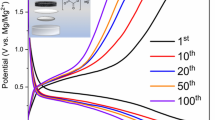
Abstract
Functional compatible electrolyte with Mg2+ intercalation cathodes represents one of the largest obstacles in the development of practical Mg batteries MBs. In current work, we report for the first time magnesium hexakis(methanol)-dinitrate complex (MHMD) electrolyte product reaction of 2,2-dimethoxypropane with magnesium nitrate hexahydrate via ‘Solvent-in-Salt’ method. 2,2-Dimethoxypropane as a water scavenger can capture reducible molecules like H2O and dehydrate Mg(NO3)2.6H2O to form magnesium hexakis(methanol)-dinitrate complex. Meanwhile, Mg cloud bonds will become weak—something which frees up the mobility of Mg2+. This electrolyte exhibits high ionic conductivity with low activation energy ~ 0.18 eV. The general aim of the investigation was to demonstrate a potential application of MHMD electrolyte in Mg-ion cell. Mg cells were analyzed with the use of cyclic voltammetry (CV), galvanostatic charging/discharging tests, and electrochemical impedance spectroscopy. A comparative study between different cathodes like V2O5, GeO2, TiO2, and S using MHMD electrolyte was performed. The S cathode has an initial discharge capacity of 370 mAh g−1 and retained a reversible capacity at 60 mAh g−1 after 20 cycles exhibiting better electrochemical performances than those of V2O5, GeO2, and TiO2 cathodes. This work opens up a new pathway to explore new electrolytic materials for MBs with high performance.







Similar content being viewed by others
References
Kang S-J, Lim S-C, Kim H, Heo JW, Hwang S, Jang M, Yang D, Hong S-T, Lee H (2017) Non-Grignard and Lewis acid-free sulfone electrolytes for rechargeable magnesium batteries. Chem Mater 29(7):3174–3180
Shterenberg I, Salama M, Gofer Y, Levi E, Aurbach D (2014) The challenge of developing rechargeable magnesium batteries. MRS Bull 39(5):453–460
Tutusaus O, Mohtadi R, Arthur TS, Mizuno F, Nelson EG, Sevryugina YV (2015) An efficient halogen-free electrolyte for use in rechargeable magnesium batteries. Angew Chem Int Ed 54(27):7900–7904
Aurbach D, Lu Z, Schechter A, Gofer Y, Gizbar H, Turgeman R, Cohen Y, Moshkovich M, Levi E (2000) Prototype systems for rechargeable magnesium batteries. Nature 407(6805):724–727
Du A, Zhang Z, Qu H, Cui Z, Qiao L, Wang L, Chai J, Lu T, Dong S, Dong T (2017) An efficient organic magnesium borate-based electrolyte with non-nucleophilic characteristics for magnesium–sulfur battery. Energy Environ Sci 10(12):2616–2625
Samuel D, Steinhauser C, Smith JG, Kaufman A, Radin MD, Naruse J, Hiramatsu H, Siegel DJ (2017) Ion pairing and diffusion in magnesium electrolytes based on magnesium borohydride. ACS Appl Mater Interfaces 9(50):43755–43766
Mohtadi R, Matsui M, Arthur TS, Hwang SJ (2012) Magnesium borohydride: from hydrogen storage to magnesium battery. Angew Chem Int Ed 51(39):9780–9783
Zhao-Karger Z, Bardaji MEG, Fuhr O, Fichtner M (2017) A new class of non-corrosive, highly efficient electrolytes for rechargeable magnesium batteries. J Mater Chem A 5(22):10815–10820
Ha S-Y, Lee Y-W, Woo SW, Koo B, Kim J-S, Cho J, Lee KT, Choi N-S (2014) Magnesium (II) bis (trifluoromethane sulfonyl) imide-based electrolytes with wide electrochemical windows for rechargeable magnesium batteries. ACS Appl Mater Interfaces 6(6):4063–4073
Yoo HD, Liang Y, Dong H, Lin J, Wang H, Liu Y, Ma L, Wu T, Li Y, Ru Q (2017) Fast kinetics of magnesium monochloride cations in interlayer-expanded titanium disulfide for magnesium rechargeable batteries. Nat Commun 8(1):339
Mao M, Yan F, Cui C, Ma J, Zhang M, Wang T, Wang C (2017) Pipe-wire TiO2–Sn@ carbon nanofibers paper anodes for lithium and sodium ion batteries. Nano Lett 17(6):3830–3836
McNulty D, Geaney H, Buckley D, O'Dwyer C (2018) High capacity binder-free nanocrystalline GeO2 inverse opal anodes for Li-ion batteries with long cycle life and stable cell voltage. Nano Energy 43(Supplement C):11–21
Su D, Wang G (2013) Single-crystalline bilayered V2O5 nanobelts for high-capacity sodium-ion batteries. ACS Nano 7(12):11218–11226
Li Z, Guan BY, Zhang J, Lou XWD (2017) A compact nanoconfined sulfur cathode for high-performance lithium-sulfur batteries. Joule 1(3):576–587
Chang C-C, Her L-J, Chen L-C, Lee Y-Y, Liu S-J, Tien H-J (2007) 2, 2-Dimethoxy-propane as electrolyte additive for lithium-ion batteries. J Power Sources 163(2):1059–1063
Critchfield F, Bishop E (1961) Water determination by reaction with 2, 2-dimethoxypropane. Anal Chem 33(8):1034–1035
Sa N, Wang H, Proffit DL, Lipson AL, Key B, Liu M, Feng Z, Fister TT, Ren Y, Sun C-J (2016) Is alpha-V 2 O 5 a cathode material for Mg insertion batteries? J Power Sources 323:44–50
Sinha NN, Munichandraiah N (2008) Electrochemical conversion of LiMn2O4 to MgMn2O4 in aqueous electrolytes. Electrochem Solid-State Lett 11(11):F23–F26
Zhang H, Ye K, Shao S, Wang X, Cheng K, Xiao X, Wang G, Cao D (2017) Octahedral magnesium manganese oxide molecular sieves as the cathode material of aqueous rechargeable magnesium-ion battery. Electrochim Acta 229:371–379
van Ingen Schenau A, Groeneveld W, Reedijk J (1972) Alcohols as ligands. Part II: metal (II) salts containing coordinated methanol. Recueil des Travaux Chimiques des Pays-Bas 91(1):88–94
Al-Abadleh HA, Grassian V (2003) Phase transitions in magnesium nitrate thin films: a transmission FT-IR study of the deliquescence and efflorescence of nitric acid reacted magnesium oxide interfaces. J Phys Chem B 107(39):10829–10839
Sulaiman M, Rahman A, Mohamed N (2013) Structural, thermal and conductivity studies of magnesium nitrate–alumina composite solid electrolytes prepared via sol-gel method. Int J Electrochem Sci 8:6647–6655
Natal-Santiago M, Dumesic J (1998) Microcalorimetric, FTIR, and DFT studies of the adsorption of methanol, ethanol, and 2, 2, 2-trifluoroethanol on silica. J Catal 175(2):252–268
Chen L, Fan LZ (2018) Dendrite-free Li metal deposition in all-solid-state lithium sulfur batteries with polymer-in-salt polysiloxane electrolyte. Energy Storage Mater 15:37–45
Zhang M, MacRae AC, Liu H, Meng YS (2016) Communication—investigation of anatase-TiO2 as an efficient electrode material for magnesium-ion batteries. J Electrochem Soc 163(10):A2368–A2370
Chen L, Li Y, Li S-P, Fan L-Z, Nan C-W, Goodenough JB (2017) PEO/garnet composite electrolytes for solid-state lithium batteries: from “ceramic-in-polymer” to “polymer-in-ceramic”. Nano Energy 46:176–184
Tan C, Farhana N, Saidi NM, Ramesh S, Ramesh K (2018) Conductivity, dielectric studies and structural properties of P (VA-co-PE) and its application in dye sensitized solar cell. Org Electron 56:116–124
Panero S, Scrosati B, Sumathipala H, Wieczorek W (2007) Dual-composite polymer electrolytes with enhanced transport properties. J Power Sources 167(2):510–514
Zhao-Karger Z, Zhao X, Wang D, Diemant T, Behm RJ, Fichtner M (2015) Performance improvement of magnesium sulfur batteries with modified non-nucleophilic electrolytes. Adv Energy Mater 5 (3)
Vinayan B, Zhao-Karger Z, Diemant T, Chakravadhanula VSK, Schwarzburger NI, Cambaz MA, Behm RJ, Kübel C, Fichtner M (2016) Performance study of magnesium–sulfur battery using a graphene based sulfur composite cathode electrode and a non-nucleophilic Mg electrolyte. Nano 8(6):3296–3306
Li X, Yang Z, Fu Y, Qiao L, Li D, Yue H, He D (2015) Germanium anode with excellent lithium storage performance in a germanium/lithium–cobalt oxide lithium-ion battery. ACS Nano 9(2):1858–1867
Zhang Z, Cui Z, Qiao L, Guan J, Xu H, Wang X, Hu P, Du H, Li S, Zhou X (2017) Novel design concepts of efficient mg-ion electrolytes toward high-performance magnesium–selenium and magnesium–sulfur batteries. Adv Energy Mater 7(11):1602055–1602065
Zhou X, Tian J, Hu J, Li C (2018) High rate magnesium–sulfur battery with improved cyclability based on metal–organic framework derivative carbon host. Adv Mater 30(7):1704166–1704172
Zeng L, Wang N, Yang J, Wang J, NuLi Y (2017) Application of a sulfur cathode in nucleophilic electrolytes for magnesium/sulfur batteries. J Electrochem Soc 164(12):A2504–A2512
Rui X, Lu Z, Yu H, Yang D, Hng HH, Lim TM, Yan Q (2013) Ultrathin V 2 O 5 nanosheet cathodes: realizing ultrafast reversible lithium storage. Nano 5(2):556–560
Hou J, Wu R, Zhao P, Chang A, Ji G, Gao B, Zhao Q (2013) Graphene–TiO2 (B) nanowires composite material: synthesis, characterization and application in lithium-ion batteries. Mater Lett 100:173–176
Rashad M, Zhang H, Asif M, Feng K, Li X, Zhang H (2018) Low cost room temperature synthesis of NaV3O8. 1.69 H2O Nanobelts for Mg batteries. ACS Appl Mater Interfaces 10(5):4757–4766
Yuan Z, Jiang Q, Feng C, Chen X, Guo Z (2017) Synthesis and performance of tungsten disulfide/carbon (WS2/C) composite as anode material. J Electron Mater 47:251–261
Ihsan M, Wang H, Majid SR, Yang J, Kennedy SJ, Guo Z, Liu HK (2016) MoO 2/Mo 2 C/C spheres as anode materials for lithium ion batteries. Carbon 96:1200–1207
Zhang H, Ye K, Zhu K, Cang R, Yan J, Cheng K, Wang G, Cao D (2017) The FeVO4· 0.9 H2O/graphene composite as anode in aqueous magnesium ion battery. Electrochim Acta 256:357–364
Funding
This work is partial financially supported by the Support Development of Scientific Research Centre of Benha University (SDSRC) (Grant No. 1076) and Science Technology Development Fund (Grant No. 12564).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sheha, E., El-Deftar, M. Magnesium hexakis(methanol)-dinitrate complex electrolyte for use in rechargeable magnesium batteries. J Solid State Electrochem 22, 2671–2679 (2018). https://doi.org/10.1007/s10008-018-3986-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-018-3986-z