Abstract
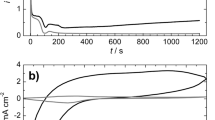
The electrochemical synthesis of polyaniline on a titanium surface in aqueous sulfuric acid solutions with various concentrations of added aniline has been investigated by cyclic voltammetry. By utilizing a more cathodic potential range (up to −0.6 V) for the cyclization than is usual (up to −0.2 V) on Pt and Au electrodes, the new voltammetric waves have been deconvoluted from the already well-known ones for polyaniline.
By simultaneous electrochemical and in situ Raman spectroscopic measurements, the Raman bands of polyaniline electrodeposited on a Ti electrode, were assigned for potentials of −0.15 V and −0.6 V. It was found that the new monitored waves were closely related to the so-called “middle” peaks and appear only when the polyaniline reaches an overoxidized state.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 7 August 1997 / Accepted: 4 November 1997
Rights and permissions
About this article
Cite this article
Arsov, L. Electrochemical study of polyaniline deposited on a titanium surface. J Solid State Electrochem 2, 266–272 (1998). https://doi.org/10.1007/s100080050099
Issue Date:
DOI: https://doi.org/10.1007/s100080050099