Abstract.
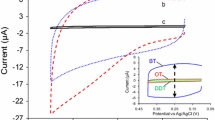
Three thiols with three aromatic rings and different structure – terphenyl-4-methanethiol (TPMT), terphenyl-4-thiol (TPT), and anthracene-2-thiol (AT) – have been used to form self-assembled monolayers (SAM) on vapour-deposited and flame-annealed Au films on glass substrates. All three SAMs effectively block the anodic formation of Au oxide, indicating densely packed layers which prevent the access of water and hydrated ions through the organic layer to the metal surface. The film improves its inhibiting properties with duration of exposure to the thiol solutions, reaching completion after 1 hour [1]. The charge-transfer reaction of the Fe(CN)6 3–/Fe(CN)6 4– system is blocked for TPMT films with an insulation of the π-electron system from the Au surface by the methylene group. TPT and especially AT films show the current density of the redox reactions. It is proposed that the charge transfer occurs via the aromatic molecules of the SAMs to the Au surface.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Scharf, J., Strehblow, HH., Zeysing, B. et al. Electrochemical and surface analytical studies of self-assembled monolayers of three aromatic thiols on gold electrodes. J Solid State Electrochem 5, 396–401 (2001). https://doi.org/10.1007/s100080000167
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s100080000167