Abstract
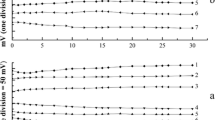
A new method is described for the determination of ammonium in aqueous solutions with electrodes modified by Prussian blue (PB). The specific voltammetric response of PB-modified electrodes to ammonium ions is used for their analytical determination. In the presence of ammonium ions, a concentration-dependent inhibition of the low-spin iron(II/III) system of PB occurs. Only thallium and rubidium ions cause similar inhibition. A useful electrochemical determination method is thus available for detecting ammonium ions in the presence of frequently interfering potassium and sodium ions. Paraffin-impregnated graphite electrodes modified with a mechanically transferred PB layer and bulk-modified PB-composite electrodes are studied. The method is applicable within a concentration range which extends from 4 × 10−5 mol/l to 10−2 mol/l NH4 +. The composite electrode is used in an electrochemical flow-through system in conjunction with the Kjeldahl method.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 21 April 1997 / Accepted: 28 May 1997
Rights and permissions
About this article
Cite this article
Hermes, M., Scholz, F. The electrochemical determination of ammonium based on the selective inhibition of the low-spin iron(II)/(III) system of Prussian blue. J Solid State Electrochem 1, 215–220 (1997). https://doi.org/10.1007/s100080050051
Issue Date:
DOI: https://doi.org/10.1007/s100080050051