Abstract
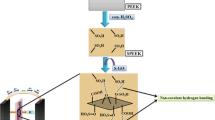
In this study, graphene (G) was blended with sulfonated polyether sulfone octyl sulfonamide (SPESOS) in different ratios (1, 1.5, and 2 wt.%) to enhance the proton conductivity of SPESOS. The results of the water absorption analysis and current impedance spectroscopy of a G/SPESOS hybrid film showed that the proton conductivity improved due to the hydrophilic-hydrophobic nature of the hybrid. Further analysis showed that the synthesized hybrid film had a desirable contact angle and good ion exchange capacity, water absorption, and thermal stability. The hybrid film with 2 wt.% graphene showed better proton conductivity (92 mS/cm) than the pristine SPESOS film (34 mS/cm) at 100% relative humidity. These results suggested that the synthesized hybrid films offered superior proton conductivity, ion exchange capacity, and hydrophilicity compared to pristine SPESOS.






Similar content being viewed by others
References
Sahu AK, Ketpang K, Shanmugam S, Kwon O, Lee SC, Kim H (2016) Sulfonated graphene-Nafion hybrid films for polymer electrolyte fuel cells operating under reduced relative humidity. J Phys Chem C120(29):15855–15866. https://doi.org/10.1021/acs.jpcc.5b11674
Vinothkannan M, Kim AR, Nahm KS, Yoo DJ (2016) Ternary hybrid (SPEEK/SPVdF-HFP/GO) based film electrolyte for the applications of fuel cells: profile of improved mechanical strength, thermal stability and proton conductivity. RSC Adv 6(110):108851–108863. https://doi.org/10.1039/C6RA22295A
Kim AR, Vinothkannan M,Yoo DJ (2018) Sulfonated fluorinated multi-block copolymer hybrid containing sulfonated(poly ether ether ketone) and graphene oxide: a ternary hybrid film architecture for electrolyte applications in proton exchange film fuel cells. J Energy Chem 27(4):1247–1260. https://doi.org/10.1016/j.jechem.2018.02.020
Wu H, Peng T, Kou Z, Cheng K, Zhang J, Zhang J, Meng T, Mu S (2017) In situ constructing of ultrastableceramic@graphene core-shell architectures as advanced metal catalyst supports toward oxygen reduction. J Energy Chem 26(6):1160–1167. https://doi.org/10.1016/j.jechem.2017.08.012
Tian XL, Wang L, Deng P, Chen Y, Xia BY (2017) Research advances in unsupported Pt-based catalysts for electrochemical methanol oxidation. J Energy Chem 26(6):1067–1076. https://doi.org/10.1016/j.jechem.2017.10.009
Neelakandan S, Kaleekkal NJ, Kanagaraj P, Sabarathinam RM, Muthumeenal A, Nagendran A (2016) Effect of sulfonated graphene oxide on the performance enhancement of acid-base hybrid films for direct methanol fuel cells. RSC Adv 6(57):51599–51608. https://doi.org/10.1039/C5RA27655A
Kim AR, Vinothkannan M, Yoo DJ (2017) Sulfonated-fluorinated copolymer blending films containing SPEEK for use as the electrolyte in polymer electrolyte fuel cell (PEFC). Int J Hydrogen Energy 42(7):4349–4365. https://doi.org/10.1016/j.ijhydene.2016.11.161
Park CK, Lee CH, Guiver MD, Lee YM (2011) Sulfonated hydrocarbon films for medium-temperature and low-humidity proton exchange film fuel cells (PEMFCs). Prog Polymer Sci 36(11):1443–1498. https://doi.org/10.1016/j.progpolymsci.2011.06.001
Kreuer KD (2001) On the development of proton conducting polymer films for hydrogen and methanol fuel cells. J Membrane Sci 185(1):29–39. https://doi.org/10.1016/S0376-7388(00)00632-3
Xing P, Robertson GP, Guiver MD, Mikhailenko SD, Kaliaguine S (2005) Synthesis and characterization of poly(aryl ether ketone) copolymers containing (hexafluoroisopropylidene)-diphenol moiety as proton exchange film materials. Polymer 46(10):3257–3263. https://doi.org/10.1016/j.polymer.2005.03.003
Xing P, Robertson GP, Guiver MD, Mikhailenko SD, Wang K, Kaliaguine S (2004) Synthesis and characterization of sulfonated poly(ether ether ketone)forproton exchange films. J Film Sci 229(2):95–106. https://doi.org/10.1016/j.memsci.2003.09.019
Kim YS, Einsla B, Sankir M, Harrison W, Pivovar BS (2006) Structure–property–performance relationships of sulfonated poly(arylene ether sulfone)s as apolymer electrolyte for fuel cell applications. Polymer 47(11):4026–4035. https://doi.org/10.1016/j.polymer.2006.02.032
Mabrouk W, Ogier L, Matoussi F, Sollogoub C, Vidal S, Dachraoui M, Fauvarque JF (2011) Preparation of new proton exchange films using sulfonated poly (ether sulfone) modified by octylamine (SPESOS). Mater Chem Phys 128(3):456–463. https://doi.org/10.1016/j.matchemphys.2011.03.031
Ahmed Z, Charradi K, Alsulami QA, Keshk SMAS, Chtourou R (2021) Physicochemical characterization of low sulfonated polyether ether ketone/smectite clay hybrid for proton exchange film fuel cells. J Appl Polym Sci 138(1):e49634. https://doi.org/10.1002/app.49634
Charradi K, Ahmed Z, Thmaini N, Aranda P, Al-Ghamdi YO, Ocon P, Keshk SAMS, Chtourou R (2021) Incorporation of layered double hydroxide/sepiolite to improve the performance of sulfonated poly(ether ether ketone) hybridfilms for proton exchange film fuel cells. J Appl Polym Sci 138(19):e50364. https://doi.org/10.1002/app.50364
Mabrouk W, Charradi K, Lafi R, AlSalem HS, Meherzi HM, Keshk SAMS (2022) Incorporation of sepiolite clay within sulfonated polyether sulfone octyl sulfonamide for the augmentation of proton conductivity. J Mater Sci 57(140):15331–15339. https://doi.org/10.1007/s10853-022-07627-5
Mabrouk W, Charradi K, Meherzi HM, Alhussein A, Keshk SAMS (2022) Proton conductivity amelioration of sulfonated poly ether sulfone octyl sulfonamide via the incorporation of Montmorillonite. J Electron Mater 51(139):6369–6378. https://doi.org/10.1007/s11664-022-09862-7
Pushparaj VL, Shaijumon MM, Kumar A, Murugesan S, Ci L, Vajtai R, Linhardt RJ, Nalamasu O, Ajayan PM (2007) Flexible energy storage devices based on nanohybrid paper. Proc Natl Acad Sci 104(39):13574–13577. https://doi.org/10.1073/pnas.070650810
Chen J, Minett AI, Liu Y, Lynam C, Sherrell P, Wang C, Wallace GG (2008) Direct growth of flexible carbon nanotube electrodes. Adv Mater 20(3):566–570. https://doi.org/10.1002/adma.200701146
Chou SL, Wang JZ, Chew SY, Liu HK, Dou SX (2008) Electrodeposition of MnO2 nanowires on carbon nanotube paper as free-standing, flexible electrode for supercapacitors. Electrochem Commun 10(11):1724–1727. https://doi.org/10.1016/j.elecom.2008.08.051
Mathew E, Manoj B (2022) Disorders in graphene: types, effects and control techniques-a review. Carbon Lett 32(240):431–450. https://doi.org/10.1007/s42823-021-00289-4
Yoo E, Kim J, Hosono E, Zhou H, Kudo T, Honma I (2008) Large reversible Li storage of graphene nanosheet families for use in rechargeable lithium ion batteries. Nano Lett 8(8):2277–2282. https://doi.org/10.1021/nl800957b
Yin H, Zhang C, Liu F, Hou Y (2014) Hybrid of iron nitride and nitrogen-doped graphene aerogel as synergistic catalyst for oxygen reduction reaction. Adv Funct Mater 24(20):2930–2937. https://doi.org/10.1002/adfm.201303902
Ye YS, Cheng MY, Xie XL, Rick J, Huang YJ, Chang FC, Hwang BJ (2013) Alkali doped polyvinyl alcohol/graphene electrolyte for direct methanol alkaline fuel cells. J Power Sources 239:424–432. https://doi.org/10.1016/j.jpowsour.2013.03.021
Wang S, Yu D, Dai L, Chang DW, Baek JB (2011) Polyelectrolyte-functionalizedgraphene as metal-free electrocatalysts for oxygen reduction. ACS Nano 5(8):6202–6209. https://doi.org/10.1021/nn200879h
Yong YC, Dong XC, Chan-Park MB, Song H, Chen P (2012) Macroporous and monolithic-anode based on polyaniline hybridized three-dimensional graphene for high-performance microbial fuel cells. ACS Nano 6(3):2394–2400. https://doi.org/10.1021/nn204656d
Dai W, Yu L, Li Z, Yan J, Liu L, Xi J, Qiu X (2014) Sulfonated poly(ether ether ketone)/graphene hybrid film for vanadium redox flow battery. Electrochi Acta 132:200–207. https://doi.org/10.1016/j.electacta.2014.03.156
Xiaobing H, Hao K, Tao C, Jie G, Yuan Z, Sang Y, Guowen H (2021) Effect of π–π stacking interfacial interaction on the properties of graphene/poly(styrene-b-isoprene-b-styrene) hybrids. Nanomaterials 11(9):2158. https://doi.org/10.3390/nano11092158
Choi E, Han TH, Hong J, Kim J, Lee SH, Kima HW (2010) Noncovalent functionalization of graphene with end-functional polymers. J Mater Chem 20(9):1907–1912. https://doi.org/10.1039/B919074K
Mabrouk W, Lafi R, Charradi K, Ogier L, Hafiane A, Fauvarque JF, Sollogoub C (2020) Synthesis and characterization of new proton exchange film deriving sulfonated polyether sulfone using ionic crosslinking electrodialysis applications. Polym Eng Sci 60(12):3149–3158. https://doi.org/10.1002/pen.25543
Charradi K, Ahmed Z, Moussa M, Beji Z, Brahmia A, Othman I, Abu-Haija M, Chtourou R, Keshk SM (2022) A facile approach for the synthesis of zinc ferrite/cellulose as an effective magnetic photocatalyst for the degradation of methylene blue in aqueous solution. Cellulose 29(466):2565–2576. https://doi.org/10.1007/s10570-021-04334-3
Bagheri A, Javanbakht M, Beydaghi H, Salarizadeh P, Shabanikiac A, Amoli HS (2016) Sulfonated poly(ether ether ketone) and sulfonated polyvinylidene fluoride-co-hexafluoropropylene based blend proton exchange films for direct methanol fuel cell applications. RSC Adv 6(45):39500–39510. https://doi.org/10.1039/C6RA00038J
Liang L, Shi Z, Tan X, Sun S, Chen M, Dastan D, Dong B, Cao L (2021) Largely improved breakdown strength and discharge efficiency of layer-structured nanocomposites by filling with a small loading fraction of 2D zirconium phosphate nanosheets. Adv Mater Interfaces 9:2101646. https://doi.org/10.1002/admi.202101646
Zhang M, Shi Z, Zhang J, Zhang K, Lei L, Dastan D, Dong B, Cao L (2021) Greatly enhanced dielectric charge storage capabilities of layered polymer composites incorporated with low loading fractions of ultrathin amorphous iron phosphate nanosheets. J Mater Chem C 9:10414–10424. https://doi.org/10.1039/DITC01974K
Liu Y, Wang J, Zhang H, Ma C, Liu J, Cao S, Zhang X (2014) Enhancement of proton conductivity of chitosan film enabled by sulfonated graphene oxide under both hydrated and anhydrous conditions. J Power Sources 269:898–911. https://doi.org/10.1016/j.jpowsour.2014.07.075
Mabrouk W, Lafi R, Fauvarque JF, Hafiane A, Sollogoub C (2021) New ion exchange film derived from sulfochloratedpolyether sulfone for electrodialysis desalination of brackish water. Polym Adv Technol 32(1):304–314. https://doi.org/10.1002/pat.5086
Altaf F, Ahmed S, Dastan D, Batool R, Rehman ZU, Shi Z, Hameed MU, Bocchetta P, Jacob K (2022) Novel sepiolite reinforced emerging composite polymer electrolyte membranes for high-performance direct methanol fuel cells. Mater Today Chem 24:100843. https://doi.org/10.1016/j.mtchem.2022.100843
Zhu P, Dastan D, Liu L, Wu LK, Shi Z, Chu QQ, Altaf F, Mohammed MKA (2023) Surface wettability of various phases of titania thin films: atomic-scale simulation studies. J Mol Graph Model 118:108335. https://doi.org/10.1016/j.jmgm.2022.108335
Acknowledgements
The authors would like to thank Research and Technology Center of Energy, Technopark Borj Cedria for their support. The authors extend their appreciation to the manager of Eras Labo for providing the SPESOS polymer free of charge.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• An organic-inorganic hybrid film having potential applications in fuel cells was prepared and characterized.
• Graphene improved the contact angle, water uptake, and other physicochemical properties of hybrid films.
• Hybrid film 2% G/SPESOS exhibited proton conductivity augmentation at elevated temperatures and humidity.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mabrouk, W., Jebri, S., Charradi, K. et al. Fabrication and characterization of graphene/sulfonated polyether sulfone octyl sulfonamide hybrid film with improved proton conductivity performance. J Solid State Electrochem 27, 991–999 (2023). https://doi.org/10.1007/s10008-023-05411-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-023-05411-2