Abstract
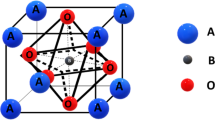
Perovskite-type compounds, Li x La(1− x )/3NbO3 and (Li0.25La0.25)1− x Sr0.5 x NbO3 as lithium ionic conductors, were synthesized by a solid-state reaction. From powder X-ray diffraction, the solid solution ranges of the two compounds were determined to be 0≤x≤0.25 and 0≤x≤0.125, respectively. In the Li x La(1− x )/3NbO3 system, the ionic conductivity of lithium at room temperature, σ25, exhibited a maximum value of 4.7 × 10−5 S · cm−1 at x = 0.10. However, because of the decrease in the lattice parameters with increasing Li concentration x˙, σ25 of the samples decreased with increasing x from 0.10 to 0.25. Also, in the (Li0.25La0.25)1− x Sr0.5 x NbO3 system, the lattice parameter increased with the increase of Sr concentration and the σ25 achieved a maximum (7.3 × 10−5 S · cm−1 at 25 °C) at x = 0.125.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 12 September 1997 / Accepted: 15 November 1997
Rights and permissions
About this article
Cite this article
Kawakami, Y., Ikuta, H. & Wakihara, M. Ionic conduction of lithium for Perovskite-type compounds, Li x La(1− x )/3NbO3 and (Li0.25La0.25)1− x Sr0.5 x NbO3 . J Solid State Electrochem 2, 206–210 (1998). https://doi.org/10.1007/s100080050089
Issue Date:
DOI: https://doi.org/10.1007/s100080050089