/10.1136/bmj.l4898
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res 27(6):1785–1805. https://doi.org/10.1177/0962280216669183
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5(1):1–10. https://doi.org/10.1186/1471-2288-5-13
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14:1–13. https://doi.org/10.1186/1471-2288-14-135
Stinson LW Jr, Roderer GT, Cross NE, Davis BE (2001) Peripheral subcutaneous electrostimulation for control of intractable post-operative inguinal pain: a case report series. Neuromodulation 4(3):99–104. https://doi.org/10.1046/j.1525-1403.2001.00099.x
DeSantana JM, Santana-Filho VJ, Guerra DR, Sluka KA, Gurgel RQ, da Silva WM, Jr., (2008) Hypoalgesic effect of the transcutaneous electrical nerve stimulation following inguinal herniorrhaphy: a randomized, controlled trial. J Pain 9(7):623–629. https://doi.org/10.1016/j.jpain.2008.01.337
Yakovlev AE, Al Tamimi M, Barolat G, Karasev SA, Merkulov YA, Resch BE, Yakovleva VE (2010) Spinal cord stimulation as alternative treatment for chronic post-herniorrhaphy pain. Neuromodulation 13(4):288–291. https://doi.org/10.1111/j.1525-1403.2010.00276.x
Dias M, Carneiro NM, Guerra LA et al (2010) Effects of electroacupuncture on local anaesthesia for inguinal hernia repair: a randomised placebo-controlled trial. Acupunct Med 28(2):65–70. https://doi.org/10.1136/aim.2009.000570
Ahmed MT (2010) Effect of transcutaneous electrical nerve stimulation on postoperative pain after inguinal hernia repair: a randomized placebo-controlled trial. Turk J Phys Med 56:170–176. https://doi.org/10.4274/tftr.56.170
Lepski G, Vahedi P, Tatagiba MS, Morgalla M (2013) Combined spinal cord and peripheral nerve field stimulation for persistent post-herniorrhaphy pain. Neuromodulation 16(1):84–89. https://doi.org/10.1111/j.1525-1403.2012.00463.x
Schu S, Gulve A, ElDabe S et al (2015) Spinal cord stimulation of the dorsal root ganglion for groin pain—a retrospective review. Pain Pract 15(4):293–299. https://doi.org/10.1111/papr.12194
Voorbrood CE, Burgmans JP, Van Dalen T, Breel J, Clevers GJ, Wille F, Simmermacher RK (2015) An algorithm for assessment and treatment of postherniorrhaphy pain. Hernia 19:571–577. https://doi.org/10.1007/s10029-015-1387-0
Eidy M, Fazel MR, Janzamini M, Haji Rezaei M, Moravveji AR (2016) Preemptive analgesic effects of transcutaneous electrical nerve stimulation (TENS) on postoperative pain: a randomized, double-blind, placebo-controlled trial. Iran Red Crescent Med J. https://doi.org/10.5812/ircmj.35050
Szmit M, Agrawal S, Goździk W et al (2021) Transcutaneous electrical acupoint stimulation reduces postoperative analgesic requirement in patients undergoing inguinal hernia repair: a randomized, placebo-controlled study. J Clin Med 10(1):146. https://doi.org/10.3390/jcm10010146
Parseliunas A, Paskauskas S, Simatoniene V, Kubiliute E, Dainius E, Subocius A, Venclauskas L, Venskutonis D (2022) Transcutaneous electric nerve stimulation reduces pathological sensation of mesh one week after open inguinal hernia surgery: follow-up results from a randomized, double blind and placebo-controlled trial. Medicina (Kaunas) 58(6):725. https://doi.org/10.3390/medicina58060725
Parseliunas A, Paskauskas S, Kubiliute E, Vaitekunas J, Venskutonis D (2021) Transcutaneous electric nerve stimulation reduces acute postoperative pain and analgesic use after open inguinal hernia surgery: a randomized, double-blind, placebo-controlled trial. J Pain 22(5):533–544. https://doi.org/10.1016/j.jpain.2020.11.006
Fränneby U, Sandblom G, Nordin P, Nyrén O, Gunnarsson U (2006) Risk factors for long-term pain after hernia surgery. Ann Surg 244(2):212–219. https://doi.org/10.1097/01.sla.0000218081.53940.01
Hansen MB, Andersen KG, Crawford ME (2010) Pain following the repair of an abdominal hernia. Surg Today 40:8–21. https://doi.org/10.1007/s00595-009-4001-1
Callesen T (2003) Inguinal hernia repair: anaesthesia, pain and convalescence. Dan Med Bull 50(3):203–218
Bay-Nielsen M, Perkins FM, Kehlet H, Danish Hernia Database (2001) Pain and functional impairment 1 year after inguinal herniorrhaphy: a nationwide questionnaire study. Ann Surg 233(1):1. https://doi.org/10.1097/00000658-200101000-00001
Loos MJ, Roumen RM, Scheltinga MR (2007) Chronic sequelae of common elective groin hernia repair. Hernia 11:169–173. https://doi.org/10.1007/s10029-007-0196-5
Florence CS, Zhou C, Luo F, Xu L (2016) The economic burden of prescription opioid overdose, abuse and dependence in the United States, 2013. Med Care 54(10):901. https://doi.org/10.1097/MLR.0000000000000625
Bates C, Laciak R, Southwick A, Bishoff J (2011) Overprescription of postoperative narcotics: a look at postoperative pain medication delivery, consumption and disposal in urological practice. J Urol 185(2):551–555. https://doi.org/10.1016/j.juro.2010.09.088
Bateman BT, Franklin JM, Bykov K et al (2016) Persistent opioid use following cesarean delivery: patterns and predictors among opioid-naïve women. Am J Obstet Gynecol 215(3):353.e1-353.e18. https://doi.org/10.1016/j.ajog.2016.03.016
Verhagen T, Loos MJA, Scheltinga MRM, Roumen RMH (2018) The groinpain trial: a randomized controlled trial of injection therapy versus neurectomy for postherniorraphy inguinal neuralgia. Ann Surg 267(5):841–845. https://doi.org/10.1097/SLA.0000000000002274
Khoshmohabat H, Panahi F, Alvandi AA, Mehrvarz S, Mohebi HA, Shams KE (2012) Effect of ilioinguinal neurectomy on chronic pain following herniorrhaphy. Trauma Mon 17(3):323. https://doi.org/10.5812/traumamon.6581
Sluka KA, Deacon M, Stibal A, Strissel S, Terpstra A (1999) Spinal blockade of opioid receptors prevents the analgesia produced by TENS in arthritic rats. J Pharmacol Exp Ther 289(2):840–846
Kalra A, Urban MO, Sluka KA (2001) Blockade of opioid receptors in rostral ventral medulla prevents antihyperalgesia produced by transcutaneous electrical nerve stimulation (TENS). J J Pharmacol Exp Ther 298(1):257–263
Chandran P, Sluka KA (2003) Development of opioid tolerance with repeated transcutaneous electrical nerve stimulation administration. Pain 102(1–2):195–201. https://doi.org/10.1016/s0304-3959(02)00381-0
Pourmokhtari M, Shahriarirad R, Shekouhi R (2023) Effectiveness of transcutaneous electrical nerve stimulation alongside quadriceps exercise in the correction of soccer genu varum in adolescents 14–18 years old: a randomized controlled trial. Sport Sci Health. https://doi.org/10.1007/s11332-023-01049-9
Funding
No funding was obtained for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Compliance with ethical standards, Human and animal rights
This article is fully compliant with ethical standards, human, and animal rights.
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shekouhi, R., Farz, F., Sohooli, M. et al. Investigating the safety and efficacy of nerve stimulation for management of groin pain after surgical herniorrhaphy: a systematic review and meta-analysis. Hernia 27, 1363–1373 (2023). https://doi.org/10.1007/s10029-023-02861-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10029-023-02861-5