Abstract
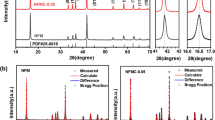
In this work, Cu-doped NaFe0.5Co0.5O2 was easily prepared by a one-step solid-state reaction and studied the effect of copper salt precursors including CuCl2 and Cu(OAc)2 on the structure and electrochemical properties. XRD patterns of the synthesized materials all exhibited diffraction peaks of O3-type layered oxide with high crystallinity and negligible impurity of halide compound. Using Cu(OAc)2 precursor showed a well-defined voltage profile feature of NaFe0.5Co0.5O2 material and a superior performance in sodium half-cell. Doped samples exhibited a capacity of 130 mAh g−1 which is higher than pristine NaFe0.5Co0.5O2 and good capacity retention for 100 cycles. Ex situ XRD results indicated the complex phase transition above 4 V on the first charge, but the structure of the Cu-doped materials remained O3-type layered after the end of cycling. Additionally, Cu-doping effectively enhanced Na+ diffusion coefficients also indicated using the cyclic voltammetry method.






Similar content being viewed by others
References
Nayak PK, Yang L, Brehm W, Adelhelm P (2018) Angew Chem Int Ed 57(1):102–120
Kubota K, Dahbi M, Hosaka T, Kumakura S, Komaba S (2018) Chem Rec 18(4):459–479
Massé RC, Uchaker E, Cao G (2015) Sci China Mater 58(9):715–766
Nitta N, Wu F, Lee JT, Yushin G (2015) Mater Today 18(5):252–264
Wang P-F, You Y, Yin Y-X, Guo Y-G (2018) Adv Energy Mater 8(8):1701912
Ye H, Wang C-Y, Zuo T-T, Wang P-F, Yin Y-X, Zheng Z-J, Wang P, Cheng J, Cao F-F, Guo Y-G (2018) Nano Energy 48:369–376
Tripathi A, Rudola A, Gajjela SR, Xi S, Balaya P (2019) J Mater Chem A 7(45):25944–25960
Guo J-Z, Wang P-F, Wu X-L, Zhang X-H, Yan Q, Chen H, Zhang J-P, Guo Y-G (2017) Adv Mater 29(33):1701968
Deng J, Luo W-B, Lu X, Yao Q, Wang Z, Liu H-K, Zhou H, Dou S-X (2018) Adv Energy Mater 8(5):1701610
Liu J, Yang G, Zhang X, Wang J, Wang R (2012) J Power Sources 197:253–259
Churikov AV, Kachibaya EI, Sycheva VO, Ivanishcheva IA, Imnadze RI, Paikidze TV, Ivanishchev AV (2009) Russ J Electrochem 45(2):175–182
Kim H-S, Kim W-S, Gu H-B, Wang G (2017) J New Mater Electrochem Syst 12:207–212
Cao X, Zhao Y, Zhu L, Xie L, Cao X, Xiong S, Wang C (2016) Int J Electrochem Sci 5267–5278
Li Z-Y, Zhang J, Gao R, Zhang H, Hu Z, Liu X (2016) ACS Appl Mater Interfaces 8(24):15439–15448
Yabuuchi N, Kubota K, Dahbi M, Komaba S (2014) Chem Rev 114(23):11636–11682
Zhou P, Liu X, Weng J, Wang L, Wu X, Miao Z, Zhao J, Zhou J, Zhou S (2019) J Mater Chem A 7(2):657–663
Manikandan P, Heo S, Kim HW, Jeong HY, Lee E, Kim Y (2017) J Power Sources 363:442–449
Yoshida H, Yabuuchi N, Komaba S (2013) Electrochem Commun 34:60–63
Wang X, Tamaru M, Okubo M, Yamada A (2013) J Phys Chem C 117(30):15545–15551
Wang H, Gu M, Jiang J, Lai C, Ai X (2016) J Power Sources 327:653–657
Amaha K, Kobayashi W, Akama S, Mitsuishi K, Moritomo Y (2017) Phys Status Solidi RRL – Rapid Res Lett 11(1):1600284
Van Hoang N, Dai Quang T, Van Man T, My Loan Phung L (2019) Vietnam J Chem 57:302–307
Plewa A, Kulka A, Kondracki Ł, Lu L, Molenda J (2019) J Power Sources 419:42–51
Yao H-R, Wang P-F, Wang Y, Yu X, Yin Y-X, Guo Y-G (2017) Adv Energy Mater 7(15):1700189
Li L, Wang H, Han W, Guo H, Hoser A, Chai Y, Liu X (2018) J Electrochem Soc 165(16):A3854–A3861
Xu S-Y, Wu X-Y, Li Y-M, Hu Y-S, Chen L-Q (2014) Chin Phys B 23(11):118202
Pang W-L, Guo J-Z, Zhang X-H, Fan C-Y (2019) J Alloys Compd 790:1092–1100
Wang L, Sun Y-G, Hu L-L, Piao J-Y, Guo J, Manthiram A, Ma J, Cao A-M (2017) J Mater Chem A 5(18):8752–8761
Ding JJ, Zhou YN, Sun Q, Yu XQ, Yang XQ, Fu ZW (2013) Electrochim Acta 87:388–393
Zhao J, Zhao L, Dimov N, Okada S, Nishida T (2013) J Electrochem Soc 160(5):A3077–A3081
Hoang NV, Quang TD, Man TV, Phung LML (2019) Vietnam J Chem 4E12:302–307
Delmas C, Fouassier C, Hagenmuller P (1980) Physica 99B:81–85
Kubota K, Asari T, Yoshida H, Yaabuuchi N, Shiiba H, Nakayama M, Komaba S (2016) Adv Funct Mater 26(33):6047–6059
Lei Y, Li X, Liu L, Ceder G (2014) Chem Mater 26(18):5288–5296
Hoang NV, Phuoc MN, Nguyen HLT, Quan ND (2019) Vietnam J Sci Technol 57(2):198–206
Ramasamy HV, Kaliyappan K, Tangavel R, Aravindan V, Kang K, Kim DU, Sun X, Lee Y-S (2017) J Mater Chem A
Mason CW, Lange F, Saravanan K, Lin F, Nordlund D (2015) ECS Electrochem Lett 4(5):A41–A44
Mu L, Xu S, Li Y, Hu Y-S, Li H, Chen L, Huang X (2015) Adv Mater 27(43):6928–6933
Wang P-F, You Y, Yin Y-X, Guo Y-G (2016) J Mater Chem A 4(45):17660–17664
Zhu Q, Nan B, Shi Y, Zhu Y, Wu S, He L, Deng Y, Wang L, Chen Q, Lu Z (2017) J Solid State Electrochem 21(10):2985–2995
Guo S, Sun Y, Yi J, Zhu K, Liu P, Zhu Y, Zhu G-Z, Chen M, Ishida M, Zhou H (2016) NPG Asia Mat 8(4):e266
Clément RJ, Bruce PG, Grey CP (2015) J Electrochem Soc 162(14):A2589–A2604
Yabuuchi N, Komaba S (2014) Sci Technol Adv Mater 15(4):043501
Di Lecce D, Campanella D, Hassoun J (2018) J Phys Chem C 122(42):23925–23933
Rong X, Qi X, Lu Y, Wang Y, Li Y, Jiang L, Yang K, Gao F, Huang X, Chen L, Hu Y-S (2019) J Energy Chem 31:132–137
Funding
This work was funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.06-2018.359.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• High crystallize Cu-doped NaCu0.05Fe0.45Co0.5O2 samples were synthesized using halide and acetate as copper precursors.
• The acetate-based precursor (NFCCu-Ac) enables high purity and better performance with the capacity of 130 mAhg−1 and stable cycling over 100 cycles.
• Phase structure at high potential and Na+-ion diffusion coefficient of the NFCCu-Ac sample were explored.
Rights and permissions
About this article
Cite this article
Nguyen, V.H., Le Nguyen, M., Tran, H.P. et al. Cu-doped NaCu0.05Fe0.45Co0.5O2 as promising cathode material for Na-ion batteries: synthesis and characterization. J Solid State Electrochem 25, 767–775 (2021). https://doi.org/10.1007/s10008-020-04851-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-020-04851-4