Abstract
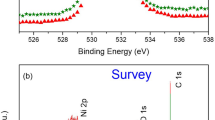
Sulfur-induced degradation of the passive film on Alloy 800 in a simulated alkaline crevice (AKC) environment at 300 °C was evaluated using electrochemical impedance spectroscopy (EIS), scanning electron microscopy (SEM), secondary ion mass spectrometry (SIMS), X-ray photoelectron spectroscopy (XPS), and Auger electron spectroscopy (AES). The reduced sulfur species (mainly S−2) from thiosulfate was incorporated into the passive film, increasing the Ni/Cr ratio in the film. Film thickness was reduced from 700 nm in an alkaline crevice solution without thiosulfate to 400 nm in an AKC solution with thiosulfate. The results from EIS modeling using an electrochemical equivalent circuit (EEC) and point defect model (PDM) also confirmed that reduced sulfur species increased the dissolution rate of the passive layer.









Similar content being viewed by others
References
Xia DH, Luo JL (2015) Acta Phys Sin 31:467–475
Luo B, Xia DH (2014) Acta Phys Chim Sin 30:59–66
Xia DH, Zhou C, Liu YH, Wang JH, Fu CW, Wang K, Li M (2013) Electrochemistry 81:262–268
Grant IS, Andrews PJD (1999) Br Med J 319:110–113
Lu BT, Luo JL, Lu YC (2008) Electrochim Acta 53:4122–4136
Staehle RW, Gorman JA (2004) Corrosion 60:115–180
Tsai W-T, Wu T-F (2000) J Nucl Mater 277:169–174
Kappes M, Frankel GS, Sridhar N, Carranza RM (2012) J Electrochem Soc 159:C195
Tsujikawa S, Miyasaka A, Ueda M, Ando S, Shibata T, Haruna T, Katahira M, Yamane Y, Aoki T, Yamada T (1993) Corrosion 49:409–419
Hemmingsen T (1992) Electrochim Acta 37:2775–2784
Schmitt G (1991) Corrosion 47:285–308
Marcus P, Protopopoff E (1997) Corros Sci 39:1741–1752
Bentz RW, Neville HA (1949) J Polym Sci 4:673–688
Jung RH, Tsuchiya H, Fujimoto S (2012) Corros Sci 58:62–68
Hamzah RY, Eltorkey NM (1995) Arab Gulf J Sci Res 13:13–23
Zuili D, Maurice V, Marcus P (1999) J Phy Chem B 103:7896–7905
Hai NTM, Huemann S, Hunger R, Jaegermann W, Wandelt K, Broekmann P (2007) J Phy Chem C 111:14768–14781
Zhang X, Zagidulin D, Shoesmith DW (2013) Electrochim Acta 89:814–822
Lewandowski BR, Lytle DA, Garno JC (2010) Langmuir 26:14671–14679
Oblonsky LJ, Davenport AJ, Ryan MP, Isaacs HS, Newman RC (1997) J Electrochem Soc 144:2398–2404
Magnussen OM, Scherer J, Ocko BM, Behm RJ (2000) J Phys Chem B 104:1222–1226
Zhao R, Zhang Z, Shi JB, Tao L, Song SZ (2010) J Cent South Univ Technol 17:13–18
Neville HA, Theis ER, K'Burg RB (1930) Ind Eng Chem 22:57–60
Neville HA, Theis ER, Oswald CT (1930) Ind Eng Chem 22:60–62
Guo HX, Lu BT, Luo JL (2006) Electrochim Acta 52:1108–1116
de Oliveira MCL, Pereira VSM, Correa OV, de Lima NB, Antunes RA (2013) Corros Sci 69:311–321
Zhu RK, Luo JL (2010) Electrochem Commun 12:1752–1755
Zhu RK, Lu BT, Luo JL, Lu YC (2013) Appl Surf Sci 270:755–762
Lu BT, Luo JL, Lu YC (2013) Electrochim Acta 87:824–838
Dukstiene N, Sinkeviciute D (2013) J Solid State Electrochem 17:1175–1184
Hakiki NE (2011) Corros Sci 53:2688–2699
Marchetti L, Perrin S, Wouters Y, Martin F, Pijolat M (2010) Electrochim Acta 55:5384–5392
Li DG, Wang JD, Chen DR (2012) Electrochim Acta 60:134–146
Andrews PLR (1999) Dig Dis Sci 44:31s–38s
Andrews PC, Hardie MJ, Raston CL (1999) Coord Chem Rev 189:169–198
Hoog CO, Birbilis N, Estrin Y (2008) Adv Eng Mater 10:579–582
Graham AC, Reid MM, Andrews PJD (1999) Anaesthesia 54:814–815
Xia DH, Song SZ, Wang JH, Bi HC, Han ZW (2012) Trans Tianjin Univ 18:15–20
Zhang Y, Urquidi-Macdonald M, Engelhardt GR, Macdonald DD (2012) Electrochim Acta 69:1–11
Zhang Y, Urquidi-Macdonald M, Engelhardt GR, Macdonald DD (2012) Electrochim Acta 69:12–18
Zhang Y, Urquidi-Macdonald M, Engelhardt GR, Macdonald DD (2012) Electrochim Acta 69:19–29
Lu Z, Macdonald DD (2008) Electrochim Acta 53:7696–7702
Macdonald DD, Sun A (2006) Electrochim Acta 51:1767–1779
Shao HB, Wang JM, He WC, Zhang JQ, Cao CN (2005) Electrochem Commun 7:1429–1433
Shao HB, Wang JM, Wang XY, Zhang JQ, Cao CN (2004) Electrochem Commun 6:6–9
Xia DH, Song SZ, Zhu RK, Behnamian Y, Shen C, Wang JH, Luo JL, Lu YC, Klimas S (2013) Electrochim Acta 111:510–525
Seyeux A, Maurice V, Marcus P (2013) J Electrochem Soc 160:C189–C196
Leistner K, Toulemonde C, Diawara B, Seyeux A, Marcus P (2013) J Electrochem Soc 160:C197–C205
Xia D-H, Zhu R-K, Behnamian Y, Shen C, Luo J-L, Lu Y-C, Klimas S (2014) J Electrochem Soc 161:C201–C214
Huang J, Wu X, Han E-H (2009) Corros Sci 51:2976–2982
Huang J, Wu X, Han E-H (2010) Corros Sci 52:3444–3452
Lu BT, Luo JL, Lu YC (2007) J Electrochem Soc 154:C379
Sharifi-Asl S, Taylor ML, Lu Z, Engelhardt GR, Kursten B, Macdonald DD (2013) Electrochim Acta 102:161–173
Macdonald DD (2012) Russ J Electrochem 48:235–258
Macdonald DD (2011) Electrochim Acta 56:1761–1772
Sharifi-Asl S, Macdonald DD, Almarzooqi A, Kursten B, Engelhardt GR (2013) J Electrochem Soc 160:C316–C325
Liu XH, Wu XQ, Han EH (2013) Electrochim Acta 108:554–565
Zhou C, Wang J, Song S, Xia D, Wang K, Shen C, Luo B, Shi J (2013) J Wuhan Univ Technol Mater Sci Ed 28:367–372
Hsu CH, Mansfeld F (2001) Corrosion 57:747–748
Xia DH, Song SZ, Wang JH, Bi HC, Han ZW (2012) Trans Nonferrous Metals Soc China 22:717–724
Zheng X, Xia D, Wang H, Fu C (2013) Anti Corros Methods Mater 60:153–159
Zhou C, Wang JH, Song SZ, Xia DH, Wang K, Shen C, Luo B, Shi JB (2013) J Wuhan Univ Technol Mater Sci Ed 28:367–372
Wang J, Fu C, Gao Z, Xia D (2013) Trans Tianjin Univ 19:235–240
Xia D, Wang J, Jiang Y, Li N, Zhou C (2013) J Tianjin Univ 46:503–509
Staehle RW, Gorman JA (2003) Corrosion 59:931–994
Staehle RW, Gorman JA (2004) Corrosion 60:5–63
Macdonald DD, Sharifi-Asl S (2014) Corros Sci 81:102–109
Mao F, Sharifi-Asl S, Yu J, Macdonald DD (2014) J Electrochem Soc 161:C254–C260
Kuang W, Mathews JA, Taylor ML, Macdonald DD (2014) Electrochim Acta 136:493–503
Zhang Y, Macdonald DD, Urquidi-Macdonald M, Engelhardt GR, Dooley RB (2006) Corros Sci 48:3812–3823
Acknowledgments
This work was supported by AECL and Natural Science Foundation of Tianjin (No. 14JCYBJC1770). We acknowledge the support of Dr. R.L. Tapping and Dr. P. Angell of AECL. The authors thank Dr. Anqiang He, Dr. Shihong Xu, and Dr. Dimitre Karpuzov of the Alberta Center for Surface Engineering and Science, University of Alberta, for performing AES, SEM, XPS and SIMS analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xia, DH., Behnamian, Y., Chen, XY. et al. A mechanistic study of sulfur-induced passivity degradation of Alloy 800 in a simulated alkaline crevice environment at 300 °C. J Solid State Electrochem 19, 3567–3578 (2015). https://doi.org/10.1007/s10008-015-2822-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-015-2822-y