Abstract.
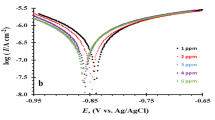
The mechanism of pit growth of pure aluminum (Al) in sulfate ion (SO4 2–)- or nitrate ion (NO3 –)-containing 0.1 M sodium chloride solutions has been studied in terms of the morphological changes of artificial pits using potentiodynamic polarization experiment, potentiostatic current transient technique and optical microscopy. The increase in SO4 2– and NO3 – ion concentrations in NaCl solution raised the pitting potential E pit of pure Al, which is ascribed to the impediment to pit initiation on pure Al by the addition of SO4 2– or NO3 – ions. From the potentiostatic current transients of artificial pits in aqueous 0.1 M NaCl solution, the average value of the pit current was observed to increase with increasing SO4 2– ion concentration, whereas that value of the pit current in the presence of NO3 – ions increased up to ca. 0.4 M NO3 – ion concentration and then decreased abruptly with increasing NO3 – ion concentration. From observations of the morphologies of the pits, it appears that the pit grows preferentially in the lateral direction or in the downward direction by adding SO4 2– or NO3 – ions to aqueous 0.1 M NaCl solution, respectively. Based upon the experimental results, two different pit growth mechanisms by anion additives can be proposed: (1) pit growth by the preferential attack of both SO4 2– and Cl– to the pit wall in SO4 2–-containing solutions; (2) pit growth by the creation of an aggressive environment at the pit bottom up to 0.4 M NO3 – ion concentration due to the lower mobility of NO3 – than Cl– in NO3 –-containing solutions.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Pyun, SI., Na, KH. & Park, JJ. Morphological studies of the mechanism of pit growth of pure aluminum in sulfate ion- or nitrate ion-containing 0.1 M NaCl solutions. J Solid State Electrochem 5, 473–478 (2001). https://doi.org/10.1007/s100080000191
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s100080000191