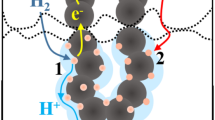
Abstract
This paper reports the development of mathematical model that bridges PEMFC transport phenomena with surface reaction kinetics in rough and heterogeneous electrodes, in which specifically the anode is modeled under carbon monoxide contamination. The mathematical bridging is done by converting surface concentration of reactants and contaminant into surface coverage of relevant adsorbates using the Langmuir-Freundlich isotherm to statistically include heterogeneity in binding site energetics. Thermodynamically optimized kinetic rate and equilibrium constants are calculated using coverage-dependent activation energies and provided as input to the HOR, ORR, and a kinetic-based limiting current model developed to solve for the distribution of activation overpotential. A novel model for a closed-form calculation of activation overpotential is proposed to allow numerical investigation via a galvanostatic approach. The kinetic reaction models are highly coupled with three-dimensional transport equations and solved iteratively under single-phase and steady-state conditions. Comparison is done with respect to two sets of available literature data in order to test the kinetic model validity under variation of CO concentrations and cell operating temperatures, in which good agreement is found. The results confirm that a Langmuir-Freundlich isotherm could be a more suitable isotherm compared to the extensively used Langmuir-specific isotherm for rough heterogeneous surfaces physically found in PEMFC catalysts. Results from the model show that rate of HOR is higher under the ribs of the cell since most of the active sites under channel are blocked with CO, with the effect exacerbated at lower temperatures.








Similar content being viewed by others
Abbreviations
- a :
-
Water activity
- c :
-
Concentration, mol m−3
- c p :
-
Specific heat, J kg−1 K−1 or J mol−1 K−1
- A :
-
Area, m2
- D :
-
Diffusion coefficient, m2 s−1
- \( \widehat{D} \) :
-
Pseudo-diffusion coefficient, m2 s−1
- E :
-
Electrode potential, V
- \( {\tilde{E}}_{\mathrm{a}} \) :
-
Activation energy, J mol−1
- F :
-
Faraday’s constant, 96,485 C mol−1
- h :
-
Heterogeneity index
- i :
-
Local current density based on geometric area, A m−2
- \( \overline{i} \) :
-
Local current density based on Pt area, A m − 2Pt
- j :
-
Volumetric local current density, A m−3
- k :
-
Thermal conductivity, W m−1 K−1
- k f :
-
Forward rate constant, first order, m3 mol−1 s−1, second order, m5 mol−2 s−1
- k b :
-
Backward rate constant, first order, s−1, second order, m2 mol−1 s−1
- K :
-
Equilibrium constant, H, CO, O, and H2O adsorption, kPa−1, H2O desorption, kPa
- L Pt :
-
Catalyst loading, mgPt cm−2
- K p :
-
Hydraulic permeability, m2
- M :
-
Molecular weight, kg mol−1
- P :
-
Pressure, Pa
- r f :
-
Roughness factor
- R :
-
Universal gas constant, 8.314 J mol−1 K−1
- R ohmic :
-
Ohmic resistance, Ω m2
- s 0 :
-
Sticking coefficient
- S :
-
Source term
- \( \widehat{S} \) :
-
Pseudo-source term
- ΔS :
-
Entropy change, J mol−1 K−1
- T :
-
Temperature, K
- u :
-
Velocity, m s−1
- X :
-
Mole fraction
- \( \widehat{X} \) :
-
Pseudo-mole fraction
- x, y, z :
-
Cartesian coordinates, m
- Γ:
-
Site density, mol m−2
- α :
-
Symmetry coefficient
- β :
-
Temperature exponent, K
- ε :
-
Porosity
- η :
-
Overpotential, V
- θ :
-
Surface coverage
- λ :
-
Hydration
- μ :
-
Dynamic viscosity, Pa s
- ρ :
-
Density, kg m−3
- τ :
-
Tortuosity
- σ :
-
Conductivity, S m−1
- φ :
-
Relative humidity, %
- ϕ :
-
Local potential, V
- 0:
-
Standard
- a:
-
Anode
- c:
-
Cathode
- eff:
-
Effective
- geom:
-
geometric
- i, j :
-
Species i or j
- in:
-
Inlet
- M:
-
Ionomer phase
- r:
-
Reactive
- rev:
-
reversible
- s:
-
solid phase
References
Li X (2006) Principles of fuel cells. Taylor & Francis, New York
Gottesfeld S, Pafford J (1988) J Electrochem Soc 135(10):2651–2652
Somorjai GA (2010) Introduction to surface chemistry and catalysis. Wiley, New Jersey
Gilman S (1964) J Phys Chem 68(1):70–80
Hasmady S, Hatakeyama T, Wacker MP, Fushinobu K, Okazaki K (2009) Therm Sci Eng 17:147–156
Springer TE, Rockward T, Zawodzinski TA, Gottesfeld S (2001) J Electrochem Soc 148(1):A11–A23
Camara GA, Ticianelli EA, Mukerjee S, Lee SJ, McBreen J (2002) J Electrochem Soc 149(6):A748–A53
Bhatia KK, Wang C-Y (2004) Electrochim Acta 49:2333–2341
Baschuk JJ, Li X (2003) Int J Energy Res 27:1095–1116
Zhou T, Liu H (2004) J Power Sources 138:101–110
Ju H, Lee KS, Um S (2008) J Mech Sci Technol 22:991–998
Siegel C (2008) Energy 33:1331–1352
Shah AA, Luo KH, Ralph TR, Walsh FC (2011) Electrochim Acta 56:3731–3757
Franco AA, Schott P, Jallut C, Maschke B (2006) J Electrochem Soc 153(6):A1053–A1061
Wei ZD, Ran HB, Liu XA, Liu Y, Sun CX, Chan SH, Shen PK (2006) Electrochim Acta 51:3091–3096
Hartnig C, Koper MTM (2002) J Electroanal Chem 532:165–170
Mazumder S, Cole JV (2003) J Electrochem Soc 150(11):A1503–A1509
Nguyen PT, Berning T, Djilali N (2004) J Power Sources 130:149–157
Ju H, Meng H, Wang C-Y (2005) Int J Heat Mass Transfer 48:1303–1315
Hwang JJ (2006) J Electrochem Soc 153(2):A216–A224
Weber AZ, Newman J (2004) Chem Rev 104:4679–4726
Wang C-Y (2004) Chem Rev 104:4727–4766
Yi JS, Nguyen TV (1999) J Electrochem Soc 146(1):38–45
Berning T, Djilali N (2003) J Electrochem Soc 150(12):A1589–A1598
Lum KW, McGuirk JJ (2005) J Power Sources 143:103–124
Khajeh-Hosseini-Dalasm N, Fushinobu K, Okazaki K (2010) J Electrochem Soc 157(10):B1358–B1369
Oetjen HF, Schmidt VM, Stimming U, Trila F (1996) J Electrochem Soc 143(12):3838–3842
Lee SJ, Mukerjee S, Ticianelli EA, McBreen J (1999) Electrochim Acta 44:3283–3293
Reid RC, Sherwood TK (1958) The properties of gases and liquids. McGraw-Hill, New York
Hirschfielder JO, Curtis CF, Bird RB (1954) Molecular theory of gases and liquids. Wiley, New York
Bird RB, Stewart WE, Lightfoot EN (2002) Transport phenomena. Wiley, New York
Taylor R, Krishna R (1993) Multicomponent mass transfer. Wiley, New York
Springer TE, Zawodzinski TA, Gottesfeld S (1991) J Electrochem Soc 138(8):2334–2342
Zamel N (2011) Transport properties of the gas diffusion layer of PEM fuel cells, PhD Thesis, University of Waterloo, Ontario, Canada
Masuda M (2003) Hydrogen-based integrated energy system for building equipment, PhD Thesis (in Japanese), Tokyo Institute of Technology, Tokyo, Japan
Japan Society of Mechanical Engineers (1986) JSME data book: heat transfer. Maruzen, Tokyo
Gasteiger HA, Gu W, Makharia R, Mathias MF, Sompalli B (2003) Handbook of fuel cells. In: Vielstich W, Lamm A, Gasteiger HA (eds) Fuel cell technology & applications, vol 3. Wiley, West Sussex, pp 593–610
Markovic NM, Grgur BN, Lucas CA, Ross PN (1999) J Phys Chem B 103:487–495
Koper MTM, Jansen APJ, van Santen RA, Lukkien JJ, Hilbers PAJ (1998) J Chem Phys 109:6051–6062
Breiter MW (2003) Handbook of fuel cells. In: Vielstich W, Lamm A, Gasteiger HA (eds) Electrocatalysis, vol 2. Wiley, West Sussex
Umpleby RJ, Baxter SC, Chen Y, Shah RN, Shimizu KD (2001) Anal Chem 73(19):4584–4591
Sips R (1948) J Chem Phys 16(5):490–495
de Becdelièvre AM, de Becdelièvre J, Clavilier J (1990) J Electroanal Chem 294:97–100
Balasubramanian S, Lakshmanan B, Hetzke CE, Sethuraman VA, Weidner JW (2011) Electrochim Acta 58:723–728
Hauptmann W, Votsmeiera M, Vogel H, Vlachos DG (2011) Appl Catal A Gen 397:174–182
Kee RJ, Coltrin ME, Glarborg P (2003) Chemically reacting flow: theory & practice. Wiley, New Jersey
Zhdanov VP, Kasemo B (2006) Electrochem Commun 8:1132–1136
Gurau V, Liu H, Kakac S (1998) AIChE J 44(11):2410–2422
Benziger J, Kimball E, Mejia-Ariza R, Kevrekidis I (2011) AIChE J 57(9):2505–2517
Nitta I, Hottinen T, Himanen O, Mikkola M (2007) J Power Sources 171:26–36
Gurau B, ELAT® Gas Diffusion Layers. http://fuelcellsetc.com/store/DS/ELAT-Property-Sheet.pdf. Accessed 26 November 2013
Pasaogullari U, Mukherjee PP, Wang C-Y, Chen KS (2007) J Electrochem Soc 154(8):B823–B834
Lampinen MJ, Fomino M (1993) J Electrochem Soc 140(12):3537–3546
Ralph TR, Hogarth MP (2002) Platin Met Rev 46(3):117–135
Acknowledgment
Part of this work was supported by the Grant-in-Aid for Scientific Research from MEXT/JSPS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasmady, S., Fushinobu, K. Inclusion of surface heterogeneity in bridging PEM fuel cell electrode contamination kinetics and transport via a competitive Langmuir-Freundlich isotherm. J Solid State Electrochem 18, 3387–3405 (2014). https://doi.org/10.1007/s10008-014-2521-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2521-0