ABSTRACT
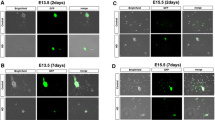
Intestinal aganglionosis results from homologous genetic defects in humans and mice, including mutations of Sox10, which encodes a transcription factor expressed in neural crest cells. To gain insight into the embryological basis for this condition, the phenotype and pathogenesis of intestinal aganglionosis in Sox10 Dom/Sox10 Dom embryos were studied. The distribution of enteric neural precursors and other neural crest derivatives in Sox10 Dom/Sox10 Dom embryos was analyzed with immunochemical and transgenic markers. The ability of wild-type neural crest cells to colonize Sox10 Dom/Sox10 Dom intestinal explants was evaluated by appositional grafts under the renal capsule. Apoptosis was studied by TUNEL labeling. Sox10 Dom/Sox10 Dom embryos died pre- or perinatally with total enteric aganglionosis and hypoplasia or agenesis of nonenteric ganglia. Mutant crest cells failed to colonize any portion of the Sox10 Dom/Sox10 Dom gut, but wild-type neural crest cells were able to colonize explanted segments of Sox10 Dom/Sox10 Dom embryonic intestine. In Sox10 Dom/Sox10 Dom embryos, apoptosis was increased in sites of early neural crest cell development, before these cells enter the gut. Sox10 Dom/Sox10 Dom embryos are one of many genetic animal models for human Hirschsprung disease. The underlying problem is probably not the enteric microenvironment, since Sox10 Dom/Sox10 Dom intestine supports colonization and neuronal differentiation by wild-type neural crest cells. Instead, excessive cell death occurs in mutant neural crest cells early in their migratory pathway. Comparison with other models suggests that genetic heterogeneity of aganglionosis correlates with different pathogenetic mechanisms.
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received January 8, 1999; accepted March 16, 1999.
Rights and permissions
About this article
Cite this article
Kapur, R. Early Death of Neural Crest Cells Is Responsible for Total Enteric Aganglionosis in Sox10 Dom/Sox10 Dom Mouse Embryos. Pediatr. Dev. Pathol. 2, 559–569 (1999). https://doi.org/10.1007/s100249900162
Issue Date:
DOI: https://doi.org/10.1007/s100249900162