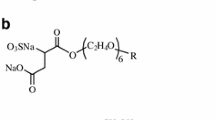
Abstract
The function Δ(ΔG A 0), which is the difference of Gibbs energies characterizing surface-active substance (surfactant, SAS) adsorption at metal/solution and air/solution surfaces, has been introduced. The equation connecting the function Δ(ΔG A 0) with SAS ionization potential has been obtained using the elementary theory of donor-acceptor interactions. Published experimental data on SAS adsorption at mercury, bismuth and gold have been used for Δ(ΔG A 0) calculation. The dependence of Δ(ΔG A 0) on ionization potentials can be described by an equation derived in this work. It has been demonstrated that the value of the hydrophilicity of gold is much higher than the values for mercury and bismuth. The lifetime of SAS molecules at a metal surface has been estimated. The question of the possibility of theoretica l estimation of standard energies ΔG A 0 characterizing SAS adsorption at a metal/solution surface has been discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 9 December 1996 / Accepted: 13 January 1997
Rights and permissions
About this article
Cite this article
Afanasyev, B., Akulova, Y. & (Balobanova), M. Dependence of the energy of surface-active substances/metal interaction on their ionization potentials. Evaluation of hydrophilicity of metal. J Solid State Electrochem 1, 68–76 (1997). https://doi.org/10.1007/s100080050024
Issue Date:
DOI: https://doi.org/10.1007/s100080050024